FULL PRESCRIBING INFORMATION
WARNING: SERIOUS AND SOMETIMES FATAL INFECTIONS OR BLEEDING
Serious and sometimes fatal infections and bleeding occur very rarely following spontaneous, surgical, and medical abortions, including following Mifepristone tablets, 200 mg use. No causal relationship between the use of Mifepristone tablets, 200 mg and misoprostol and these events has been established.
- Atypical Presentation of Infection. Patients with serious bacterial infections (e.g., Clostridium sordellii) and sepsis can present without fever, bacteremia, or significant findings on pelvic examination following an abortion. Very rarely, deaths have been reported in patients who presented without fever, with or without abdominal pain, but with leukocytosis with a marked left shift, tachycardia, hemoconcentration, and general malaise. A high index of suspicion is needed to rule out serious infection and sepsis [see Warnings and Precautions ( 5.1)]
- Bleeding. Prolonged heavy bleeding may be a sign of incomplete abortion or other complications and prompt medical or surgical intervention may be needed. Advise patients to seek immediate medical attention if they experience prolonged heavy vaginal bleeding [see Warnings and Precautions ( 5.2)].
Because of the risks of serious complications described above, Mifepristone tablets, 200mg is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Mifepristone REMS Program [see Warnings and Precautions ( 5.3)].
Before prescribing Mifepristone, inform the patient about the risk of these serious events. Ensure that the patient knows whom to call and what to do, including going to an Emergency Room if none of the provided contacts are reachable, if they experience sustained fever, severe abdominal pain, prolonged heavy bleeding, or syncope, or if they experience abdominal pain or discomfort, or general malaise (including weakness, nausea, vomiting or diarrhea) for more than 24 hours after taking misoprostol.
1 INDICATIONS AND USAGE
Mifepristone tablets, 200 mg is indicated, in a regimen with misoprostol, for the medical termination of intrauterine pregnancy through 70 days gestation.
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Regimen
For purposes of this treatment, pregnancy is dated from the first day of the last menstrual period. The duration of pregnancy may be determined from menstrual history and clinical examination. Assess the pregnancy by ultrasonographic scan if the duration of pregnancy is uncertain or if ectopic pregnancy is suspected.
Remove any intrauterine device (“IUD”) before treatment with Mifepristone tablets, 200mg begins [see
Contraindications (
4)].
The dosing regimen for Mifepristone tablets, 200 mg and misoprostol is:
Mifepristone 200 mg orally + misoprostol 800 mcg buccally
• Day One: Mifepristone 200 mg Administration
One 200 mg tablet of Mifepristone is taken in a single oral dose.
• Day Two or Three: Misoprostol Administration (minimum 24-hour interval between, Mifepristone and misoprostol)
Four 200 mcg tablets (total dose 800 mcg) of misoprostol are taken by the buccal route.
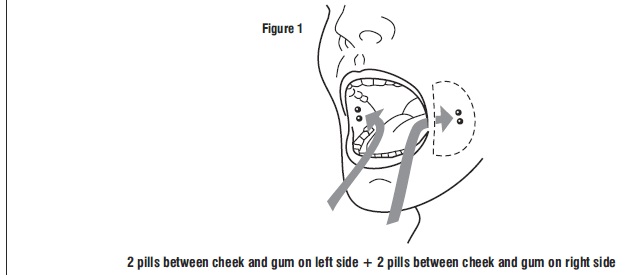
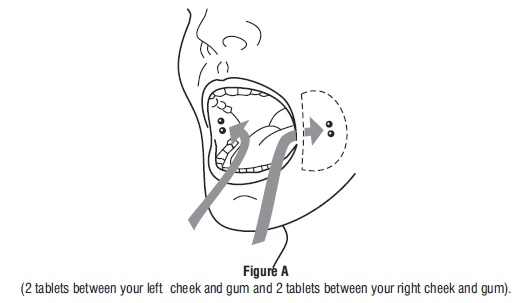
Tell the patient to place two 200 mcg misoprostol tablets in each cheek pouch (the area between the cheek and gums) for 30 minutes and then swallow any remnants with water or another liquid (see Figure 1).
Patients taking Mifepristone tablets, 200 mg must take misoprostol within 24 to 48 hours after taking Mifepristone. The effectiveness of the regimen may be lower if misoprostol is administered less than 24 hours or more than 48 hours after mifepristone administration.
Because most women will expel the pregnancy within 2 to 24 hours of taking misoprostol
[see Clinical Studies (
14)]
, discuss with the patient an appropriate location for them to be when taking the misoprostol, taking into account that expulsion could begin within 2 hours of administration.
2.2 Patient Management Following Misoprostol Administration
During the period immediately following the administration of misoprostol, the patient may need medication for cramps or gastrointestinal symptoms [see Adverse Reactions ( 6)] .
Give the patient:
- Instructions on what to do if significant discomfort, excessive vaginal bleeding or other adverse reactions occur
- A phone number to call if the patient has questions following the administration of the misoprostol
- The name and phone number of the healthcare provider who will behandling emergencies.
2.3 Post-treatment Assessment: Day 7 to 14
Patients should follow-up with their healthcare provider approximately 7 to 14 days after the administration of Mifepristone. This assessment is very important to confirm that complete termination of pregnancy has occurred and to evaluate the degree of bleeding. Termination can be confirmed by medical history, clinical examination, human Chorionic Gonadotropin (hCG) testing, or ultrasonographic scan. Lack of bleeding following treatment usually indicates failure; however, prolonged or heavy bleeding is not proof of a complete abortion.
The existence of debris in the uterus (e.g., if seen on ultrasonography) following the treatment procedure will not necessarily require surgery for its removal.
Patients should expect to experience vaginal bleeding or spotting for an average of 9 to 16 days. Women report experiencing heavy bleeding for a median duration of 2 days. Up to 8% of women may experience some type of bleeding for more than 30 days. Persistence of heavy or moderate vaginal bleeding at the time of follow-up, however, could indicate an incomplete abortion.
If complete expulsion has not occurred, but the pregnancy is not ongoing, patients may be treated with another dose of misoprostol 800mcg buccally. There have been rare reports of uterine rupture in women who took mifepristone tablets, 200 mg and misoprostol, including women with prior uterine rupture or uterine scar and patients who received multiple doses of misoprostol within 24 hours. Patients who choose to use a repeat dose of misoprostol should have a follow-up visit with their healthcare provider in approximately 7 days to assess for complete termination.
Surgical evacuation is recommended to manage ongoing pregnancies after medical abortion
[see
Use in Specific Populations (
8.1)]
. Advise the patient whether you will provide such care or will refer her to another provider as part of counseling prior to prescribing Mifepristone tablets, 200 mg.
3 DOSAGE FORMS AND STRENGTHS
Tablets containing 200 mg of mifepristone each, supplied as 1 tablet on one blister card. Mifepristone tablets are light yellow in color, circular, bio-convex tablets, approximately 11mm in diameter and imprinted on one side with “S.”
4 CONTRAINDICATIONS
- Administration of Mifepristone tablets, 200 mg and misoprostol for the termination of pregnancy (the “treatment procedure”) is contraindicated in patients with any of the following conditions:
- Confirmed or suspected ectopic pregnancy or undiagnosed adnexal mass (the treatment procedure will not be effective to terminate an ectopic pregnancy) [see Warnings and Precautions ( 5.4)]
- Chronic adrenal failure (risk of acute adrenal insufficiency)
- Concurrent long-term corticosteroid therapy (risk of acute adrenal insufficiency)
- History of allergy to mifepristone, misoprostol, or other prostaglandins (allergic reactions including anaphylaxis, angioedema, rash, hives, and itching have been reported [see Adverse Reactions ( 6.2)])
- Hemorrhagic disorders or concurrent anticoagulant therapy (risk of heavy bleeding)
- Inherited porphyrias (risk of worsening or of precipitation of attacks)
- Use of Mifepristone tablets, 200 mg and misoprostol for termination of intrauterine pregnancy is contraindicated in patients with an intrauterine device (“IUD”) in place (the IUD might interfere with pregnancy termination). If the IUD is removed, Mifepristone tablets, 200 mg may be used.
5 WARNINGS AND PRECAUTIONS
5.1 Infection and Sepsis
As with other types of abortion, cases of serious bacterial infection, including very rare cases of fatal septic shock, have been reported following the use of Mifepristone tablets, 200 mg [see Boxed Warning]. Healthcare providers evaluating a patient who is undergoing a medical abortion should be alert to the possibility of this rare event. A sustained (>4 hours) fever of 100.4°F or higher, severe abdominal pain, or pelvic tenderness in the days after a medical abortion may be an indication of infection.
A high index of suspicion is needed to rule out sepsis (e.g., from Clostridium sordellii) if a patient reports abdominal pain or discomfort or general malaise (including weakness, nausea, vomiting or diarrhea) more than 24 hours after taking misoprostol. Very rarely, deaths have been reported in patients who presented without fever, with or without abdominal pain, but with leukocytosis with a marked left shift, tachycardia, hemoconcentration, and general malaise. No causal relationship between Mifepristone tablets, 200 mg and misoprostol use and an increased risk of infection or death has been established. Clostridium sordellii infections have also been reported very rarely following childbirth (vaginal delivery and caesarian section), and in other gynecologic and non-gynecologic conditions.
5.2 Uterine Bleeding
Uterine bleeding occurs in almost all patients during a medical abortion. Prolonged heavy bleeding (soaking through two thick full-size sanitary pads per hour for two consecutive hours) may be a sign of incomplete abortion or other complications and prompt medical or surgical intervention may be needed to prevent the development of hypovolemic shock. Counsel patients to seek immediate medical attention if they experience prolonged heavy vaginal bleeding following a medical abortion [see Boxed Warning].
Women should expect to experience vaginal bleeding or spotting for an average of 9 to 16 days. Women report experiencing heavy bleeding for a median duration of 2 days.
Up to 8% of all subjects may experience some type of bleeding for 30 days or more. In general, the duration of bleeding and spotting increased as the duration of the pregnancy increased.
Decreases in hemoglobin concentration, hematocrit, and red blood cell count may occur in patients who bleed heavily.
Excessive uterine bleeding usually requires treatment by uterotonics, vasoconstrictor drugs, surgical uterine evacuation, administration of saline infusions, and/or blood transfusions. Based on data from several large clinical trials, vasoconstrictor drugs were used in 4.3% of all subjects, there was a decrease in hemoglobin of more than 2 g/dL in 5.5% of subjects, and blood transfusions were administered to ≤0.1% of subjects. Because heavy bleeding requiring surgical uterine evacuation occurs in about 1% of patients, special care should be given to patients with hemostatic disorders, hypocoagulability, or severe anemia.
5.3 Mifepristone REMS Program
Mifepristone tablets, 200 mg is available only through a restricted program under a REMS called the Mifepristone REMS Program, because of the risks of serious complications [see Warnings and Precautions ( 5.1, 5.2)]
Notable requirements of the Mifepristone REMS Program include the following:
- Prescribers must be certified with the program by completing the Prescriber Agreement Form
- Patients must sign a Patient Agreement Form
- Mifepristone tablets, 200 mg must be dispensed to patients by or under the supervision of a certified prescriber, or by certified pharmacies on prescriptions issued by certified prescribers
Further information is available at 1-855-MIFEINFO (1-855-643-3463).
5.4 Ectopic Pregnancy
Mifepristone tablets, 200 mg is contraindicated in patients with a confirmed or suspected ectopic pregnancy because mifepristone is not effective for terminating ectopic pregnancies [see Contraindications ( 4)]. Healthcare providers should remain alert to the possibility that a patient who is undergoing a medical abortion could have an undiagnosed ectopic pregnancy because some of the expected symptoms experienced with a medical abortion (abdominal pain, uterine bleeding) may be similar to those of a ruptured ectopic pregnancy. The presence of an ectopic pregnancy may have been missed even if the patient underwent ultrasonography prior to being prescribed Mifepristone tablets, 200 mg.
Patients who became pregnant with an IUD in place should be assessed for ectopic pregnancy.
6 ADVERSE REACTIONS
The following adverse reactions are described in greater detail in other sections:
- Infection and sepsis [see Warnings and Precautions ( 5.1)]
- Uterine bleeding [see Warnings and Precautions ( 5.2)]
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Information presented on common adverse reactions relies solely on data from US studies, because rates reported in non-US studies were markedly lower and are not likely generalizable to the US population. In three US clinical studies totaling 1,248 women through 70 days gestation who used mifepristone 200 mg orally followed 24-48 hours later by misoprostol 800mcg buccally, women reported adverse reactions in diaries and in interviews at the follow-up visit. These studies enrolled generally healthy women of reproductive age without contraindications to mifepristone or misoprostol use according to the Mifepristone tablets, 200 mg product label.Gestational age was assessed prior to study enrollment using the date of the woman's last menstrual period, clinical evaluation, and/or ultrasound examination.
About 85% of patients report at least one adverse reaction following administration of Mifepristone tablets, 200 mg and misoprostol, and many can be expected to report more than one such reaction. The most commonly reported adverse reactions (>15%) were nausea, weakness, fever/chills, vomiting, headache, diarrhea, and dizziness (see Table 1). The frequency of adverse reactions varies between studies and may be dependent on many factors including the patient population and gestational age.
Abdominal pain/cramping is expected in all medical abortion patients and its incidence is not reported in clinical studies. Treatment with Mifepristone tablets, 200 mg and misoprostol is designed to induce uterine bleeding and cramping to cause termination of an intrauterine pregnancy. Uterine bleeding and cramping are expected consequences of the action of Mifepristone tablets, 200 mg and misoprostol as used in the treatment procedure. Most patients can expect bleeding more heavily than they do during a heavy menstrual period [see Warnings and Precautions ( 5.2) ].
Table 1 lists the adverse reactions reported in US clinical studies with incidence >15% of women.
Table 1 Adverse Reactions Reported in Women Following Administration of Mifepristone (oral) and Misoprostol (buccal) in US Clinical Studies
| Adverse Reaction | # US
studies | Number of Evaluable
Women | Range of
frequency (%) | Upper Gestational Age of Studies
Reporting Outcome |
| Nausea | 3 | 1,248 | 51-75% | 70 days |
| Weakness | 2 | 630 | 55-58% | 63 days |
| Fever/chills | 1 | 414 | 48% | 63 days |
| Vomiting | 3 | 1,248 | 37-48% | 70 days |
| Headache | 2 | 630 | 41-44% | 63 days |
| Diarrhea | 3 | 1,248 | 18-43% | 70 days |
| Dizziness | 2 | 630 | 39-41% | 63 days |
One study provided gestational-age stratified adverse reaction rates for women who were 57-63 and 64-70 days; there was little difference in frequency of the reported common adverse reactions by gestational age.
Information on serious adverse reactions was reported in six US and four non-US clinical studies, totaling 30,966 women through 70 days gestation who used mifepristone 200mg orally followed 24-48 hours later by misoprostol 800mcg buccally. Serious adverse reaction rates were similar between US and non-US studies, so rates from both US and non-US studies are presented. In the US studies, one studied women through 56 days gestation, four through 63 days gestation, and one through 70 days gestation, while in the non-US studies, two studied women through 63 days gestation, and two through 70 days gestation. Serious adverse reactions were reported in <0.5% of women. Information from the US and non-US studies is presented in Table 2.
Table 2 Serious Adverse Reactions Reported in Women Following Administration of Mifepristone (oral) and Misoprostol (buccal) in US and Non-US Clinical Studies
|
NR= Not reported *This outcome represents a single patient who experienced death related to sepsis. |
||||||
| Adverse
Reaction | US | Non-US | ||||
| # of studies | Number of Evaluable Women | Range of
frequency (%) | # of
studies | Number of
Evaluable Women | Range of
frequency (%) |
|
| Transfusion | 4 | 17,774 | 0.03-0.5% | 3 | 12,134 | 0-0.1% |
| Sepsis | 1 | 629 | 0.2% | 1 | 11,155 | <0.01% * |
| ER visit | 2 | 1,043 | 2.9-4.6% | 1 | 95 | 0 |
| Hospitalization
Related to Medical Abortion | 3 | 14,339 | 0.04-0.6% | 3 | 1,286 | 0-0.7% |
| Infection without
sepsis | 1 | 216 | 0 | 1 | 11,155 | 0.2% |
| Hemorrhage | NR | NR | NR | 1 | 11,155 | 0.1% |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of Mifepristone tablets, 200 mg and misoprostol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and infestations: post-abortal infection (including endometritis, endomyometritis, parametritis, pelvic infection, pelvic inflammatory disease, salpingitis)
Blood and the lymphatic system disorders: anemia
Immune system disorders: allergic reaction (including anaphylaxis, angioedema, hives, rash, itching)
Psychiatric disorders: anxiety
Cardiac disorders: tachycardia (including racing pulse, heart palpitations, heart pounding)
Vascular disorders: syncope, fainting, loss of consciousness, hypotension (including orthostatic), light-headedness
Respiratory, thoracic and mediastinal disorders: shortness of breath
Gastrointestinal disorders: dyspepsia
Musculoskeletal, connective tissue and bone disorders: back pain, leg pain
Reproductive system and breast disorders: uterine rupture, ruptured ectopic pregnancy, hematometra, leukorrhea
General disorders and administration site conditions: pain
7 DRUG INTERACTIONS
7.1 Drugs that May Reduce Mifepristone tablets, 200 mg Exposure (Effect of CYP 3A4 Inducers on Mifepristone tablets, 200mg)
CYP450 3A4 is primarily responsible for the metabolism of mifepristone. CYP3A4 inducers such as rifampin, dexamethasone, St. John's Wort, and certain anticonvulsants (such as phenytoin, phenobarbital, carbamazepine) may induce mifepristone metabolism (lowering serum concentrations of mifepristone). Whether this action has an impact on the efficacy of the dose regimen is unknown. Refer to the follow-up assessment [see Dosage and Administration ( 2.3)] to verify that treatment has been successful.
7.2 Drugs that May Increase Mifepristone tablets, 200 mg Exposure (Effect of CYP 3A4 Inhibitors on Mifepristone tablets, 200mg)
Although specific drug or food interactions with mifepristone have not been studied, on the basis of this drug's metabolism by CYP 3A4, it is possible that ketoconazole, itraconazole erythromycin, and grapefruit juice may inhibit its metabolism (increasing serum concentrations of mifepristone). Mifepristone tablets, 200mg should be used with caution in patients currently or recently treated with CYP 3A4 inhibitors.
7.3 Effects of Mifepristone tablets, 200 mg on Other Drugs (Effect of Mifepristone tablets, 200 mg on CYP 3A4Substrates)
Based on in vitro inhibition information, coadministration of mifepristone may lead to an increase in serum concentrations of drugs that are CYP 3A4 substrates. Due to the slow elimination of mifepristone from the body, such interaction may be observed for a prolonged period after its administration. Therefore, caution should be exercised when mifepristone is administered with drugs that are CYP 3A4 substrates and have a narrow therapeutic range.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Mifepristone is indicated, in a regimen with misoprostol, for the medical termination of intrauterine pregnancy through 70 days gestation. Risks to pregnant patients are discussed throughout the labeling.
Refer to misoprostol labeling for risks to pregnant patients with the use of misoprostol.
The risk of adverse developmental outcomes with a continued pregnancy after a failed pregnancy termination with Mifepristone tablets, 200 mg in a regimen with misoprostol is unknown; however, the process of a failed pregnancy termination could disrupt normal embryo-fetal development and result in adverse developmental effects. Birth defects have been reported with a continued pregnancy after a failed pregnancy termination with Mifepristone tablets, 200 mg in a regimen with misoprostol. In animal reproduction studies, increased fetal losses were observed in mice, rats, and rabbits and skull deformities were observed in rabbits with administration of mifepristone at doses lower than the human exposure level based on body surface area.
Data
Animal Data
In teratology studies in mice, rats and rabbits at doses of 0.25 to 4.0mg/kg (less than 1/100 to approximately 1/3 the human exposure based on body surface area), because of the antiprogestational activity of mifepristone, fetal losses were much higher than in control animals. Skull deformities were detected in rabbit studies at approximately 1/6 the human exposure, although no teratogenic effects of mifepristone have been observed to date in rats or mice.
These deformities were most likely due to the mechanical effects of uterine contractions resulting from inhibition of progesterone action.
8.2 Lactation
Mifepristone tablets, 200 mg is present in human milk. Limited data demonstrate undetectable to low levels of the drug in human milk with the relative (weight-adjusted) infant dose 0.5% or less as compared to maternal dosing. There is no information on the effects of Mifepristone tablets, 200 use of misoprostol. The developmental and health benefits of breast-feeding should be considered along with any potential adverse effects on the breast-fed child from Mifepristone tablets, 200 mg in a regimen with misoprostol.
8.4 Pediatric Use
Safety and efficacy of Mifepristone tablets, 200 mg have been established in pregnant females. Data from a clinical study of Mifepristone tablets, 200 mg that included a subset of 322 females under age 17 demonstrated a safety and efficacy profile similar to that observed in adults.
10 OVERDOSAGE
No serious adverse reactions were reported in tolerance studies in healthy non-pregnant female and healthy male subjects where mifepristone was administered in single doses greater than 1,800 mg (ninefold the recommended dose for medical abortion). If a patient ingests a massive overdose, the patient should be observed closely for signs of adrenal failure.
11 DESCRIPTION
Mifepristone tablets each contain 200 mg of mifepristone, a synthetic steroid with antiprogestational effects. The tablets are light yellow in color, circular, bi-convex, and are intended for oral administration only. The tablets include the inactive ingredients colloidal silicon dioxide, corn starch, povidone, microcrystalline cellulose, and magnesium stearate.
Mifepristone is a substituted 19-nor steroid compound chemically designated as 11ß-[ p- (Dimethylamino) phenyl]-17ß-hydroxy- 17-(1-propynyl) estra-4,9-dien-3-one. Its empirical formula is C 29H 35NO 2. Its structural formula is:
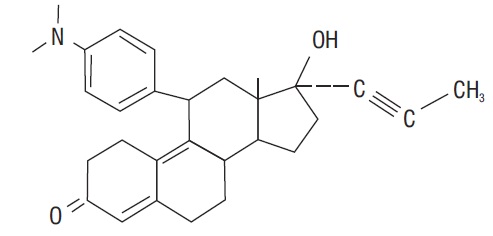
The compound is a yellow powder with a molecular weight of 429.6 and a melting point of 192- 196°C. It is freely soluble in methanol, chloroform and acetone and poorly soluble in water, hexane and isopropyl ether.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The anti-progestational activity of mifepristone results from competitive interaction with progesterone at progesterone-receptor sites. Based on studies with various oral doses in several animal species (mouse, rat, rabbit, and monkey), the compound inhibits the activity of endogenous or exogenous progesterone, resulting in effects on the uterus and cervix that, when combined with misoprostol, result in termination of an intrauterine pregnancy.
During pregnancy, the compound sensitizes the myometrium to the contraction-inducing activity of prostaglandins.
12.2 Pharmacodynamics
Use of Mifepristone tablets, 200 mg in a regimen with misoprostol disrupts pregnancy by causing decidual necrosis, myometrial contractions, and cervical softening, leading to the expulsion of the products of conception.
Doses of 1mg/kg or greater of mifepristone have been shown to antagonize the endometrial and myometrial effects of progesterone in women.
Antiglucocorticoid and antiandrogenic activity: Mifepristone also exhibits antiglucocorticoid and weak antiandrogenic activity. The activity of the glucocorticoid dexamethasone in rats was inhibited following doses of 10 to 25 mg/kg of mifepristone. Doses of 4.5mg/kg or greater in human beings resulted in a compensatory elevation of adrenocorticotropic hormone (ACTH) and cortisol. Antiandrogenic activity was observed in rats following repeated administration of doses from 10 to 100 mg/kg.
12.3 Pharmacokinetics
Mifepristone is rapidly absorbed after oral ingestion with non-linear pharmacokinetics for C max after single oral doses of 200 mg and 600 mg in healthy subjects.
Absorption
The absolute bioavailability of a 20 mg mifepristone oral dose in females of childbearing age is 69%. Following oral administration of a single dose of 600mg, mifepristone is rapidly absorbed, with a peak plasma concentration of 1.98 ± 1.0mg/L occurring approximately 90 minutes after ingestion.
Following oral administration of a single dose of 200 mg in healthy men (n=8), mean C max was1.77 ± 0.7 mg/L occurring approximately 45 minutes after ingestion. Mean AUC0-∞ was 25.8± 6.2 mg*hr/L.
Distribution
Mifepristone is 98% bound to plasma proteins, albumin, and 1-acid glycoprotein. Binding to the latter protein is saturable, and the drug displays nonlinear kinetics with respect to plasma concentration and clearance.
Elimination
Following a distribution phase, elimination of mifepristone is slow at first (50% eliminated between 12 and 72 hours) and then becomes more rapid with a terminal elimination half-life of 18 hours.
Metabolism
Metabolism of mifepristone is primarily via pathways involving N-demethylation and terminal hydroxylation of the 17-propynyl chain.
In vitro studies have shown that CYP450 3A4 is primarily responsible for the metabolism. The three major metabolites identified in humans are: (1) RU 42 633, the most widely found in plasma, is the N-monodemethylated metabolite; (2) RU 42 848, which results from the loss of two methyl groups from the 4-dimethylaminophenyl in position 11ß; and (3) RU 42 698, which results from terminal hydroxylation of the 17-propynyl chain.
Excretion
By 11 days after a 600mg dose of tritiated compound, 83% of the drug has been accounted for by the feces and 9% by the urine. Serum concentrations are undetectable by 11 days.
Specific Populations
The effects of age, hepatic disease and renal disease on the safety, efficacy and pharmacokinetics of mifepristone have not been investigated.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No long-term studies to evaluate the carcinogenic potential of mifepristone have been performed.
Mutagenesis
Results from studies conducted
in vitro and in animals have revealed no genotoxic potential for mifepristone. Among the tests carried out were: Ames test with and without metabolic activation; gene conversion test in
Saccharomyces cerevisiae D4 cells; forward mutation in
Schizosaccharomyces pompe P1 cells; induction of unscheduled DNA synthesis in cultured HeLa cells; induction of chromosome aberrations in CHO cells;
in vitro test for gene mutation in V79 Chinese hamster lung cells; and micronucleus test in mice.
Impairment of Fertility
In rats, administration of 0.3mg/kg mifepristone per day caused severe disruption of the estrus cycles for the three weeks of the treatment period. Following resumption of the estrus cycle, animals were mated and no effects on reproductive performance were observed.
14 CLINICAL STUDIES
Safety and efficacy data from clinical studies of mifepristone 200 mg orally followed 24-48 hours later by misoprostol 800 mcg buccally through 70 days gestation are reported below. Success was defined as the complete expulsion of the products of conception without the need for surgical intervention. The overall rates of success and failure, shown by reason for failure based on 22 worldwide clinical studies (including 7 US studies) appear in Table 3.
The demographics of women who participated in the US clinical studies varied depending on study location and represent the racial and ethnic variety of American females. Females of all reproductive ages were represented, including females less than 18 and more than 40 years of age; most were 27 years or younger.
Table 3 Outcome Following Treatment with Mifepristone (oral) and Misoprostol (buccal) Through 70 Days Gestation
| US Trials | Non-US Trials | |
| N | 16,794 | 18,425 |
| Complete Medical Abortion | 97.4% | 96.2% |
| Surgical Intervention * | 2.6% | 3.8% |
| Ongoing Pregnancy ** | 0.7% | 0.9% |
| * Reasons for surgical intervention include ongoing pregnancy, medical necessity, persistent or heavy bleeding after treatment, patient request, or incomplete expulsion.
** Ongoing pregnancy is a subcategory of surgical intervention, indicating the percent of women who have surgical intervention due to an ongoing pregnancy. |
||
The results for clinical studies that reported outcomes, including failure rates for ongoing pregnancy, by gestational age are presented in Table 4.
Table 4 Outcome by Gestational Age Following Treatment with Mifepristone and Misoprostol (buccal) for US and Non-US Clinical Studies
| <49 days | 50-56 days | 57-63 days | 64-70 days | |||||||||
| N | % | Number of
Evaluable Studies | N | % | Number of
Evaluable Studies | N | % | Number of
Evaluable Studies | N | % | Number of
Evaluable Studies |
|
| Complete
medical abortion | 12,046 | 98.1 | 10 | 3,941 | 96.8 | 7 | 2,294 | 94.7 | 9 | 479 | 92.7 | 4 |
| Surgical
intervention for ongoing pregnancy | 10,272 | 0.3 | 6 | 3,788 | 0.8 | 6 | 2,211 | 2 | 8 | 453 | 3.1 | 3 |
One clinical study asked subjects through 70 days gestation to estimate when they expelled the pregnancy, with 70% providing data. Of these, 23-38% reported expulsion within 3 hours and over 90% within 24 hours of using misoprostol.
16 HOW SUPPLIED/STORAGE AND HANDLING
Mifepristone tablets, 200 mg is only available through a restricted program called the Mifepristone REMS Program [see Warnings and Precautions ( 5.3)]
Mifepristone tablets, 200 mg is supplied as light yellow, circular, bi-convex, uncoated tablets debossed with “S” on one side and plain on other side. Each tablet contains 200 mg of mifepristone. One tablet is individually blistered on one blister card that is packaged in an individual package (National Drug Code 43393-001-01).
Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide), included with each package of Mifepristone tablets, 200 mg. Additional copies of the Medication Guide are available by contacting GenBioPro, Inc., at 1-855-MIFEINFO (1-855-643-3463) or from www.MIFEINFO.com.
Serious Infections and Bleeding
- Inform the patient that uterine bleeding and uterine cramping will occur [see Warnings and Precautions ( 5.2)].
- Advise the patient that serious and sometimes fatal infections and bleeding can occur very rarely [see Warnings and Precautions ( 5.1, 5.2)].
- Mifepristone tablets, 200 mg are only available through a restricted program called the Mifepristone REMS Program [see
Warnings and Precautions (
5.3)]. Under the Mifepristone REMS Program:
- Patients must sign a Patient Agreement Form.
- Mifepristone tablets, 200mg is only available by or under the supervision of certified prescribers, or by certified pharmacies on prescriptions issued by certified prescribers.
Provider Contacts and Actions in Case of Complications
- Ensure that the patient knows whom to call and what to do, including going to an emergency room if none of the provided contacts are reachable, or if the patient experiences complications including prolonged heavy bleeding, severe abdominal pain, or sustained fever [see Boxed Warning].
Compliance with Treatment Schedule and Follow-up Assessment
- Advise the patient that it is necessary to complete the treatment schedule; including a follow-up assessment approximately 7 to14 days after taking Mifepristone tablets, 200mg [see Dosage and Administration ( 2.3)].
- Explain that:
- prolonged heavy vaginal bleeding is not proof of a complete abortion,
- if the treatment fails and the pregnancy continues, the risk of fetal malformation is unknown,
- it is recommended that ongoing pregnancy be managed by surgical termination [see Dosage and Administration ( 2.3)] . Advise the patient whether you will provide such care or will refer her to another provider.
Subsequent Fertility
- Inform the patient that another pregnancy can occur following medical abortion and before resumption of normal menses.
- Inform the patient that contraception can be initiated as soon as pregnancy expulsion has been confirmed, or before she resumes sexual intercourse.
Manufactured for:
GenBioPro, Inc.
P.O. Box 32011
Las Vegas, NV 89103
1-855-MIFEINFO (1-855-643-3463)
www.MIFEINFO.com
01/2023
PI.MIF.R3
MEDICATION GUIDE
Mifepristone (MIF-eh-pris-tone) tablets, 200 mg for oral use
Read this information carefully before taking Mifepristone tablets, 200 mg and misoprostol. It will help you understand how the treatment works. This Medication Guide does not take the place of talking with your healthcare provider.
What is the most important information I should know about Mifepristone tablets, 200 mg?
What symptoms should I be concerned with? Although cramping and bleeding are an expected part of ending a pregnancy, rarely, serious and potentially life-threatening bleeding, infections, or other problems can occur following a miscarriage, surgical abortion, medical abortion, or childbirth. Seeking medical attention as soon as possible is needed in these circumstances. Serious infection has resulted in death in a very small number of cases. There is no information that use of Mifepristone tablets, 200mg and misoprostol caused these deaths. If you have any questions, concerns, or problems, or if you are worried about any side effects or symptoms, you should contact your healthcare provider. You can write down your healthcare provider's telephone number here _______________________.
Be sure to contact your healthcare provider promptly if you have any of the following:
- Heavy Bleeding. Contact your healthcare provider right away if you bleed enough to soak through two thick full-size sanitary pads per hour for two consecutive hours or if you are concerned about heavy bleeding. In about 1 out of 100 women, bleeding can be so heavy that it requires a surgical procedure (surgical aspiration or D&C).
- Abdominal Pain or “Feeling Sick.” If you have abdominal pain or discomfort, or you are “feeling sick,” including weakness, nausea, vomiting, or diarrhea, with or without fever, more than 24 hours after taking misoprostol, you should contact your healthcare provider without delay. These symptoms may be a sign of a serious infection or another problem (including an ectopic pregnancy, a pregnancy outside the womb).
- Fever. In the days after treatment, if you have a fever of 100.4°F or higher that lasts for more than 4 hours, you should contact your healthcare provider right away. Fever may be a symptom of a serious infection or another problem.
If you cannot reach your healthcare provider, go to the nearest hospital emergency room.
What to do if you are still pregnant after Mifepristone tablets, 200 mg with misoprostol treatment. If you are still pregnant, your healthcare provider will talk with you about a surgical procedure to end your pregnancy. In many cases, this surgical procedure can be done in the office/clinic. The chance of birth defects if the pregnancy is not ended is unknown.
Talk with your healthcare provider. Before you take Mifepristone tablets, 200 mg you should read this Medication Guide and you and your healthcare provider should discuss the benefits and risks of your using Mifepristone tablets, 200 mg.
What is Mifepristone tablets, 200 mg?
Mifepristone tablets, 200 mg is used in a regimen with another prescription medicine called misoprostol, to end an early pregnancy. Early pregnancy means it is 70 days (10 weeks) or less since your last menstrual period began. Mifepristone tablets, 200 mg is not approved for ending pregnancies that are further along. Mifepristone tablets, 200 mg blocks a hormone needed for your pregnancy to continue. When you use Mifepristone tablets, 200 mg on Day 1, you also need to take another medicine called misoprostol 24 to 48 hours after you take Mifepristone tablets, 200 mg to cause the pregnancy to be passed from your uterus.
The pregnancy is likely to be passed from your uterus within 2 to 24 hours after taking Mifepristone tablets, 200 mg and misoprostol. When the pregnancy is passed from the uterus, you will have bleeding and cramping that will likely be heavier than your usual period. About 2 to 7 out of 100 women taking Mifepristone tablets, 200 mg will need a surgical procedure because the pregnancy did not completely pass from the uterus or to stop bleeding.
Who should not take Mifepristone tablets, 200 mg?
Some patients should not take Mifepristone tablets, 200 mg. Do not take Mifepristone tablets, 200 mg if you:
- Have a pregnancy that is more than 70 days (10 weeks). Your healthcare provider may do a clinical examination, an ultrasound examination, or other testing to determine how far along you are in pregnancy.
- Are using an IUD (intrauterine device or system). It must be taken out before you take Mifepristone tablets, 200 mg.
- Have been told by your healthcare provider that you have a pregnancy outside the uterus (ectopic pregnancy).
- Have problems with your adrenal glands (chronic adrenal failure).
- Take a medicine to thin your blood.
- Have a bleeding problem.
- Have porphyria.
- Take certain steroid medicines.
- Are allergic to mifepristone, misoprostol, or medicines that contain misoprostol, such as Cytotec or Arthrotec.
Ask your healthcare provider if you are not sure about all your medical conditions before taking this medicine to find out if you can take Mifepristone tablets, 200 mg.
What should I tell my healthcare provider before taking Mifepristone tablets, 200 mg? Before you take Mifepristone tablets, 200 mg, tell your healthcare provider if you:
- cannot follow-up within approximately 7 to 14 days of your first visit
- are breastfeeding. Mifepristone tablets, 200 mg can pass into your breast milk. The effect of the Mifepristone tablets, 200 mg and misoprostol regimen on the breastfed infant or on milk production is unknown.
- are taking medicines, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Mifepristone tablets, 200 mg and certain other medicines may affect each other if they are used together. This can cause side effects.
How should I take Mifepristone tablets, 200 mg?
- Mifepristone tablets, 200 mg will be given to you by a healthcare provider or pharmacy.
- You and your healthcare provider will plan the most appropriate location for you to take the misoprostol, because it may cause bleeding, cramps, nausea, diarrhea, and other symptoms that usually begin within 2 to 24 hours after taking it.
- Most women will pass the pregnancy within 2 to 24 hours after taking the misoprostol tablets.
Follow the instruction below on how to take Mifepristone tablets, 200 mg and misoprostol: Mifepristone tablets, 200 mg (1 tablet) orally + misoprostol (4 tablets) buccally
Day 1:
- Take 1 Mifepristone 200 mg tablet by mouth.
24 to 48 hours after taking Mifepristone tablets, 200 mg:
- Take 4 misoprostol tablets by placing 2 misoprostol tablets in each cheek pouch (the area between your teeth and cheek - see Figure A) for 30 minutes and then swallow anything left over with a drink of water or another liquid.
- The medicines may not work as well if you take misoprostol sooner than 24 hours after Mifepristone tablets, 200mg or later than 48 hours after Mifepristone tablets, 200mg.
- Misoprostol often causes cramps, nausea, diarrhea, and other symptoms. Your healthcare provider may send you home with medicines for these symptoms.
Follow-up Assessment at Day 7 to 14:
- This follow-up assessment is very important. You must follow-up with your healthcare provider about 7 to 14 days after you have taken Mifepristone tablets, 200 mg to be sure you are well and that you have had bleeding and the pregnancy has passed from your uterus.
- Your healthcare provider will assess whether your pregnancy has passed from your uterus. If your pregnancy continues, the chance that there may be birth defects is unknown. If you are still pregnant, your healthcare provider will talk with you about a surgical procedure to end your pregnancy.
- If your pregnancy has ended, but not yet completely passed from your uterus, your provider will talk with you about other choices you have, including waiting, taking another dose of misoprostol, or having surgical procedure to empty your uterus.
When should I begin birth control?
You can become pregnant again right after your pregnancy ends. If you do not want to become pregnant again, start using birth control as soon as your pregnancy ends or before you start having sexual intercourse again.
What should I avoid while taking Mifepristone tablets, 200 mg and misoprostol?
Do not take any other prescription or over-the-counter medicines (including herbal medicines or supplements) at any time during the treatment period without first asking your healthcare provider about them because they may interfere with the treatment. Ask your healthcare provider about what medicines you can take for pain and other side effects.
What are the possible side effects of Mifepristone tablets, 200 mg and misoprostol?
Mifepristone tablets, 200 mg may cause serious side effects. See “What is the most important information I should know about Mifepristone tablets, 200 mg?”
Cramping and bleeding. Cramping and vaginal bleeding are expected with this treatment. Usually, these symptoms mean that the treatment is working. But sometimes you can get cramping and bleeding and still be pregnant. This is why you must follow-up with your healthcare provider approximately 7 to 14 days after taking Mifepristone tablets, 200 mg. See “How should I take Mifepristone tablets, 200 mg?” for more information on your follow-up assessment. If you are not already bleeding after taking Mifepristone tablets, 200 mg you probably will begin to bleed once you take misoprostol, the medicine you take 24 to 48 hours after Mifepristone tablets, 200 mg. Bleeding or spotting can be expected for an average of 9 to16 days and may last for up to 30 days. Your bleeding may be similar to, or greater than, a normal heavy period. You may see blood clots and tissue. This is an expected part of passing the pregnancy.
The most common side effects of Mifepristone tablets, 200 mg treatment include: nausea, weakness, fever/chills, vomiting, headache, diarrhea and dizziness. Your provider will tell you how to manage any pain or other side effects. These are not all the possible side effects of Mifepristone tablets, 200 mg.
Call your healthcare provider for medical advice about any side effects that bother you or do not go away. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Mifepristone tablets, 200 mg.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. This Medication Guide summarizes the most important information about Mifepristone tablets, 200 mg. If you would like more information, talk with your healthcare provider. You may ask your healthcare provider for information about Mifepristone tablets, 200 mg that is written for healthcare professionals. For more information about Mifepristone tablets, 200 mg, go to www.MIFEINFO.com or call 1- 855-MIFEINFO (1- 855-643-3463).
Manufactured for:
GenBioPro, Inc.
P.O. Box 32011
Las Vegas, NV 89103
1-855-MIFEINFO (1-855-643-3463)
www.MIFEINFO.com
This Medication Guide has been approved by the U.S. Food and Drug Administration.
MG.MIF.R3
Approved 01/2023