BIVIGAM- human immunoglobulin g injection, solution
ADMA Biologics, Inc
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use BIVIGAM safely and effectively. See full prescribing information for BIVIGAM.
Immune Globulin Intravenous (Human), 10% Liquid BIVIGAM Initial U.S. Approval : 2012 WARNING: THROMBOSIS, RENAL DYSFUNCTION AND ACUTE RENAL FAILURE
|
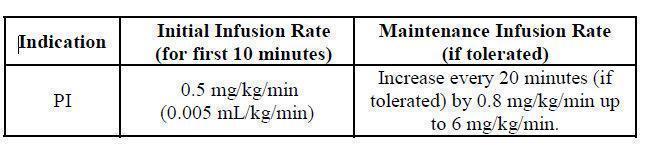
| Indication | Dose | Initial Infusion Rate |
Maintenance Infusion Rate (2) (if tolerated) (2) |
| PI |
300-800 mg/kg every 3-4 weeks (2) |
0.5 mg/kg/min for the first 10 minutes. (2) | Increase every 20 minutes (if tolerated) by 0.8 mg/kg/min up to 6 mg/kg/min. |
- Ensure that patients with pre-existing renal insufficiency are not volume depleted; discontinue BIVIGAM if renal function deteriorates. [5.3]
- For patients at risk of renal dysfunction or thrombotic events, administer BIVIGAM at the minimum infusion rate practicable. [5.1, 5.3]
(2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
(4)
- History of anaphylactic or severe systemic reactions to human immunoglobulin. [4]
- IgA deficient patients with antibodies to IgA and a history of hypersensitivity. [4, 5.2]
WARNINGS AND PRECAUTIONS
- Thrombotic events have occurred in patients receiving IGIV therapy. Monitor patients with known risk factors for thrombotic events; consider baseline assessment of blood viscosity for those at risk of hyperviscosity. [5.1, 5.4]
- IgA deficient patients with antibodies against IgA are at greater risk of developing severe hypersensitivity and anaphylactic reactions. Have medications such as epinephrine available immediately to treat any acute severe hypersensitivity reactions. [4, 5.2]
- Monitor renal function, including blood urea nitrogen (BUN), serum creatinine, and urine output in patients at risk of developing acute renal failure. [5.3, 5.9]
- Hyperproteinemia, increased serum viscosity, and hyponatremia or pseudohyponatremia can occur in patients receiving IGIV therapy. [5.4]
- Aseptic meningitis syndrome (AMS) has been reported with IGIV treatments, especially with high doses or rapid infusion. [5.5]
- Hemolytic anemia can develop subsequent to treatment with IGIV products. Monitor patients for hemolysis and hemolytic anemia. [5.6]
- Monitor patients for pulmonary adverse reactions (Transfusion-related acute lung injury [TRALI]). If transfusion-related acute lung injury is suspected, test the product and patient for antineutrophil antibodies. [5.7]
- Because this product is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. [5.8]
ADVERSE REACTIONS
The most common adverse reactions to BIVIGAM (reported more than equal to 5% of clinical study subjects) were headache, fatigue, infusion site reaction, nausea, sinusitis, blood pressure increased, diarrhea, dizziness, and lethargy. [6]
To report SUSPECTED ADVERSE REACTIONS, contact Biotest Pharmaceuticals at (800) 458-4244 or FDA at 1-800-332-1088 or www.fda.gov/medwatch.
(6)
DRUG INTERACTIONS
Passive transfer of antibodies may transiently interfere with the immune response to live virus vaccines, such as measles, mumps, rubella, and varicella. [7] Passive transfer of antibodies may confound the results of serological testing. [5.10] (7)
USE IN SPECIFIC POPULATIONS
Pregnancy: Use in pregnant women has not been evaluated. Use BIVIGAM in pregnant women only if clearly needed. [8.1]
Geriatric Use: In patients over age 65 or in any patient at risk of developing renal insufficiency, do not exceed the recommended dose, and infuse BIVIGAM at the minimum infusion rate practicable. [8.5]
(8)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2014
FULL PRESCRIBING INFORMATION
WARNING: THROMBOSIS, RENAL DYSFUNCTION AND ACUTE RENAL FAILURE
- Thrombosis may occur with immune globulin (IGIV) products, including BIVIGAM. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors. (see Warnings and Precautions [5.1], Patient Counseling Information [17.2]
-
Use of Immune Globulin Intravenous (IGIV) products, particularly those containing sucrose, has been reported to be associated with renal dysfunction, acute renal failure, osmotic nephrosis, and death
1,2. Patients at risk of acute renal failure include those with any degree of pre-existing renal insufficiency, diabetes mellitus, advanced age (above 65 years of age), volume depletion, sepsis, paraproteinemia, or receiving known nephrotoxic drugs (see Warnings and Precautions [5.3]).
-
Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV products containing sucrose. BIVIGAM does not contain sucrose.
-
For patients at risk of thrombosis, renal dysfunction or renal failure, administer BIVIGAM at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity (see Dosage and Administration [2.2, 2.3], Warnings and Precautions [5.3]).
1 INDICATIONS AND USAGE
1.1 Primary Humoral Immunodeficiency
BIVIGAM is an Immune Globulin Intravenous (Human), 10% Liquid, indicated for the treatment of patients with primary humoral immunodeficiency (PI).
This includes, but is not limited to, the humoral immune defect in common variable immunodeficiency (CVID), X-linked agammaglobulinemia, congenital agammaglobulinemia, Wiskott-Aldrich syndrome, and severe combined immunodeficiencies.
2 DOSAGE AND ADMINISTRATION
For Intravenous Use Only
2.1 Preparation and Handling
- BIVIGAM is a clear or slightly opalescent, colorless to pale yellow solution. Inspect BIVIGAM visually for particulate matter and discoloration prior to administration. Do not use if the solution is cloudy or turbid, or contains particulate matter.
- Allow refrigerated product to come to room temperature before use.
- Do not freeze or heat. Do not use any solution that has been frozen or heated.
- DO NOT SHAKE.
- Do not mix BIVIGAM with other IGIV products or other intravenous medications. If large doses of BIVIGAM are to be administered, several vials may be pooled using aseptic technique into sterile infusion bags and infused.
- Do not dilute BIVIGAM.
- BIVIGAM contains no preservatives. BIVIGAM vial is for single use only. Any vial of BIVIGAM that has been entered should be used promptly and any unused portion should be discarded immediately. Do not reuse or save for future use.
- Maintain BIVIGAM at room temperature during administration.
- Do not use after expiration date.
2.2 Recommended Dose
As there are significant differences in the half-life of IgG among patients with primary humoral immunodeficiency, the frequency and amount of immunoglobulin therapy may vary from patient to patient. The proper amount can be determined by monitoring clinical response.
The recommended dose of BIVIGAM for replacement therapy in primary humoral immunodeficiency (PI) is 300 to 800 mg/kg body weight administered every 3 to 4 weeks. The dosage may be adjusted over time to achieve the desired trough levels and clinical response.
BIVIGAM dose adjustments may be required in patients who fail to maintain trough total IgG concentrations of at least 500 mg/dL with a target of 600 mg/dL. Starting with the second infusion, the dose will be adjusted proportionally, targeting a trough of more than equal to 600 mg/dL, based on the previous trough and the associated dose.
2.3 Administration
It has been reported that the frequency of adverse drug reactions to IGIV increases with the infusion rate. Initial infusion rates should be slow. If there are no adverse drug reactions, the infusion rate for subsequent infusions can be slowly increased to the maximum rate. For patients experiencing adverse drug reactions, it is advisable to reduce the infusion rate in subsequent infusions.
Table 1: Recommended Infusion Rates for BIVIGAM
Monitor patient vital signs throughout the infusion. Slow or stop the infusion if adverse reactions occur. If symptoms subside promptly, the infusion may be resumed at a lower rate that is comfortable for the patient.
Ensure that patients with pre-existing renal insufficiency are not volume depleted. For patients judged to be at risk for renal dysfunction or thrombotic events, administer BIVIGAM at the minimum infusion rate practicable, and consider discontinuation of administration if renal function deteriorates (see Boxed Warning, Warnings and Precautions [5.1, 5.3]
3 DOSAGE FORMS AND STRENGTHS
BIVIGAM is a liquid solution containing 10% IgG (100 mg/mL) for intravenous infusion.
4 CONTRAINDICATIONS
- BIVIGAM is contraindicated in patients who have had an anaphylactic or severe systemic reaction to the administration of human immune globulin.
- BIVIGAM is contraindicated in IgA deficiency patients with antibodies to IgA and a history of hypersensitivity.
5 WARNINGS AND PRECAUTIONS
5.1 Thrombosis
Thrombosis may occur following treatment with immune globulin products, including BIVIGAM.4,5,6 Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors
Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies. For patients at risk of thrombosis, administer BIVIGAM at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity (see Boxed Warning, Dosage and Administration [2.3], Patient Counseling Information [17.2]).
5.2 Hypersensitivity
Severe hypersensitivity reactions may occur with IGIV products, including BIVIGAM. In case of hypersensitivity, discontinue BIVIGAM infusion immediately and institute appropriate treatment. Medications such as epinephrine should be available for immediate treatment of acute hypersensitivity reactions.
BIVIGAM contains trace amounts of IgA (? 200 micrograms per milliliter) (see Description [11]). Patients with known antibodies to IgA may have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions. BIVIGAM is contraindicated in IgA deficient patients with antibodies against IgA and a history of hypersensitivity reaction (see Contraindications [4]).
5.3 Acute Renal Dysfunction and Acute Renal Failure
Acute renal dysfunction/failure, osmotic nephrosis, and death1,2 may occur upon use of human IGIV products. Ensure that patients are not volume depleted before administering BIVIGAM. Periodic monitoring of renal function and urine output is particularly important in patients judged to be at increased risk of developing acute renal failure.2 Assess renal function, including measurement of blood urea nitrogen (BUN) and serum creatinine, before the initial infusion of BIVIGAM and at appropriate intervals thereafter. If renal function deteriorates, consider discontinuing BIVIGAM (see Patient Counseling Information [17.1]). In patients who are at risk of developing renal dysfunction, because of pre-existing renal insufficiency or predisposition to acute renal failure (such as diabetes mellitus, hypovolemia, overweight, use of concomitant nephrotoxic medicinal products or age of more than 65 years), administer BIVIGAM at the minimum infusion rate practicable (see Dosage and Administration [2.3]).
5.4 Hyperproteinemia, Increased Serum Viscosity, and Hyponatremia
Hyperproteinemia, increased serum viscosity, and hyponatremia may occur in patients receiving IGIV therapy, including BIVIGAM. It is critical to clinically distinguish true hyponatremia from a pseudohyponatremia that is associated with or causally related to hyperproteinemia with concomitant decreased calculated serum osmolality or elevated osmolar gap, because treatment aimed at decreasing serum free water in patients with pseudohyponatremia may lead to volume depletion, a further increase in serum viscosity, and a possible predisposition to thrombotic events.
5.5 Aseptic Meningitis Syndrome (AMS)
AMS may occur infrequently with IGIV treatments including BIVIGAM. AMS usually begins within several hours to 2 days following IGIV treatment. Discontinuation of IGIV treatment has resulted in remission of AMS within several days without sequelae. 7,8,9
AMS is characterized by the following signs and symptoms: severe headache, nuchal rigidity, drowsiness, fever, photophobia, painful eye movements, nausea, and vomiting (see Patient Counseling Information [17.3]). Cerebrospinal fluid (CSF) studies frequently reveal pleocytosis up to several thousand cells per cubic millimeter, predominantly from the granulocytic series, and elevated protein levels up to several hundred mg/dL, but negative culture results. Conduct a thorough neurological examination on patients exhibiting such signs and symptoms, including CSF studies, to rule out other causes of meningitis.
AMS may occur more frequently in association with high doses (2 g/kg) and/or rapid infusion of IGIV.
5.6 Hemolysis
IGIV products, including BIVIGAM, may contain blood group antibodies that can act as hemolysins and induce in vivo coating of red blood cells (RBCs) with immunoglobulin, causing a positive direct antiglobulin reaction and, rarely, hemolysis. 10,11,12 Delayed hemolytic anemia can develop subsequent to IGIV therapy due to enhanced RBC sequestration,13 and acute hemolysis, consistent with intravascular hemolysis, has been reported.
Monitor patients for clinical signs and symptoms of hemolysis (see Patient Counseling Information [17.4]). If these are present after BIVIGAM infusion, perform appropriate confirmatory laboratory testing. If transfusion is indicated for patients who develop hemolysis with clinically compromising anemia after receiving IGIV, perform adequate cross-matching to avoid exacerbating on-going hemolysis.
5.7 Transfusion-Related Acute Lung Injury (TRALI)
Noncardiogenic pulmonary edema may occur in patients following IGIV treatment 14 including BIVIGAM. TRALI is characterized by severe respiratorydistress, pulmonary edema, hypoxemia, normal left ventricular function, and fever. Symptoms typically appear within 1 to 6 hours following treatment.
Monitor patients for pulmonary adverse reactions. If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies in both the product and the patient s serum (see Patient Counseling Information [17.5]).
TRALI may be managed using oxygen therapy with adequate ventilatory support.
5.8 Transmissible Infectious Agents
Because BIVIGAM is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
No cases of transmission of viral diseases or CJD have been associated with the use of BIVIGAM. All infections suspected by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Biotest Pharmaceuticals Corporation at
1-800-458-4244. Before prescribing BIVIGAM, the physician should discuss the risks and benefits of its use with the patient (see Patient Counseling Information [17.6]).
5.9 Monitoring Laboratory Tests
- Periodic monitoring of renal function and urine output is particularly important in patients judged to be at increased risk of developing acute renal failure. Assess renal function, including measurement of blood urea nitrogen (BUN) and serum creatinine, before the initial infusion of BIVIGAM and at appropriate intervals thereafter.
- Because of the potentially increased risk of thrombosis with IGIV treatment, consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies.
- If signs and/or symptoms of hemolysis are present after an infusion of BIVIGAM, perform appropriate laboratory testing for confirmation.
- If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies in both the product and patient s serum.
5.10 Interference with Laboratory Tests
After infusion of immunoglobulin, the transitory rise of the various passively transferred antibodies in the patient s blood may yield positive serological testing results, with the potential for misleading interpretation. Passive transmission of antibodies to erythrocyte antigens (e.g., A, B, and D) may cause a positive direct or indirect antiglobulin (Coombs) test.
6 ADVERSE REACTIONS
Serious adverse reactions observed in clinical trial subjects receiving BIVIGAM were vomiting and dehydration in one subject.
The most common adverse reactions to BIVIGAM (reported in more than equal to 5% of clinical study subjects) were headache, fatigue, infusion site reaction, nausea, sinusitis, blood pressure increased, diarrhea, dizziness, and lethargy.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials cannot be directly compared to rates in the clinical trials of another product and may not reflect the rates observed in clinical practice.
In a multicenter, open-label, non-randomized clinical trial, 63 subjects with PI, on regular IGIV replacement therapy, received doses of BIVIGAM ranging from 254 to 1029 mg/kg (median dose 462.8 mg/kg) every 3 weeks or 4 weeks for up to 12 months (mean 317.3 days; range 66 386 days) (see Clinical Studies [14]). The use of pre-medication was discouraged; however, if subjects required pre-medication (antipyretic, antihistamine, or antiemetic agent) for recurrent reactions to immune globulins, they were allowed to continue those medications for this trial. Of the 746 infusions administered, 41 (65%) subjects received premedication prior to 415 (56%) infusions.
Fifty-nine subjects (94%) had an adverse reaction at some time during the study. The proportion of subjects who had at least one adverse reaction was the same for both the 3- and 4-week cycles. The most common adverse reactions observed in this clinical trial were headache (32 subjects, 51%), sinusitis (24 subjects, 38%), fatigue (18 subjects, 29%), upper respiratory tract infection (16 subjects, 25%), diarrhea (13 subjects, 21%), cough (14 subjects, 22%), bronchitis (12 subjects, 19%), pyrexia (12 subjects, 19%), and nausea (9 subjects, 14%).
Adverse reactions (ARs) are those occurring during or within 72 hours after the end of an infusion. In this study, the upper bound of the 1-sided 95% confidence interval for the proportion of BIVIGAM infusions with one or more temporally associated adverse reactions was 31%. The total number of adverse reactions was 431 (a rate of 0.58 ARs per infusion).
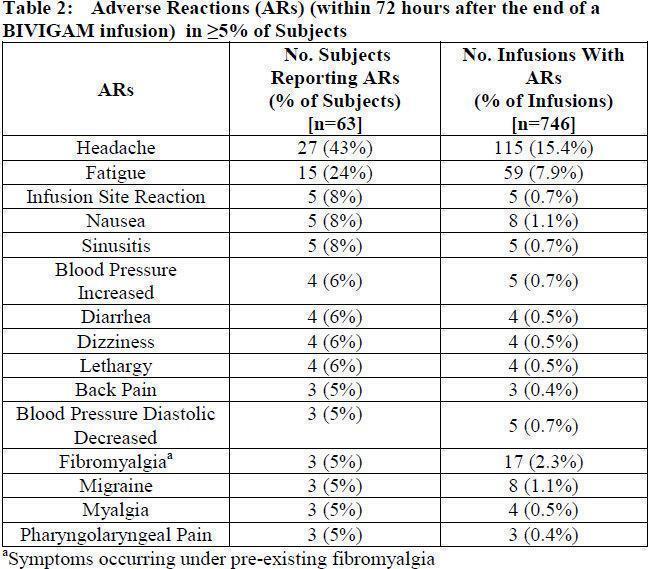
Seven subjects (11.1%) experienced 11 serious ARs. Two of these were related serious ARs (vomiting and dehydration) that occurred in one subject.
One subject withdrew from the study due to ARs related to BIVIGAM (lethargy, headache, tachycardia and pruritus).
All 63 subjects enrolled in this study had a negative direct antiglobulin (Coombs) test at baseline. During the study, no subjects showed clinical evidence of hemolytic anemia.
No cases of transmission of viral diseases or CJD have been associated with the use of BIVIGAM. During the clinical trial no subjects tested positive for infection due to human immunodeficiency virus (HIV), hepatitis B virus (HBV), or hepatitis C virus (HCV). There was a single positive finding for parvovirus (B19 virus) during the study. This subject came in contact with acute B19 virus from working at a school greeting children where a child was reported to have symptomatic Fifth's disease. There was no cluster (no other cases in other subjects) of B19 virus transmission with the IGIV batch concerned.
6.2 Postmarketing Experience
Because postmarketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure. The following adverse reactions have been identified and reported during the post-approval use of IGIV products:
- Respiratory: Apnea, Acute Respiratory Distress Syndrome (ARDS), Transfusion Associated Lung Injury (TRALI), cyanosis, hypoxemia, pulmonary edema, dyspnea, bronchospasm.
- Cardiovascular: Cardiac arrest, thromboembolism, vascular collapse, hypotension.
- Neurological: Coma, loss of consciousness, seizures, tremor.
- Integumentary: Stevens-Johnson syndrome, epidermolysis, erythema multiforme, bullous dermatitis.
- Hematologic: Pancytopenia, leukopenia, hemolysis, positive direct antiglobulin (Coombs) test.
- General/Body as a Whole: Pyrexia, rigors.
- Musculoskeletal: Back pain.
- Gastrointestinal: Hepatic dysfunction, abdominal pain.
7 DRUG INTERACTIONS
Passive transfer of antibodies may transiently interfere with the immune response to live virus vaccines, such as measles, mumps, rubella, and varicella. [7]
Passive transfer of antibodies may confound the results of serological testing. [5.10]
7. 1 Live Virus Vaccines
Immunoglobulin administration may transiently impair the efficacy of live attenuated virus vaccines such as measles, mumps, rubella, and varicella because the continued presence of high levels of passively acquired antibody may interfere with an active antibody response.15,16 The immunizing physician should be informed of recent therapy with BIVIGAM so that appropriate measures may be taken (see Patient Counseling Information [17.7]).
8 USE IN SPECIFIC POPULATION
8.1 Pregnancy
Pregnancy Category C. Animal reproduction studies have not been conducted with BIVIGAM. It is not known whether BIVIGAM can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. BIVIGAM should be given to pregnant women only if clearly needed.17,18
8.3 Nursing Mothers
Use of BIVIGAM in nursing mothers has not been evaluated. BIVIGAM should be given to nursing mothers only if clearly needed.
8.4 Pediatric Use
BIVIGAM was evaluated in 9 pediatric patients (4 children ages 6 11 years and 5 adolescents ages 12 16 years) with PI. This number of pediatric patients was too small for safety or efficacy. The safety and effectiveness of BIVIGAM has not been established in pediatric patients with PI who are under the age of 6 (see Clinical Studies [14]).
8.5 Geriatric Use
BIVIGAM should be used with caution in patients age 65 and over who are judged to be at increased risk of developing renal insufficiency or thrombotic events(see Boxed Warning, Warnings and Precautions [5.1, 5.3]). Do not exceed recommended doses and administer BIVIGAM at the minimum infusion rate practicable.
BIVIGAM was evaluated in 9 patients age 65 and older with PI. This number of geriatric patients is not being sufficient to determine whether they respond differently from younger patients (see Clinical Studies [14]).
11 DESCRIPTION
BIVIGAM is a purified, sterile, ready-to-use preparation of concentrated human immunoglobulin G (IgG) antibodies. The distribution of IgG subclasses is similar to that of normal plasma.
19,20 The active ingredient is human immunoglobulin purified from source human plasma and processed using a modified classical Cohn Method 6 / Oncley Method 9 fractionation procedure. BIVIGAM contains 100 10 mg/mL protein, of which not less than 96% is human immunoglobulin obtained from source human plasma. It is formulated in water for injection containing 0.100-0.140 M sodium chloride, 0.20-0.29 M glycine, 0.15 0.25% polysorbate 80, and pH 4.0 4.6. BIVIGAM contains ? 200 ?g/mL of IgA. Each plasma donation used for the manufacture of BIVIGAM is collected from FDA licensed facilities and undergoes rigorous testing. Plasma donations must test negative for hepatitis B virus (HBV) surface antigen (HBsAg), antibodies to human immunodeficiency virus (HIV) strains 1 and 2 (anti-HIV-1/2), and antibodies to the hepatitis C virus (anti-HCV) as determined by enzyme immuno assay (EIA). In addition, each plasma unit must test negative and/or non-reactive for HIV RNA, HCV RNA, HBV DNA, Hepatitis A Virus (HAV) RNA, and Parvovirus B19 (B19 virus) DNA as determined by Nucleic Acid Amplification Testing (NAT) of plasma minipools. NAT for B19 virus DNA is also performed on a sample of the manufacturing pool and the limit for B19 virus DNA in a manufacturing pool is set not to exceed 104 IU/mL.
The manufacturing process of BIVIGAM employs three steps to remove/inactivate adventitious viruses to minimize the risk of virus transmission. The steps are "Precipitation and removal of fraction III" during cold ethanol fractionation, classical "Solvent/detergent treatment" and "35 nm virus filtration". In compliance with current guidelines, the steps have been separately validated in a series of
in vitro experiments for their capacity to inactivate or remove both enveloped and non-enveloped viruses. Precipitation and removal of fraction III removes both enveloped and non-enveloped viruses, solvent/detergent treatment represents a virus inactivation step for enveloped viruses, and 35 nm virus filtration removes both enveloped and non-enveloped viruses by size exclusion. In addition to the steps above, low pH during several steps of the production process contributes to virus inactivation. The results of virus validation studies for BIVIGAM are shown in Table 3, expressed as log10 reduction factors.
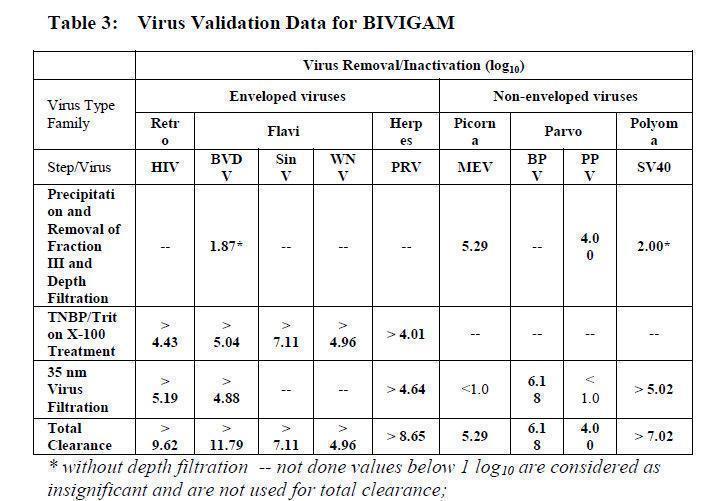
HIV, human immunodeficiency virus; BVDV, Bovine viral diarrhea virus, model virus for HCV; SinV, Sindbis virus, model virus for HCV; WNV, West Nile virus; PRV, Pseudorabies virus, model virus for herpes viruses and Hepatitis B virus; MEV, Murine encephalomyelitis virus, model virus for hepatitis A virus; BPV, Bovine parvovirus, model virus for human B19 virus; PPV, Porcine parvo virus, model virus for human B19 virus; SV40, Simian virus 40, model virus for highly resistant non- enveloped viruses.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
BIVIGAM is a replacement therapy in patients with primary humoral immunodeficiency (PI) (e.g. agammaglobulinaemia, hypogammaglobulinaemia, CVID, SCID).
The broad spectrum of neutralizing IgG antibodies against bacterial and viral pathogens and their toxins helps to avoid recurrent serious opportunistic infections. IgG antibodies are opsonins that increase phagocytosis and elimination of pathogens from the circulation. The mechanism of action has not been fully elucidated in PI.
12.3 Pharmacokinetics
In the clinical study assessing the efficacy and safety of BIVIGAM in 63 subjects with PI (
see Clinical Studies [14.1]), serum concentrations of total IgG and IgG subclasses were measured in 21 subjects (ages 18 to 75) following the 4th infusion for the 5 subjects on the 3-week dosing interval and following the 5th infusion for the 16 subjects on the 4-week dosing interval. The dose of BIVIGAM used in these subjects ranged from 300 mg/kg to 800 mg/kg. After the infusion, blood samples were taken until Day 21 and Day 28 for the 3-week and 4-week dosing intervals, respectively. Table 4 summarizes the Total IgG Pharmacokinetic Parameters of BIVIGAM, based on serum concentrations of total IgG. Trough concentrations were maintained throughout the study for both treatment cycles and mean trough concentrations were well above the target trough concentration of 500 mg/dL for both treatment cycles in pediatric (? 6 years old) as well as adult subjects at all time points.
Table 4: Total IgG Pharmacokinetic Parameter Estimates (PK Population) in Adults
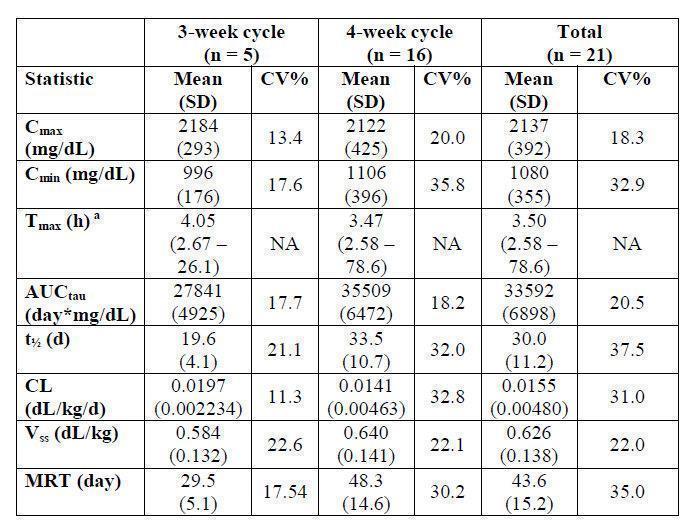
AUCtau = steady-state area under the plasma concentration versus time curve with tau = dosing interval; CL = total body clearance; Cmax = maximum concentration; Cmin = minimum concentration; CV = coefficient of variation; n = number of subjects; NA = not applicable; SD = standard deviation; Tmax = time of maximum concentration; t = terminal half-life; Vss = Volume of distribution steady-state; MRT = mean residence time; a Median and Range. The median terminal half-life of BIVIGAM was 30 days for the 21 subjects. Mean trough IgG subclass levels were consistent with physiological values.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies were conducted to evaluate the carcinogenic or mutagenic effects of BIVIGAM or its effects on fertility.
13.2 Animal Toxicology and/or Pharmacology
No animal studies were conducted to evaluate possible toxicity of BIVIGAM in animals.
BIVIGAM contains Polysorbate 80 at a concentration of up to 2.5 mg/mL. Intravenous administrations of Polysorbate 80 in multiple species have been linked with a decrease in blood pressure. In rats, single doses of Polysorbate 80 that were up to 25 times higher than the amount from 800 mg/kg BIVIGAM resulted in an increase of liver enzymes and total bilirubin.
14 CLINICAL STUDIES
14. 1 Treatment of Primary Humoral Immunodeficiency
?A prospective, open-label, single-arm, multicenter trial assessed the efficacy, safety, and pharmacokinetics of BIVIGAM in adult and pediatric subjects with PI. Study subjects were receiving regular IGIV replacement therapy, with a stable dose between 300 and 800 mg/kg for at least 3 months prior to participation. Subjects received a BIVIGAM infusion administered every 3 or 4 weeks (both the dose and schedule depending on their prior therapy) for approximately 1 year.
A total of 63 subjects were enrolled in the trial, 31 men and 32 women with a mean age of 41 years. Fourty-four subjects were adults (70%) between 18 and 64 years of age. There were 9 pediatric subjects (
see Pediatric Use [8.4]), and 9 elderly subjects (14%, ?65 years of age). The oldest subject was 75 years of age. There were 17 subjects with a 3-week cycle and 46 subjects with a 4-week cycle. There were 51 subjects (81%) with common variable immunodeficiency as their primary diagnosis, followed by X-linked agammaglobulinemia and Other (9.5% each). The intent to treat (ITT) population included 58 subjects and was used for efficacy analysis.
The primary endpoint of the study was to assess the efficacy of BIVIGAM in preventing serious bacterial infections (SBIs) defined as rate of less than 1.0 cases of bacterial pneumonia, bacteremia/septicemia, osteomyelitis/septic arthritis, visceral abscess, and bacterial meningitis per person-year. Secondary efficacy parameters included time to first SBI and time to first infection of any kind/seriousness, days on antibiotics (excluding prophylaxis), days offschool/work due to infections, all confirmed infections of any kind or seriousness, and hospitalizations due to infection.
During the 12-month study period, two serious acute bacterial infections occurred in two subjects with an onset date between the first infusion of BIVIGAM and the first follow-up visit, inclusive. Thus, the mean event rate of serious, acute, bacterial infections per year was 0.037 (with an upper 1-sided 99% confidence interval of 0.101, which met the study s primary efficacy endpoint).
The two SBIs were cases of bacterial pneumonia. Thirty-three percent of subjects had days off work or school due to an infection. Of the 197 infections reported, 2 resulted in hospitalization. Results for the pediatric subjects were similar to those for the adult subjects. (see Table 5).
Table 5: Summary of Efficacy Results in Subjects with PI
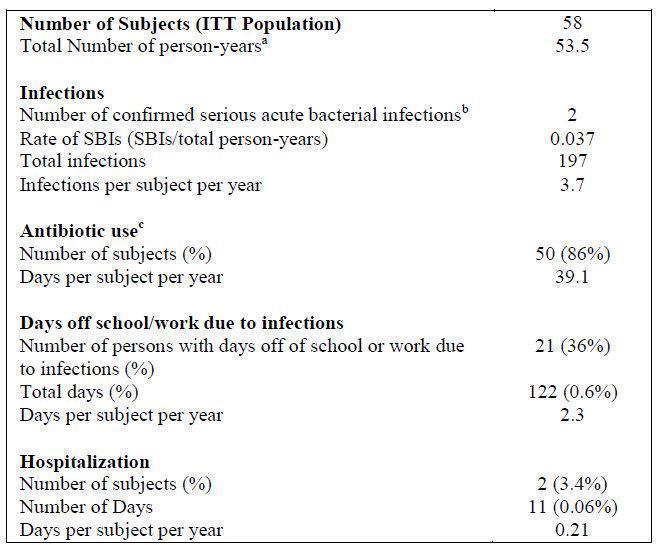
SBI = serious bacterial infections.
aPerson-years: Person-time in years with 2 decimals = (the Final Clinical Visit Date - the Day 0 date+1) / 365.25, where the final clinical visit date is defined as the specimen collection date of the final clinical visit for urinalysis, or the specimen collection date for the clinical laboratory tests at the final clinical visit and Day 0 date is the start date of the first BIVIGAM infusion.
b Defined as bacterial pneumonia, bacterial meningitis, bacteremia/septicemia, osteomyelitis/septic arthritis, and visceral abscess.
c The calculation of antibiotic use excludes 8 subjects who were on antibiotics throughout the study either prophylactically or for ongoing or recurrent conditions.
15 REFERENCES
1. Gupta N, Ahmed I, Nissel-Horowitz S, Patel D, Mehrotra B. Intravenous gammaglobulin-associated acute renal failure. Am J Hematol 2001; 66:151-152.
2. Cayco, A.V., M.A. Perazella, and J.P. Hayslett. Renal insufficiency after intravenous immune globulin therapy: a report of two cases and an analysis of the literature. J Am Soc Nephrol 1997; 8:1788-1794.
3. Steinberger BA, Ford SM, Coleman TA. Intravenous immune globulin therapy results in post-infusional hyperproteinemia, increased serum viscosity, and pseudohyponatremia. Am J Hematol 2003; 73:97-100.
4. Dalakas MC. High-dose intravenous immunoglobulin and serum viscosity: risk of precipitating thromboembolic events. Neurology 1994; 44:223-226.
5. Woodruff RK, Grigg AP, Firkin FC, Smith IL. Fatal thrombotic events during treatment of autoimmune thrombocytopenia with intravenous immunoglobulin in elderly patients. Lancet 1986; 2:217-218.
6. Wolberg AS, Kon RH, Monroe DM, Hoffman M. Coagulation factor XI is a contaminant in intravenous immunoglobulin preparations. Am J Hematol 2000; 65:30-34.
7. Casteels-Van Daele, M., et al. Intravenous immune globulin and acute aseptic meningitis [letter]. N Engl J Med 1990; 323:614-615.
8. Kato, E., et al. Administration of immune globulin associated with aseptic meningitis [letter]. Jama 1988; 259:3269-3271.
9. Scribner, C.L., et al. Aseptic meningitis and intravenous immunoglobulin therapy [editorial, comment]. Ann Intern Med 1994; 121:305-306.
10. Copelan EA, Stohm PL, Kennedy MS, Tutschka PJ. Hemolysis following intravenous immune globulin therapy. Transfusion 1986; 26:410-412.
11. Thomas MJ, Misbah SA, Chapel HM, Jones M, Elrington G, Newsom-Davis J. Hemolysis after high-dose intravenous Ig. Blood 1993; 15:3789.
12. Wilson JR, Bhoopalam N, Fisher M. Hemolytic anemia associated with intravenous immunoglobulin. Muscle and Nerve 1997; 20:1142-1145.
13. Kessary-Shoham H, Levy Y, Shoenfeld Y, Lorber M, Gershon H. In vivo administration of intravenous immunoglobulin (IVIg) can lead to enhanced erythrocyte sequestration. J Autoimmune 1999; 13:129-135.
14. Rizk A, Gorson KC, Kenney L, Weinstein R. Transfusion-related acute lung injury after the infusion of IVIG. Transfusion 2001; 41:264-268.
15. Siber GA, Werner BG, Halsey NA, et al. Interference of immune globulin with measles and rubella immunization. J Pediatr 1993; 122:204-211.
16. Salisbury D, Ramsay M, Noakes K, eds. Immunisation against infectious disease. The Stationery Office (TSO), London: UK Department of Health; 2009:426.
17. Hammarstrom L, Smith CIE. Placental transfer of intravenous immunoglobulin. Lancet 1986; 1:681.
18. Sidiropoulos D, Herrmann U, Morell A, von Muralt G, Barandun S. Transplacental passage of intravenous immunoglobulin in the last trimester of pregnancy. J Pediatr 1986; 109:505-508.
19. Skvaril F. Qualitative and quantitative aspects of IgG subclasses in i.v. immunoglobulin preparations. In: Nydegger UE, ed. Immunotherapy. London: Academic Press; 1981:118-122.
20. French M. Serum IgG subclasses in normal adults. Monogr Allergy 1986, 19:100-107.
16 HOW SUPPLIED/STORAGE AND HANDLING
BIVIGAM is supplied in a single-use, tamper-evident vial. The components used in the packaging for BIVIGAM are latex free.
BIVIGAM is supplied in the following sizes:
| NDC Number | Size | Grams Protein |
| 59730-6502-1 | 50ml | 5 |
| 59730-6503-1 | 100ml | 10 |
Storage
Refrigerate between 2 to 8 degrees C (36 to 46 degrees F).
Special Precautions for Storage
Do not freeze or heat. Do not use any solutions that have been frozen or heated.
Allow refrigerated product to come to room temperature before use. Do not use after expiration date.
Shelf-life
BIVIGAM may be stored until expiration date on vial packaging at 2 to 8 degrees C (36 to 46 degrees F).
Incompatibilities
Do not dilute.
BIVIGAM should be infused using a separate line by itself, without mixing with other intravenous fluids or medications the patient may be receiving.
17 PATIENT COUNSELING INFORMATION
17.1 Acute Renal Dysfunction and Acute Renal Failure
Instruct patients to immediately report symptoms of decreased urine output, sudden weight gain, fluid retention/edema, and/or shortness of breath. Such symptoms may suggest kidney damage (see Boxed Warning, Warnings and Precautions [5.3]).
17.2 Thrombosis
Instruct patients to immediately report symptoms of thrombosis. These symptoms may include: pain and/or swelling of an arm or legs/feet with warmth over the affected area, discoloration of an arm or leg, unexplained shortness of breath, acute chest pain or discomfort that worsens on deep breathing, unexplained rapid pulse, numbness or weakness on one side of the body., (see Warnings and Precautions [5.1]).
17.3 Aseptic Meningitis Syndrome (AMS)
Instruct patients to immediately report signs and symptoms of AMS. These symptoms include severe headache, neck stiffness, drowsiness, fever, sensitivity to light, painful eye movements, nausea and vomiting (see Warnings and Precautions [5.5]).
17.4 Hemolysis
Instruct patients to immediately report signs and symptoms of hemolysis. These symptoms include fatigue, increased heart rate, yellowing of skin or eyes, dark-colored urine (see Warnings and Precautions [5.6]).
17.5 Transfusion-Related Acute Lung Injury (TRALI)
Instruct patients to immediately report signs and symptoms of TRALI. These symptoms include trouble breathing, chest pain, blue lips or extremities, fever (see Warnings and Precautions [5.7]).
17.6 Transmissible Infectious Agents
Inform patients that BIVIGAM is made from human plasma and may contain infectious agents that can cause disease. While the risk that BIVIGAM can transmit an infection has been reduced by screening plasma donors for prior exposure, testing donated plasma, and inactivating or removing certain viruses during manufacturing, patients should report any symptoms that concern them (see Description [11] and Warnings and Precautions [5.8]).
17.7 Live Virus Vaccines
Inform patients that BIVIGAM can interfere with their immune response to live viral vaccines (e.g., measles, mumps, rubella, and varicella), and instruct patients to notify their healthcare professional of this potential interaction when they are receiving vaccinations (see Drug Interactions [7]).
| BIVIGAM
human immunoglobulin g injection, solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| BIVIGAM
human immunoglobulin g injection, solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - ADMA Biologics, Inc (117213235) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ADMA Biologics, Inc | 117213235 | manufacture(59730-6502, 59730-6503) | |