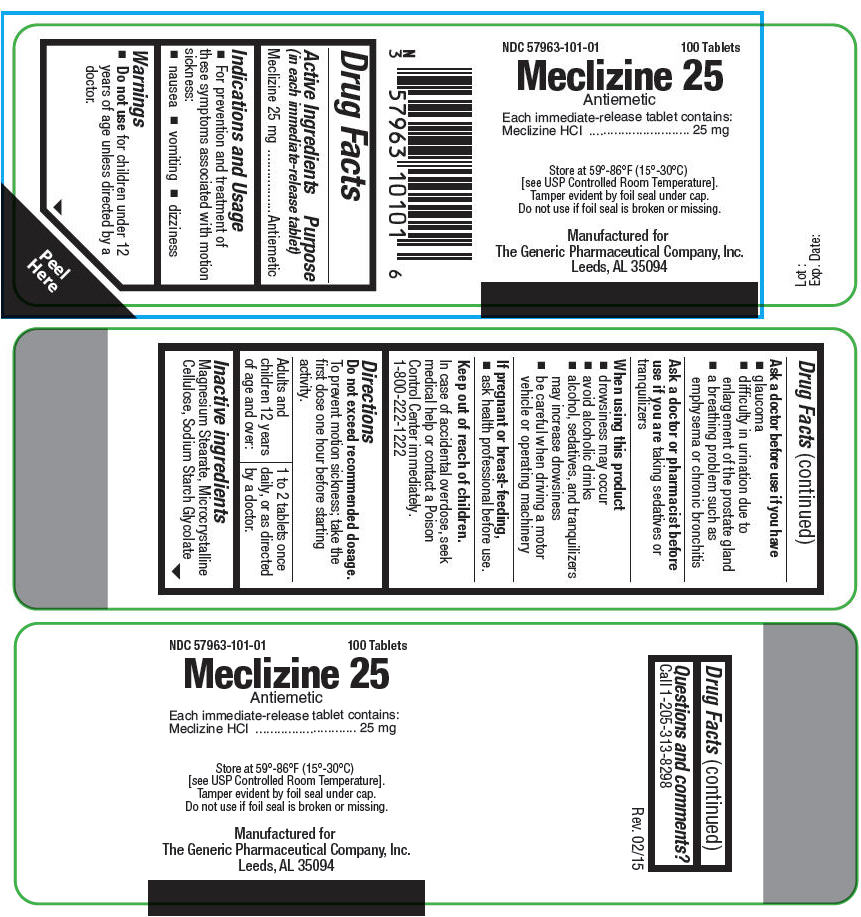
MECLIZINE 25- meclizine hydrochloride tablet
The Generic Pharmaceutical Company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredients (in each immediate-release tablet)
Meclizine 25 mg
Indications and Usage
- For prevention and treatment of these symptoms associated with motion sickness:
- nausea
- vomiting
- dizziness
Warnings
-
Do not use for children under 12 years of age unless directed by a doctor.
Ask a doctor before use if you have
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
If pregnant or breast-feeding,
- ask health professional before use.
Keep out of reach of children.
In case of accidental overdose, seek medical help or contact a Poison Control Center immediately. 1-800-222-1222
Directions
Do not exceed recommended dosage.
To prevent motion sickness; take the first dose one hour before starting activity.
| Adults and children 12 years of age and over: | 1 to 2 tablets once daily, or as directed by a doctor. |
Inactive ingredients
Magnesium Stearate, Microcrystalline Cellulose, Sodium Starch Glycolate
Questions and comments?
Call 1-205-313-8298
Manufactured for
The Generic Pharmaceutical Company, Inc.
Leeds, AL 35094
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label
NDC 57963-101-01
100 Tablets
Meclizine 25
Antiemetic
Each immediate-release tablet contains:
Meclizine HCl
25 mg
Store at 59°-86°F (15°-30°C)
[see USP Controlled Room Temperature].
Tamper evident by foil seal under cap.
Do not use if foil seal is broken or missing.
Manufactured for
The Generic Pharmaceutical Company, Inc.
Leeds, AL 35094