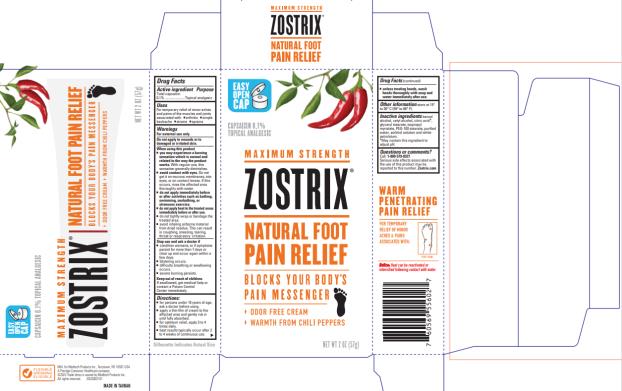
Uses
For the temporary relief of minor aches and pains of the muscles and joints associated with
- Strains
- Sprains
- Bruises
- arthritis
Warnings
For external use only.
Do not apply to wounds or to damaged or irritated skin.
When using this product
-
you may experience a burning sensation which is normal and related to the way the product works. With regular use, this sensation generally diminishes.
-
avoid contact with eyes. Do not get it on mucous membranes, into eyes, or on contact lenses. If this occurs, rinse the affected area thoroughly with water.
-
do not apply immediately before or after activities such as bathing, swimming, sun bathing, or strenuous exercise.
-
do not apply heat to the treated areas immediately before or after use.
- do not tightly wrap or bandage the treated area.
- avoid inhaling airborne material from dried residue. This can result in coughing, sneezing, tearing, throat or respiratory irritation.
Directions
- for persons under 18 years of age, ask a doctor before using.
- apply a thin film of cream to the affected area and gently rub in until fully absorbed.
- for optimum relief, apply 3 to 4 times daily
- best results typically occur after 2 to 4 weeks of continuous use.
-
wash hands thoroughly with soap and water immediately after use.
- see package insert for more information.
Inactive ingredients
benzyl alcohol, cetyl alcohol, citric acid*, glyceryl stearate, isopropyl myristate, PEG-100 stearate, purified water, sorbitol solution & white petrolatum. *May contain this ingredient to adjust pH.