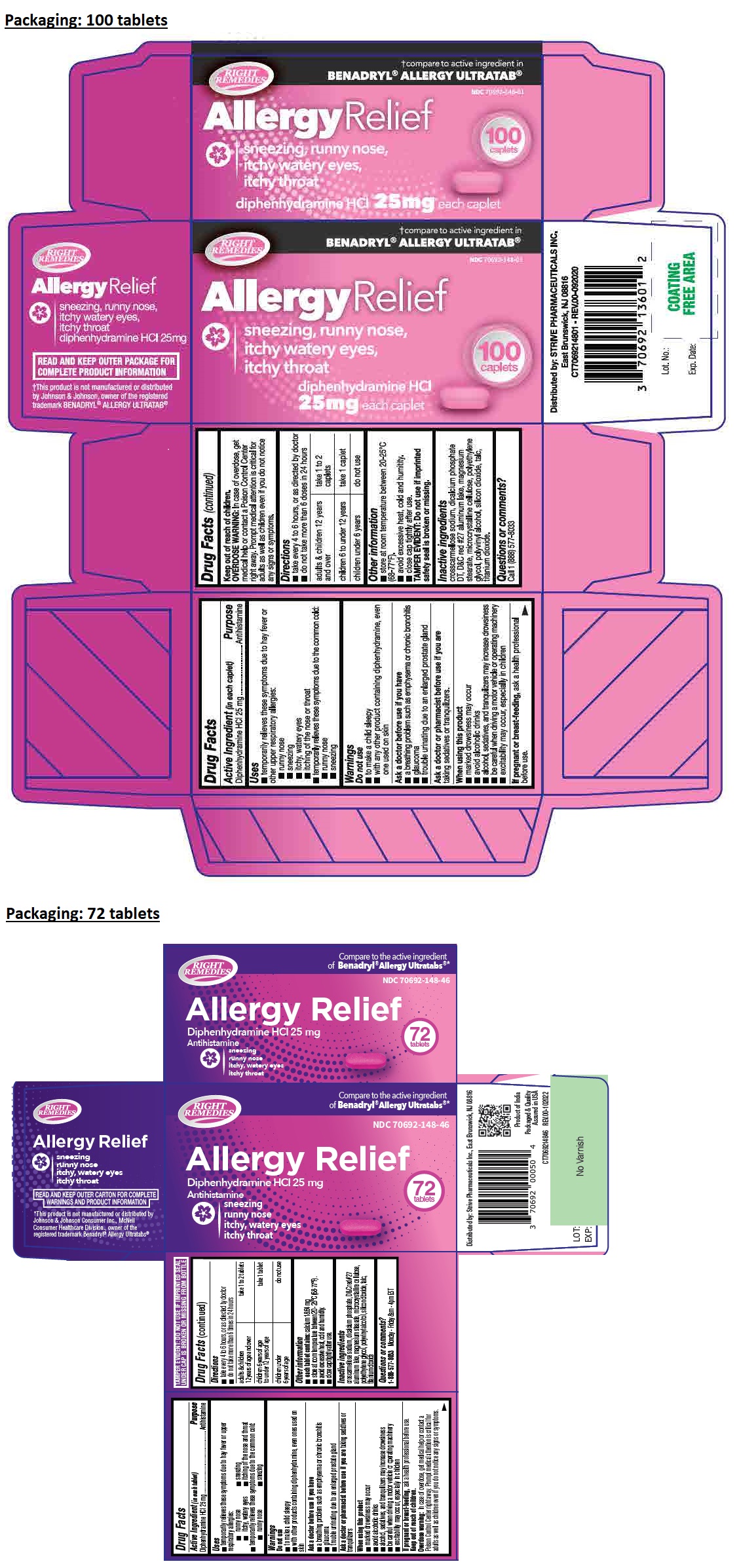
Uses
• temporarily relieves these symptoms due to hay fever or upper respiratory allergies:
• runny nose • sneezing
• itchy, watery eyes • itching of the nose and throat
• temporarily relieves these symptoms due to the common cold:
• runny nose • sneezing
Warnings
Do not use
• to make a child sleepy
• with other products containing diphenhydramine, even ones used on skin
Ask a doctor before use if you have
• a breathing problem such as emphysema or chronic bronchitis
• glaucoma
• trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
• marked drowsiness may occur
• avoid alcoholic drinks
• alcohol, sedatives, and tranquilizers may increase drowsiness
• be careful when driving a motor vehicle or operating machinery
• excitability may occur, especially in children
If pregnant or breast-feeding, ask a health professional before use.
Directions
• take every 4 to 6 hours, or as directed by doctor
• do not take more than 6 times in 24 hours
| adults & children 12 years of age and over | take 1 to 2 tablets |
| children 6 years of age to under 12 years of age | take 1 tablet |
| children under 6 years of age | do not use |
Other information
• each tablet contains: calcium 18.64 mg
• store at room temperature between 20 - 25°C (68-77°F).
• avoid excessive heat, cold and humidity.
• close cap tightly after use.
Inactive ingredients
croscarmellose sodium, dicalcium phosphate, D&C red #27 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, talc, titanium dioxide
RIGHT REMEDIES
Compare to the active ingredient of Benadryl® Allergy Ultratabs®*
Distributed by: Strive Pharmaceuticals Inc., East Brunswick, NJ 08816
Product of India
Packaged & Quality Assured in USA
REV.00-102022
READ AND KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
*This product is not manufactured or distributed by Johnson & Johnson Consumer Inc., McNeil Consumer Healthcare Division., owner of the registered trademark Benadryl® Allergy Ultratabs®
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL UNDER CAP IS BROKEN OR MISSING FROM BOTTLE