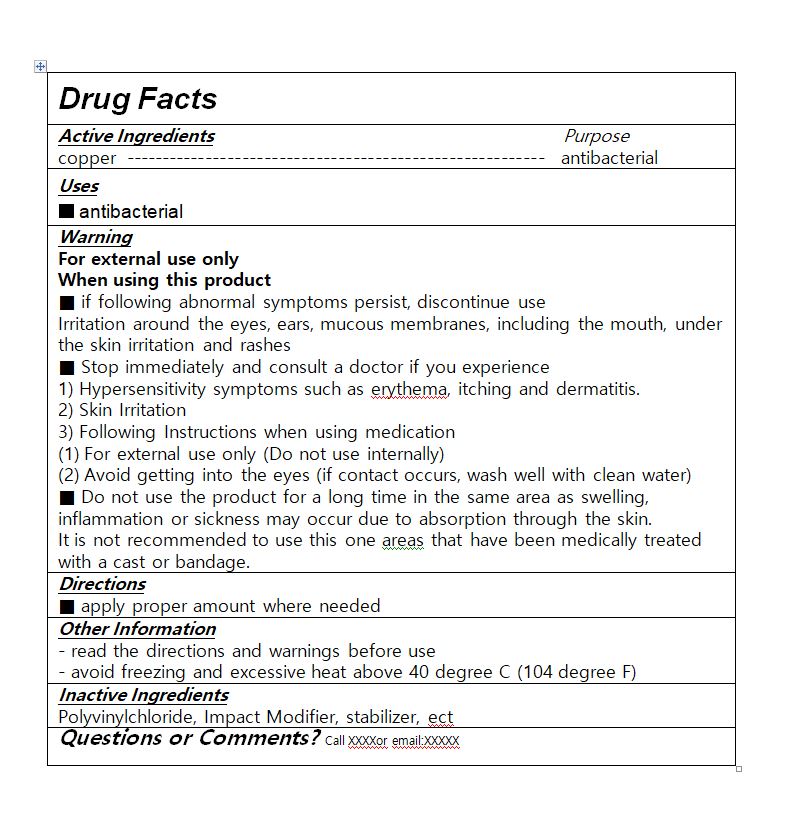
For external use only
When using this product
■ if following abnormal symptoms persist, discontinue use
Irritation around the eyes, ears, mucous membranes, including the mouth, under the skin irritation and rashes
■ Stop immediately and consult a doctor if you experience
1) Hypersensitivity symptoms such as erythema, itching and dermatitis.
2) Skin Irritation
3) Following Instructions when using medication
(1) For external use only (Do not use internally)
(2) Avoid getting into the eyes (if contact occurs, wash well with clean water)
■ Do not use the product for a long time in the same area as swelling, inflammation or sickness may occur due to absorption through the skin.
It is not recommended to use this one areas that have been medically treated with a cast or bandage.