BHI BACK- ammonium chloride, matricaria recutita,citrullus colocynthis fruit pulp, pseudognaphalium obtusifolium, and toxicodendron pubescens leaf tablet
MediNatura Inc
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
ACTIVE INGREDIENT
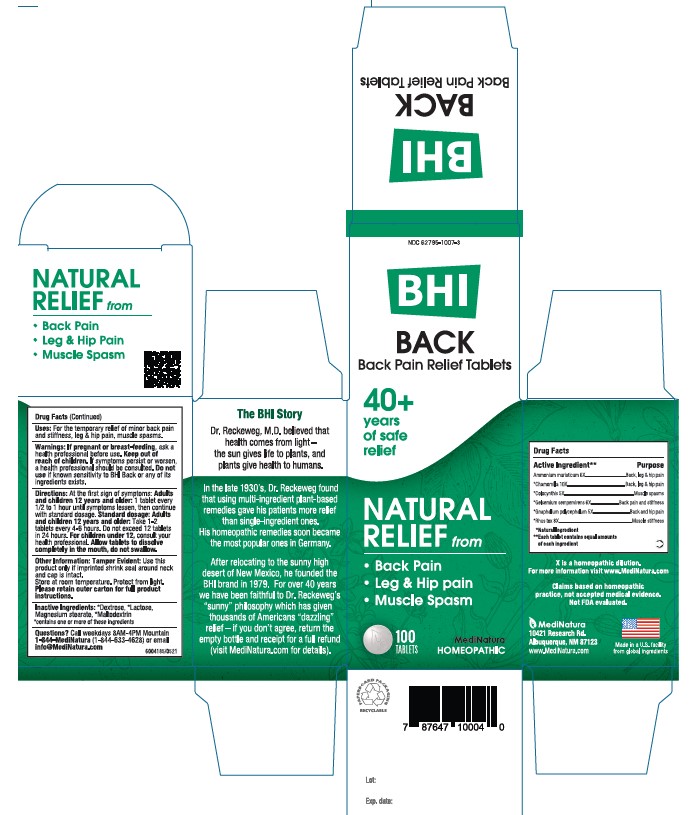
Each tablet contains: Ammonium muriaticum 6X, *Chamomilla 10X, *Colocynthis 5X, *Gelsemium sempervirens 6X, *Gnaphalium polycephalum 5X, *Rhus toxicodendron 8X 50 mg each.
*Natural Ingredients
INACTIVE INGREDIENT
Inactive Ingredients: **Dextrose, ** Lactose, Magnesium Stearate, **Maltodextrin
**contains one or more of these ingredients.
PURPOSE
Back Pain Relief Tablets
Relieves:
• Back Pain
• Leg & Hip Pain
• Muscle Spasm
USES
For the temporary relief of minor back pain, leg & hip pain, muscle spasm
DIRECTIONS
At first sign of symptoms:
Adults and children 12 years and older: 1 tablet every 1/2 to 1 hour until symptoms lessen, then continue with standard dosage.
Standard dosage: Adults and children 12 years and older: take 1-2 tablets 4 to 6 hours. Do not exceed 12 tablets in 24 hours. For children under 12, consult your health professional.
Allow tablets to dissolve completely in the mouth, do not swallow.
WARNINGS
Warnings: If pregnant or breast-feeding, ask a health professional before use. Keep out of reach of children. If symptoms persist or worsen, a health professional should be consulted. Do not use if known sensitivity to BHI Back or any of its ingredients exists.
KEEP OUT OF REACH OF CHILDREN
Keep out of reach of children.