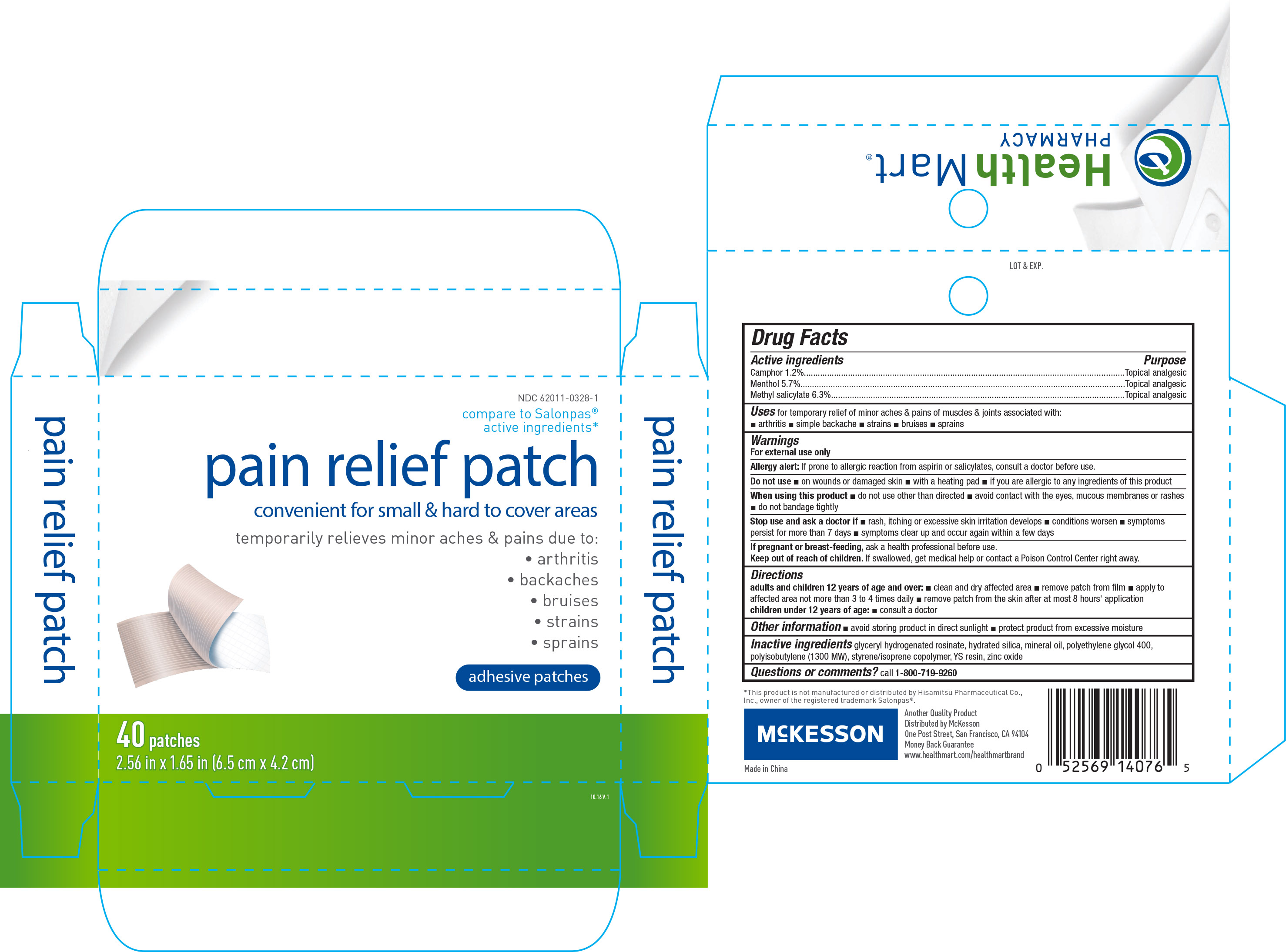
Active ingredients Purpose
Camphor 1.2%.........................................................Topical analgesic
Menthol 5.7%...........................................................Topical analgesic
Methyl salicylate 6.3%...............................................Topical analgesic
Uses
for temporary relief of minor aches & pains of muscles & joints associated with:
- arthritis
- simple backache
- strains
- bruises
- sprains
Warnings
For external use only
Allergy alert: If prone to allergic reaction from aspirin or salicylates, consult a doctor before use.
Do not use
- on wounds or damaged skin
- with a heating pad
- if you are allergic to any ingredients of this product
Stop use and ask a doctor if
- rash, itching or excessive skin irritation develops
- conditions worsen
- smptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
adults and children 12 years of age and over:
- clean and dry affected area
- remove patch from film
- apply to affected area not more than 3 to 4 times daily
- remove patch from the skin after at most 8 hours' application
children under 12 years of age:
- consult a doctor
Other information
- avoid storing product in direct sunlight
- protect product from excessive moisture