DESCRIPTION
Fenofibrate, USP (micronized), is a lipid regulating agent available as capsules for oral administration. Each capsule contains 67 mg, 134 mg or 200 mg of micronized fenofibrate, USP. The chemical name for fenofibrate, USP is 2-[4-(4- chlorobenzoyl) phenoxy]-2-methyl-propanoic acid, 1-methylethyl ester with the following structural formula:
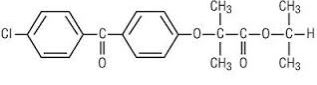
The empirical formula is C20H21O4Cl and the molecular weight is 360.83; fenofibrate, USP is very soluble in methylene chloride; slightly soluble in alcohol; practically insoluble in water. The melting point is 79° to 82°C. Fenofibrate, USP is a white or almost white crystalline powder which is stable under ordinary conditions.
Each 67 mg fenofibrate capsule, USP (micronized) contains the following inactive ingredients: sodium lauryl sulfate, croscarmellose sodium, pregelatinized starch, microcrystalline cellulose, colloidal silicon dioxide, sodium stearyl fumarate, titanium dioxide and gelatin. The capsule shell imprinting ink contains the following inactive ingredients: shellac, black iron oxide and potassium hydroxide.
Each 134 mg fenofibrate capsule, USP (micronized) contains the following inactive ingredients: sodium lauryl sulfate, croscarmellose sodium, pregelatinized starch, microcrystalline cellulose, colloidal silicon dioxide, sodium stearyl fumarate, titanium dioxide, FD&C Yellow 6, D&C Yellow 10 and gelatin. The capsule shell imprinting ink contains the following inactive ingredients: shellac, black iron oxide and potassium hydroxide.
Each 200 mg fenofibrate capsule, USP (micronized) contains the following inactive ingredients: sodium lauryl sulfate, croscarmellose sodium, pregelatinized starch, microcrystalline cellulose, colloidal silicon dioxide, sodium stearyl fumarate, titanium dioxide, FD&C Yellow 6, D&C Yellow 10 and gelatin. The capsule shell imprinting ink contains the following inactive ingredients: shellac, black iron oxide and potassium hydroxide.
CLINICAL PHARMACOLOGY
A variety of clinical studies have demonstrated that elevated levels of total cholesterol (total-C), low density lipoprotein cholesterol (LDL-C), and apolipoprotein B (apo B), an LDL membrane complex, are associated with human atherosclerosis. Similarly, decreased levels of high density lipoprotein cholesterol (HDL-C) and its transport complex, apolipoprotein A (apo AI and apo AII) are associated with the development of atherosclerosis. Epidemiologic investigations have established that cardiovascular morbidity and mortality vary directly with the level of total-C, LDL-C, and triglycerides, and inversely with the level of HDL-C. The independent effect of raising HDL-C or lowering triglycerides (TG) on the risk of cardiovascular morbidity and mortality has not been determined.
Fenofibric acid, the active metabolite of fenofibrate, produces reductions in total cholesterol, LDL cholesterol, apolipoprotein B, total triglycerides and triglyceride rich lipoprotein (VLDL) in treated patients. In addition, treatment with fenofibrate results in increases in high density lipoprotein (HDL) and apoproteins apo AI and apo AII.
The effects of fenofibric acid seen in clinical practice have been explained in vivo in transgenic mice and in vitro in human hepatocyte cultures by the activation of peroxisome proliferator activated receptor α (PPARα). Through this mechanism, fenofibrate increases lipolysis and elimination of triglyceride-rich particles from plasma by activating lipoprotein lipase and reducing production of apoproteins C-III (an inhibitor of lipoprotein lipase activity). The resulting fall in triglycerides produces an alteration in the size and composition of LDL from small, dense particles (which are thought to be atherogenic due to their susceptibility to oxidation), to large buoyant particles. These larger particles have a greater affinity for cholesterol receptors and are catabolized rapidly. Activation of PPARα also induces an increase in the synthesis of apoproteins A-I, A-II and HDL-cholesterol.
Fenofibrate also reduces serum uric acid levels in hyperuricemic and normal individuals by increasing the urinary excretion of uric acid.
Pharmacokinetics/Metabolism
Clinical experience has been obtained with two different formulations of fenofibrate: a “micronized” and “non-micronized” formulation, which have been demonstrated to be bioequivalent. Comparisons of blood levels following oral administration of both formulations in healthy volunteers demonstrate that a single capsule containing 67 mg of the “micronized” formulation is bioequivalent to 100 mg of the “non-micronized” formulation. Three capsules containing 67 mg fenofibrate (micronized) are bioequivalent to a single 200 mg fenofibrate (micronized) capsule.
Absorption
The absolute bioavailability of fenofibrate cannot be determined as the compound is virtually insoluble in aqueous media suitable for injection. However, fenofibrate is well absorbed from the gastrointestinal tract. Following oral administration in healthy volunteers, approximately 60% of a single dose of radiolabelled fenofibrate appeared in urine, primarily as fenofibric acid and its glucuronate conjugate, and 25% was excreted in the feces. Peak plasma levels of fenofibric acid occur within 6 to 8 hours after administration.
The absorption of fenofibrate is increased when administered with food. With micronized fenofibrate, the absorption is increased by approximately 35% under fed as compared to fasting conditions.
Distribution
In healthy volunteers, steady-state plasma levels of fenofibric acid were shown to be achieved within 5 days of dosing with single oral doses equivalent to 67 mg of fenofibrate and did not demonstrate accumulation across time following multiple dose administration. Serum protein binding was approximately 99% in normal and hyperlipidemic subjects.
Metabolism
Following oral administration, fenofibrate is rapidly hydrolyzed by esterases to the active metabolite, fenofibric acid; no unchanged fenofibrate is detected in plasma.
Fenofibric acid is primarily conjugated with glucuronic acid and then excreted in urine. A small amount of fenofibric acid is reduced at the carbonyl moiety to a benzhydrol metabolite which is, in turn, conjugated with glucuronic acid and excreted in urine.
In vivometabolism data indicate that neither fenofibrate nor fenofibric acid undergo oxidative metabolism
(e.g., cytochrome P450) to a significant extent.
Excretion
After absorption, fenofibrate is mainly excreted in the urine in the form of metabolites, primarily fenofibric acid and fenofibric acid glucuronide. After administration of radiolabelled fenofibrate, approximately 60% of the dose appeared in the urine and 25% was excreted in the feces.
Fenofibric acid is eliminated with a half-life of 20 hours, allowing once daily administration in a clinical setting.
Special Populations
Geriatrics
In elderly volunteers 77 to 87 years of age, the oral clearance of fenofibric acid following a single oral dose of fenofibrate was 1.2 L/h, which compares to 1.1 L/h in young adults. This indicates that a similar dosage regimen can be used in the elderly, without increasing accumulation of the drug or metabolites.
Pediatrics
Fenofibrate has not been investigated in adequate and well-controlled trials in pediatric patients.
Race
The influence of race on the pharmacokinetics of fenofibrate has not been studied, however fenofibrate is not metabolized by enzymes known for exhibiting inter-ethnic variability. Therefore, inter-ethnic pharmacokinetic differences are very unlikely.
Renal Insufficiency
The pharmacokinetics of fenofibric acid was examined in patients with mild, moderate, and severe renal impairment. Patients with severe renal impairment (creatinine clearance [CrCl] ≤ 30 mL/min) showed 2.7-fold increase in exposure for fenofibric acid and increased accumulation of fenofibric acid during chronic dosing compared to that of healthy subjects. Patients with mild to moderate renal impairment (CrCl 30 to 80 mL/min) had similar exposure but an increase in the half-life for fenofibric acid compared to that of healthy subjects. Based on these findings, the use of fenofibrate should be avoided in patients who have severe renal impairment and dose reduction is required in patients having mild to moderate renal impairment.
Hepatic Insufficiency
No pharmacokinetic studies have been conducted in patients having hepatic insufficiency.
Drug-drug Interactions
In vitro studies using human liver microsomes indicate that fenofibrate and fenofibric acid are not inhibitors of cytochrome (CYP) P450 isoforms CYP3A4, CYP2D6, CYP2E1, or CYP1A2. They are weak inhibitors of CYP2C19 and CYP2A6, and mild-to-moderate inhibitors of CYP2C9 at therapeutic concentrations.
Potentiation of coumarin-type anti-coagulants has been observed with prolongation of the prothrombin time/INR.
Bile acid sequestrants have been shown to bind other drugs given concurrently. Therefore, fenofibrate should be taken at least 1 hour before or 4 to 6 hours after a bile acid binding resin to avoid impeding its absorption. (see WARNINGS and PRECAUTIONS).
Concomitant administration of a single dose of fenofibrate (administered as 3 x 67 mg fenofibrate capsules) with a single dose of pravastatin (40 mg) in 23 healthy subjects increased the mean Cmax and mean AUC for pravastatin by 13%. The Cmax and AUC of fenofibrate decreased by 2% and 1%, respectively, after concomitant administration of fenofibrate and pravastatin. The mean Cmax and AUC for 3α-hydroxy-iso-pravastatin increased by 29% and 26%, respectively.
Concomitant administration of a single dose of fenofibrate (equivalent to 145 mg fenofibrate) and a single dose of fluvastatin (40 mg) resulted in a small increase (approximately 15% to 16%) in exposure to (+)3R,5S-fluvastatin, the active enantiomer of fluvastatin.
A single dose of either pravastatin or fluvastatin had no clinically important effect on the pharmacokinetics of fenofibric acid.
Concomitant administration of fenofibrate (equivalent to fenofibrate 200 mg) with atorvastatin (20 mg) once daily for 10 days resulted in approximately 17% decrease (range from 67% decrease to 44% increase) in atorvastatin AUC values in 22 healthy males. The atorvastatin Cmax values were not significantly affected by fenofibrate. The pharmacokinetics of fenofibric acid were not significantly affected by atorvastatin.
Concomitant administration of fenofibrate (equivalent to fenofibrate 200 mg) once daily for 10 days with glimepiride (1 mg tablet) single dose simultaneously with the last dose of fenofibrate resulted in a 35% increase in mean AUC of glimepiride in healthy subjects. Glimepiride Cmax was not significantly affected by fenofibrate coadministration. There was no statistically significant effect of multiple doses of fenofibrate on glucose nadir or AUC with the baseline glucose concentration as the covariate after glimepiride administration in healthy volunteers. However, glucose concentrations at 24 hours remained statistically significantly lower after pretreatment with fenofibrate than with glimepiride alone. Glimepiride had no significant effect on the pharmacokinetics of fenofibric acid.
Concomitant administration of fenofibrate (54 mg) and metformin (850 mg) three times a day for 10 days resulted in no significant changes in the pharmacokinetics of fenofibric acid and metformin when compared with the two drugs administered alone in healthy subjects.
Concomitant administration of fenofibrate (equivalent to fenofibrate 200 mg) once daily for 14 days with rosiglitazone tablet (rosiglitazone maleate) (8 mg) once daily for 5 days, Day 10 through Day 14, resulted in no significant changes in the pharmacokinetics of fenofibric acid and rosiglitazone when compared with the two drugs administered alone in healthy subjects.
Clinical Trials
Hypercholesterolemia (Heterozygous Familial and Nonfamilial) and Mixed Dyslipidemia (Fredrickson Types IIa and IIb)
The effects of fenofibrate at a dose equivalent to 200 mg fenofibrate capsules per day were assessed from four randomized, placebo-controlled, double-blind, parallel-group studies including patients with the following mean baseline lipid values: total-C 306.9 mg/dL; LDL-C 213.8 mg/dL; HDL-C 52.3 mg/dL; and triglycerides
191 mg/dL. Fenofibrate capsules therapy lowered LDL-C, total-C, and the LDL-C/HDL-C ratio. Fenofibrate capsules therapy also lowered triglycerides and raised HDL-C (see Table 1).
| Treatment Group | Total-C | LCL-C | HDL-C | TG |
| Pooled Cohort | ||||
| Mean baseline lipid values (n=646) | 306.9 mg/dL | 213.8 mg/dL | 52.3 mg/dL | 191 mg/dL |
| All FEN (n=361) | -18.7%b | -20.6%b | +11%b | -28.9%b |
| Placebo (n=285) | -0.4% | -2.2% | +0.7% | +7.7% |
|
Baseline LDL-C > 160 mg/dL and TG < 150 mg/dL (Type IIa) | ||||
| Mean baseline lipid values (n=334) | 307.7 mg/dL | 227.7 mg/dL | 58.1 mg/dL | 101.7 mg/dL |
| All FEN (n=193) | -22.4%b | -31.4%b | +9.8% | -23.5%b |
| Placebo (n=141) | +0.2% | -2.2% | +2.6% | +11.7% |
| Baseline LDL-C > 160 mg/dL and TG < 150 mg/dL (Type IIb) | ||||
| Mean baseline lipid values (n=646) | 312.8 mg/dL | 219.8 mg/dL | 46.7 mg/dL | 231.9 mg/dL |
| All FEN (n=126) | -16.8%b | -20.1%b | +14.6%b | -35.9%b |
| Placebo (n=116) | -3% | -6.6% | +2.3% | +0.9% |
a Duration of study treatment was 3 to 6 months
b p = <0.05 vs. Placebo
In a subset of the subjects, measurements of apo B were conducted. Fenofibrate treatment significantly reduced apo B from baseline to endpoint as compared with placebo (-25.1% vs. 2.4%, p<0.0001, n=213 and 143 respectively).
Hypertriglyceridemia (Fredrickson Type IV and V)
The effects of fenofibrate on serum triglycerides were studied in two randomized, double-blind, placebo-controlled clinical trials1 of 147 hypertriglyceridemia patients (Fredrickson Type IV and V). Patients were treated for eight weeks under protocols that differed only in that one entered patients with baseline triglyceride (TG) levels of 500 to 1,500 mg/dL, and the other TG levels of 350 to 500 mg/dL. In patients with hypertriglyceridemia and normal cholesterolemia with or without hyperchylomicronemia (Type IV/V hyperlipidemia), treatment with fenofibrate at dosages equivalent to 200 mg fenofibrate per day decreased primarily very low density lipoprotein (VLDL) triglycerides and VLDL cholesterol. Treatment of patients with Type IV hyperlipoproteinemia and elevated triglycerides often results in an increase of low density lipoprotein (LDL) cholesterol (see Table 2).
Table 2: Effects of Fenofibrate Capsules in Patients with Fredrickson Type IV/V Hyperlipidemia
|
Study 1 Baseline TG levels 350 to 499 mg/dL | Placebo | Fenofibrate Capsules | |||||||
| N | Baseline (Mean) | Endpoint (Mean) | % Change (Mean) | N | Baseline (Mean) | Endpoint (Mean) | % Change (Mean) | ||
| Triglycerides | 28 | 449 | 450 | -0.5 | 27 | 432 | 223 | -46.2* | |
| VLDL Triglycerides | 19 | 367 | 350 | 2.7 | 19 | 350 | 178 | -44.1* | |
| Total Cholesterol | 28 | 255 | 261 | 2.8 | 27 | 252 | 227 | -9.1* | |
| HDL Cholesterol | 28 | 35 | 36 | 4 | 27 | 34 | 40 | 19.6* | |
| LDL Cholesterol | 28 | 120 | 129 | 12 | 27 | 128 | 137 | 14.5 | |
| VLDL Cholesterol | 27 | 99 | 99 | 5.8 | 27 | 92 | 46 | -44.7* | |
| Study 2 Baseline TG levels 500 to 1500 mg/dL | Placebo | Fenofibrate Capsules | |||||||
| N | Baseline (Mean) | Endpoint (Mean) | % Change (Mean) | N | Baseline (Mean) | Endpoint (Mean) | % Change (Mean) | ||
| Triglycerides | 44 | 710 | 750 | 7.2 | 48 | 726 | 308 | -54.5* | |
| VLDL Triglycerides | 29 | 537 | 571 | 18.7 | 33 | 543 | 205 | -50.6* | |
| Total Cholesterol | 44 | 272 | 271 | 0.4 | 48 | 261 | 223 | -13.8* | |
| HDL Cholesterol | 44 | 27 | 28 | 5 | 48 | 30 | 36 | 22.9* | |
| LDL Cholesterol | 42 | 100 | 90 | -4.2 | 45 | 103 | 131 | 45* | |
| VLDL Cholesterol | 42 | 137 | 142 | 11 | 45 | 126 | 54 | -49.4* | |
* = p <0.05 vs. Placebo
The effect of fenofibrate on cardiovascular morbidity and mortality has not been determined.
INDICATIONS AND USAGE
Treatment of Hypercholesterolemia
Fenofibrate capsules are indicated as adjunctive therapy to diet for the reduction of LDL-C, Total-C, Triglycerides and Apo B in adult patients with primary hypercholesterolemia or mixed dyslipidemia (Fredrickson Types IIa and IIb). Lipid-altering agents should be used in addition to a diet restricted in saturated fat and cholesterol when response to diet and non-pharmacological interventions alone has been inadequate (see National Cholesterol Education Program [NCEP] Treatment Guidelines, below).
Treatment of Hypertriglyceridemia
Fenofibrate capsules are also indicated as adjunctive therapy to diet for treatment of adult patients with hypertriglyceridemia (Fredrickson Types IV and V hyperlipidemia).
Improving glycemic control in diabetic patients showing fasting chylomicronemia will usually reduce fasting triglycerides and eliminate chylomicronemia thereby obviating the need for pharmacologic intervention.
Markedly elevated levels of serum triglycerides (e.g. > 2,000 mg/dL) may increase the risk of developing pancreatitis. The effect of fenofibrate therapy on reducing this risk has not been adequately studied.
Drug therapy is not indicated for patients with Type I hyperlipoproteinemia, who have elevations of chylomicrons and plasma triglycerides, but who have normal levels of very low density lipoprotein (VLDL). Inspection of plasma refrigerated for 14 hours is helpful in distinguishing Types I, IV and V hyperlipoproteinemia2.
The initial treatment for dyslipidemia is dietary therapy specific for the type of lipoprotein abnormality. Excess body weight and excess alcoholic intake may be important factors in hypertriglyceridemia and should be addressed prior to any drug therapy. Physical exercise can be an important ancillary measure. Diseases contributory to hyperlipidemia, such as hypothyroidism or diabetes mellitus should be looked for and adequately treated. Estrogen therapy, like thiazide diuretics and beta-blockers, is sometimes associated with massive rises in plasma triglycerides, especially in subjects with familial hypertriglyceridemia. In such cases, discontinuation of the specific etiologic agent may obviate the need for specific drug therapy of hypertriglyceridemia.
The use of drugs should be considered only when reasonable attempts have been made to obtain satisfactory results with non-drug methods. If the decision is made to use drugs, the patient should be instructed that this does not reduce the importance of adhering to diet (See WARNINGS and PRECAUTIONS).
| Type | Lipoprotein Elevated | Lipid Elevation | |
|---|---|---|---|
| Major | Minor | ||
| C = cholesterol TG = triglycerides LDL = low density lipoprotein VLDL = very low density lipoprotein IDL = intermediate density lipoprotein |
|||
| I (rare) | Chylomicrons | TG | ↑↔C |
| IIa | LDL | C | — |
| IIb | LDL, VLDL | C | TG |
| III (rare) | IDL | C, TG | — |
| IV | VLDL | TG | ↑↔C |
| V (rare) | Chylomicrons, VLDL | TG | ↑↔ |
| Definite Athlerosclerotic Disease* | Two or More Other Risk Factors† | LDL-Cholesterol mg/dL (mmol/L) | |
|---|---|---|---|
| Initiation Level | Goal | ||
| No | No | ≥ 190 (≥ 4.9) | < 160 (< 4.1) |
| No | Yes | ≥ 160 (≥ 4.1) | < 130 (< 3.4) |
| Yes | Yes or No | ≥ 130‡ (≥ 3.4) | < 100 (< 2.6) |
CONTRAINDICATIONS
Fenofibrate capsules are contraindicated in patients who exhibit hypersensitivity to fenofibrate.
Fenofibrate capsules are contraindicated in patients with hepatic or severe renal dysfunction, including primary biliary cirrhosis, and patients with unexplained persistent liver function abnormality.
Fenofibrate capsules are contraindicated in patients with preexisting gallbladder disease (see WARNINGS).
WARNINGS
Hepatotoxicity:
Serious drug-induced liver injury (DILI), including liver transplantation and death, have been reported postmarketing with fenofibrate. DILI has been reported within the first few weeks of treatment or after several months of therapy and in some cases has reversed with discontinuation of fenofibrate treatment. Patients with DILI have experienced signs and symptoms including dark urine, abnormal stool, jaundice, malaise, abdominal pain, myalgia, weight loss, pruritus, and nausea. Many patients had concurrent elevations of total bilirubin, serum alanine transaminase (ALT), and aspartate transaminase (AST). DILI has been characterized as hepatocellular, chronic active, and cholestatic hepatitis, and cirrhosis has occurred in association with chronic active hepatitis.
In clinical trials, fenofibrate at doses equivalent to 134 mg to 200 mg fenofibrate daily has been associated with increases in serum AST orALT.
The incidence of increases in transaminases may be dose-related.
Fenofibrate is contraindicated in patients with active liver disease, including those with primary biliary cirrhosis and unexplained persistent liver function abnormalities [see Contraindications (4)]. Monitor patient’s liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy with fenofibrate. Discontinue fenofibrate if signs or symptoms of liver injury develop or if elevated enzyme levels persist (ALT or AST > 3 times the upper limit of normal, or if accompanied by elevation of bilirubin). Do not restart fenofibrate in these patients if there is no alternative explanation for the liver injury.
Cholelithiasis:
Fenofibrate, like clofibrate and gemfibrozil, may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated. Fenofibrate therapy should be discontinued if gallstones are found.
Concomitant Oral Anticoagulants:
Caution should be exercised when anticoagulants are given in conjunction with fenofibrate because of the potentiation of coumarin-type anticoagulants in prolonging the prothrombin time/INR. The dosage of the anticoagulant should be reduced to maintain the prothrombin time/lNR at the desired level to prevent bleeding complications. Frequent prothrombin time/INR determinations are advisable until it has been definitely determined that the prothrombin time/INR has stabilized.
Concomitant HMG-CoA Reductase Inhibitors:
The combined use of fenofibrate and HMG-CoA reductase inhibitors should be avoided unless the benefit of further alterations in lipid levels is likely to outweigh the increased risk of this drug combination.
Concomitant administration of fenofibrate (equivalent to fenofibrate 200 mg) and pravastatin (40 mg) once daily for 10 days increased the mean Cmax and AUC values for pravastatin by 36% (range from 69% decrease to 321% increase) and 28% (range from 54% decrease to 128% increase), respectively, and for 3α-hydroxy-iso-pravastatin by 55% (range from 32% decrease to 314% increase) and 39% (range from 24% decrease to 261% increase), respectively. (See also CLINICAL PHARMACOLOGY, Drug-drug interactions.)
The combined use of fibric acid derivatives and HMG-CoA reductase inhibitors has been associated, in the absences of a marked pharmacokinetic interaction, in numerous case reports, with rhabdomyolysis, markedly elevated creatine kinase (CK) levels and myoglobinuria, leading in a high proportion of cases to acute renal failure.
The use of fibrates alone, including fenofibrate capsules may occasionally be associated with myositis, myopathy, or rhabdomyolysis. Patients receiving fenofibrate and complaining of muscle pain, tenderness, or weakness should have prompt medical evaluation for myopathy, including serum creatine kinase level determination. If myopathy/myositis is suspected or diagnosed, fenofibrate therapy should be stopped.
Mortality:
The effect of fenofibrate on coronary heart disease morbidity and mortality and non-cardiovascular mortality has not been established.
Other Considerations:
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study was a 5-year randomized, placebo-controlled study of 9,795 patients with type 2 diabetes mellitus treated with fenofibrate.
Fenofibrate demonstrated a non-significant 11% relative reduction in the primary outcome of coronary heart disease events (hazard ratio [HR] 0.89, 95% CI 0.75 to 1.05, p=0.16) and a significant 11% reduction in the secondary outcome of total cardiovascular disease events (HR 0.89 [0.8 to 0.99], p=0.04). There was a non-significant 11% (HR 1.11 [0.95, 1.29], p=0.18) and 19% (HR 1.19 [0.9, 1.57], p=0.22) increase in total and coronary heart disease mortality, respectively, with fenofibrate as compared to placebo.
In the Coronary Drug Project, a large study of post myocardial infarction of patients treated for 5 years with clofibrate, there was no difference in mortality seen between the clofibrate group and the placebo group. There was however, a difference in the rate of cholelithiasis and cholecystitis requiring surgery between the two groups (3% vs. 1.8%).
Because of chemical, pharmacological, and clinical similarities between fenofibrate micronized. Atromid-S (clofibrate), and Lopid (gemfibrozil), the adverse findings in 4 large randomized, placebo-controlled clinical studies with these other fibrate drugs may also apply to fenofibrate.
In a study conducted by the World Health Organization (WHO), 5,000 subjects without known coronary artery disease were treated with placebo or clofibrate for 5 years and followed for an additional one year. There was a statistically significant, higher age-adjusted all-cause mortality in the clofibrate group compared with the placebo group (5.7% vs. 3.96%, p=<0.01). Excess mortality was due to a 33% increase in non-cardiovascular causes, including malignancy, post-cholecystectomy complications, and pancreatitis. This appeared to confirm the higher risk of gallbladder disease seen in clofibrate-treated patients studied in the Coronary Drug Project.
The Helsinki Heart Study was a large (n=4,081) study of middle-aged men without a history of coronary artery disease. Subjects received either placebo or gemfibrozil for 5 years, with a 3.5-year open extension afterward. Total mortality was numerically higher in the gemfibrozil randomization group but did not achieve statistical significance (p=0.19, 95% confidence interval for relative risk G:P=0.91 to 1.64). Although cancer deaths trended higher in the gemfibrozil group (p=0.11), cancers (excluding basal cell carcinoma) were diagnosed with equal frequency in both study groups. Due to the limited size of the study, the relative risk of death from any cause was not shown to be different than that seen in the 9 year follow-up data from World Health Organization study (RR=1.29). Similarly, the numerical excess of gallbladder surgeries in the gemfibrozil group did not differ statistically from that observed in the WHO study.
A secondary prevention component of the Helsinki Heart Study enrolled middle-aged men excluded from the primary prevention study because of known or suspected coronary heart disease. Subjects received gemfibrozil or placebo for 5 years. Although cardiac deaths trended higher in the gemfibrozil group, this was not statistically significant (hazard ratio 2.2, 95% confidence interval: 0.94 to 5.05). The rate of gallbladder surgery was not statistically significant between study groups, but did trend higher in the gemfibrozil group,
(1.9% vs. 0.3%, p=0.07). There was a statistically significant difference in the number of appendectomies in the gemfibrozil group (6/311 vs. 0/317, p=0.029).
PRECAUTIONS
Initial Therapy:
Laboratory studies should be done to ascertain that the lipid levels are consistently abnormal before instituting fenofibrate therapy. Every attempt should be made to control serum lipids with appropriate diet, exercise, weight loss in obese patients, and control of any medical problems such as diabetes mellitus and hypothyroidism that are contributing to the lipid abnormalities. Medications known to exacerbate hypertriglyceridemia (beta-blockers, thiazides, estrogens) should be discontinued or changed if possible prior to consideration of triglyceride-lowering drug therapy.
Continued Therapy:
Periodic determination of serum lipids should be obtained during initial therapy in order to establish the lowest effective dose of fenofibrate capsules. Therapy should be withdrawn in patients who do not have an adequate response after two months of treatment with the maximum recommended dose of 200 mg per day.
Pancreatitis:
Pancreatitis has been reported in patients taking fenofibrate, gemfibrozil, and clofibrate. This occurrence may represent a failure of efficacy in patients with severe hypertriglyceridemia, a direct drug effect, or a secondary phenomenon mediated through biliary tract stone or sludge formation with obstruction of the common bile duct.
Hypersensitivity Reactions:
Acute Hypersensitivity:
Anaphylaxis and angioedema have been reported postmarketing with fenofibrate. In some cases, reactions were life-threatening and required emergency treatment. If a patient develops signs or symptoms of an acute hypersensitivity reaction, advise them to seek immediate medical attention and discontinue fenofibrate.
Delayed Hypersensitivity:
Severe cutaneous adverse drug reactions (SCAR), including Stevens-Johnson Syndrome, Toxic Epidermal Necrolysis, and Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), have been reported postmarketing, occurring days to weeks after initiation of fenofibrate. The cases of DRESS were associated with cutaneous reactions (such as rash or exfoliative dermatitis) and a combination of eosinophilia, fever, systemic organ involvement (renal, hepatic, or respiratory). Discontinue fenofibrate and treat patients appropriately if SCAR is suspected.
Hematologic Changes:
Mild to moderate hemoglobin, hematocrit, and white blood cell decreases have been observed in patients following initiation of fenofibrate therapy. However, these levels stabilize during long-term administration. Extremely rare spontaneous reports of thrombocytopenia and agranulocytosis have been received during post-marketing surveillance outside of the U.S. Periodic blood counts are recommended during the first 12 months of fenofibrate administration.
Myopathy and Rhabdomyolysis:
The use of fibrates alone, including fenofibrate, may occasionally be associated with myopathy. Treatment with drugs of the fibrate class has been associated on rare occasions with rhabdomyolysis, usually in patients with impaired renal function. Myopathy should be considered in any patient with diffuse myalgias, muscle tenderness or weakness, and/or marked elevations of creatine phosphokinase levels.
Patients should be advised to report promptly unexplained muscle pain, tenderness or weakness, particularly if accompanied by malaise or fever. CPK levels should be assessed in patients reporting these symptoms and fenofibrate therapy should be discontinued if markedly elevated CPK levels occur or myopathy is diagnosed.
Venothromboembolic Disease:
In the FIELD trial, pulmonary embolus (PE) and deep vein thrombosis (DVT) were observed at higher rates in the fenofibrate- than the placebo-treated group. Of 9,795 patients enrolled in FIELD, there were 4,900 in the placebo group and 4,895 in the fenofibrate group. For DVT, there were 48 events (1%) in the placebo group and 67 (1%) in the fenofibrate group (p = 0.074); and for PE, there were 32 (0.7%) events in the placebo group and 53 (1%) in the fenofibrate group (p = 0.022).
In the Coronary Drug Project, a higher proportion of the clofibrate group experienced definite or suspected fatal or nonfatal pulmonary embolism or thrombophlebitis than the placebo group (5.2% vs. 3.3% at 5 years;
p < 0.01).
Serum Creatinine:
Elevations in serum creatinine have been reported in patients on fenofibrate. These elevations tend to return to baseline following discontinuation of fenofibrate. The clinical significance of these observations is unknown.
Drug Interactions
Oral Anticoagulants:
CAUTION SHOULD BE EXERCISED WHEN COUMARIN ANTICOAGULANTS ARE GIVEN IN CONJUNCTION WITH FENOFIBRATE CAPSULES. THE DOSAGE OF THE ANTICOAGULANTS SHOULD BE REDUCED TO MAINTAIN THE PROTHROMBIN TIME/INR AT THE DESIRED LEVEL TO PREVENT BLEEDING COMPLICATIONS. FREQUENT PROTHROMBIN TIME/INR DETERMINATIONS ARE ADVISABLE UNTIL IT HAS BEEN DEFINITELY DETERMINED THAT THE PROTHROMBIN TIME/INR HAS STABILIZED.
HMG-CoA Reductase Inhibitors:
The combined use of fenofibrate and HMG-CoA reductase inhibitors should be avoided unless the benefit of further alterations in lipid levels is likely to outweigh the increased risk of this drug combination (see WARNINGS).
Resins:
Since bile acid sequestrants may bind other drugs given concurrently, patients should take fenofibrate capsules at least one hour before or 4 to 6 hours after a bile acid binding resin to avoid impeding its absorption.
Cyclosporine:
Because cyclosporine can produce nephrotoxicity with decreases in creatinine clearance and rises in serum creatinine, and because renal excretion is the primary elimination route of fibrate drugs including fenofibrate, there is a risk that an interaction will lead to deterioration. The benefits and risks of using fenofibrate with immunosuppressants and other potentially nephrotoxic agents should be carefully considered, and the lowest effective dose employed.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Two dietary carcinogenicity studies have been conducted in rats with fenofibrate. In the first 24-month study, rats were dosed with fenofibrate at 10, 45, and 200 mg/kg/day, approximately 0.3, 1, and 6 times the maximum recommended human dose (MRHD), based on body surface area comparisons (mg/m2). At a dose of 200 mg/kg/day (at 6 times the MRHD), the incidence of liver carcinomas was significantly increased in both sexes. A statistically significant increase in pancreatic carcinomas was observed in males at 1 and 6 times the MRHD; an increase in pancreatic adenomas and benign testicular interstitial cell tumors was observed at 6 times the MRHD in males. In a second 24-month rat carcinogenicity study in a different strain of rats, doses of 10 and 60 mg/kg/day (0.3 and 2 times the MRHD) produced significant increases in the incidence of pancreatic acinar adenomas in both sexes and increases in testicular interstitial cell tumors in males at 2 times the MRHD.
A 117-week carcinogenicity study was conducted in rats comparing three drugs:
fenofibrate 10 and 60 mg/kg/day (0.3 and 2 times the MRHD), clofibrate (400 mg/kg/day; 2 times the human dose), and gemfibrozil (250 mg/kg/day; 2 times the human dose, based on mg/m2surface area). Fenofibrate increased pancreatic acinar adenomas in both sexes. Clofibrate increased hepatocellular carcinoma and pancreatic acinar adenomas in males and hepatic neoplastic nodules in females. Gemfibrozil increased hepatic neoplastic nodules in males and females, while all three drugs increased testicular interstitial cell tumors in males.
In a 21-month study in mice, fenofibrate 10, 45, and 200 mg/kg/day (approximately 0.2, 1 and 3 times the MRHD on the basis of mg/m2 surface area) significantly increased the liver carcinomas in both sexes at 3 times the MRHD. In a second 18-month study at 10, 60, and 200 mg/kg/day, fenofibrate significantly increased the liver carcinomas in male mice and liver adenomas in female mice at 3 times the MRHD.
Electron microscopy studies have demonstrated peroxisomal proliferation following fenofibrate administration to the rat. An adequate study to test for peroxisome proliferation in humans has not been done, but changes in peroxisome morphology and numbers have been observed in humans after treatment with other members of the fibrate class when liver biopsies were compared before and after treatment in the same individual.
Fenofibrate has been demonstrated to be devoid of mutagenic potential in the following tests: Ames, mouse lymphoma, chromosomal aberration and unscheduled DNA synthesis in primary rat hepatocytes.
In fertility studies rats were given oral dietary doses of fenofibrate, males received 61 days prior to mating and females 15 days prior to mating through weaning which resulted in no adverse effect on fertility at doses up to 300 mg/kg/day (~ 10 times the MRHD, based on mg/m2 surface area comparisons).
Teratogenic Effects:
Safety in pregnant women has not been established. There are no adequate and well controlled studies of fenofibrate in pregnant women. Fenofibrate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In female rats given oral dietary doses of 15, 75, and 300 mg/kg/day of fenofibrate from 15 days prior to mating through weaning, maternal toxicity was observed at 0.3 times the MRHD, based on body surface area comparisons; mg/m2.
In pregnant rats given oral dietary doses of 14, 127, and 361 mg/kg/day from gestation day 6 to 15 during the period of organogenesis, adverse developmental findings were not observed at 14 mg/kg/day (less than 1 times the MRHD, based on body surface area comparisons; mg/m2). At higher multiples of human doses evidence of maternal toxicity was observed. In pregnant rabbits given oral gavage doses of 15, 150, and 300 mg/kg/day from gestation day 6 to 18 during the period of organogenesis and allowed to deliver, aborted litters were observed at 150 mg/kg/day (10 times the MRHD, based on body surface area comparisons: mg/m2). No developmental findings were observed at 15 mg/kg/day (at less than 1 times the MRHD, based on body surface area comparisons: mg/m2).
In pregnant rats given oral dietary doses of 15, 75, and 300 mg/kg/day from gestation day 15 through lactation day 21 (weaning), maternal toxicity was observed at less than 1 times the MRHD, based on body surface area comparisons: mg/m2.
Nursing Mothers:
It is not known whether fenofibrate is excreted into milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from fenofibrate, a decision should be made whether to discontinue nursing or administration of fenofibrate taking into account the importance of the drug to the lactating woman.
Geriatric Use:
Fenofibric acid is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function.
Fenofibric acid exposure is not influenced by age. However, elderly patients have a higher incidence of renal impairment, such that dose selection for the elderly should be made on the basis of renal function (see CLINICAL PHARMACOLOGY, Special Populations, Renal Insufficiency ). Elderly patients with normal renal function should require no dose modifications.
ADVERSE REACTIONS
Clinical Studies Experience:
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Adverse events reported by 2% or more of patients treated with fenofibrate (and greater than placebo) during the double-blind, placebo-controlled trials, regardless of causality, are listed in Table 3 below. Adverse events led to discontinuation of treatment in 5.0% of patients treated with fenofibrate and in 3.0% treated with placebo. Increases in liver function tests were the most frequent events, causing discontinuation of fenofibrate treatment in 1.6% of patients in double-blind trials.
Photosensitivity reactions have occurred days to months after initiation; in some of these cases, patients reported a prior photosensitivity reaction to ketoprofen.
Table 3. Adverse Reactions Reported by 2% or More of Patients Treated withFenofibrate and Greater than Placebo During the Double-Blind, Placebo-Controlled Trials
| BODY SYSTEM Adverse Reaction | Fenofibrate* (N=439) | PLACEBO (N=365) |
|---|---|---|
| BODY AS A WHOLE | ||
| Abdominal Pain | 4.6% | 4.4% |
| Back Pain | 3.4% | 2.5% |
| Headache | 3.2% | 2.7% |
| DIGESTIVE | ||
| Abnormal Liver Function Tests | 7.5%† | 1.4% |
| Nausea | 2.3% | 1.9% |
| Constipation | 2.1% | 1.4% |
| METABOLIC AND NUTRITIONAL DISORDERS | ||
| Increased ALT | 3% | 1.6% |
| Increased CPK | 3% | 1.4% |
| Increased AST | 3.4%† | 0.5% |
| RESPIRATORY | ||
| Respiratory Disorder | 6.2% | 5.5% |
| Rhinitis | 2.3% | 1.1% |
* Dosage equivalent to 145 mg fenofibrate.
† Significantly different from Placebo.
Increases in Liver Enzymes:
In a pooled analysis of 10 placebo-controlled trials, increases to >3 times the upper limit of normal in ALT occurred in 5.3% of patients taking fenofibrate versus 1.1% of patients treated with placebo. In an 8-week study, the incidence of ALT or AST elevations ≥ 3 times the upper limit of normal was 13% in patients receiving dosages equivalent to 134 mg to 200 mg fenofibrate daily and was 0% in those receiving dosages equivalent to 34 mg to 67 mg fenofibrate daily or placebo.
Post-Marketing Experience:
The following adverse reactions have been identified during post-approval use of fenofibrate: myalgia, rhabdomyolysis, pancreatitis, acute renal failure, muscle spasm, hepatitis, cirrhosis, increased total bilirubin, anemia, arthralgia, decreases in hemoglobin, decreases in hematocrit, white blood cell decreases, asthenia and interstitial lung disease. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
To report SUSPECTED ADVERSE REACTIONS, contact AustarPharma, LLC at 1-844-375-5410 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
OVERDOSAGE
There is no specific treatment for overdose with fenofibrate. General supportive care of the patient is indicated, including monitoring of vital signs and observation of clinical status, should an overdose occur. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage; usual precautions should be observed to maintain the airway. Because fenofibrate is highly bound to plasma proteins, hemodialysis should not be considered.
DOSAGE AND ADMINISTRATION
Patients should be placed on an appropriate lipid-lowering diet before receiving fenofibrate capsules, and should continue this diet during treatment with fenofibrate capsules. Fenofibrate capsules should be given with meals, thereby optimizing the bioavailability of the medication.
For the treatment of adult patients with primary hypercholesterolemia or mixed hyperlipidemia, the initial dose of fenofibrate capsules is 200 mg per day.
For adult patients with hypertriglyceridemia, the initial dose is 67 mg to 200 mg per day. Dosage should be individualized according to patient response, and should be adjusted if necessary following repeat lipid determinations at 4 to 8 week intervals. The maximum dose is 200 mg per day.
Treatment with fenofibrate capsules, should be initiated at a dose of 67 mg/day in patients having impaired renal function, and increased only after evaluation of the effects on renal function and lipid levels at this dose. In the elderly, the initial dose should likewise be limited to 67 mg/day.
Lipid levels should be monitored periodically and consideration should be given to reducing the dosage of fenofibrate capsules if lipid levels fall significantly below the targeted range.
HOW SUPPLIED
Fenofibrate capsules, USP (micronized) 67 mg are opaque white cap and body, hard gelatin capsules, printed in black ink “CL” and “22” on opposing cap and body portions of the capsule. They are supplied as follows:
NDC 35561-345-11 Bottles of 90 capsules
NDC 35561-345-12 Bottles of 100 capsules
NDC 35561-345-13 Bottles of 500 capsules
Fenofibrate capsules, USP (micronized) 134 mg are opaque white cap and opaque yellow body, hard gelatin capsules, printed in black ink “CL” and “23” on opposing cap and body portions of the capsule. They are supplied as follows:
NDC 35561-346-11 Bottles of 90 capsules
NDC 35561-346-12 Bottles of 100 capsules
NDC 35561-346-13 Bottles of 500 capsules
Fenofibrate capsules, USP (micronized) 200 mg are opaque yellow cap and body, hard gelatin capsules, printed in black ink “CL” and “24” on opposing cap and body portions of the capsule. They are supplied as follows:
NDC 35561-347-11 Bottles of 90 capsules
NDC 35561-347-12 Bottles of 100 capsules
NDC 35561-347-13 Bottles of 500 capsules
REFERENCES
- GOLDBERG AC, et al. Fenofibrate for the Treatment of Type IV and V Hyperlipoproteinemias: A Double-Blind, Placebo-Controlled Multicenter US Study. Clinical Therapeutics, 11, pp. 69-83, 1989.
- NIKKILA EA. Familial Lipoprotein Lipase Deficiency and Related Disorders of Chylomicron Metabolism. In Stanbury J.B., et al. (eds.): The Metabolic Basis of Inherited Disease, 5th edition, McGraw-Hill, 1983, Chap. 30, pp. 622-642.
- BROWN WV, et al. Effects of Fenofibrate on Plasma Lipids: Double-Blind, Multicenter Study In Patients with Type IIA or IIB Hyperlipidemia. Arteriosclerosis. 6, pp. 670-678, 1986.
All brands mentioned are trademarks of their respective owners and are not trademarks of AustarPharma, LLC.
Manufactured by:
AustarPharma, LLC
Edison, NJ 08837 USA
Revised: 05/2021
LBL342
PRINCIPAL DISPLAY PANEL - 67 mg Capsule Bottle Label
NDC 35561-345-11
Fenofibrate Capsules, USP
67 mg
Rx Only 90 Capsules

PRINCIPAL DISPLAY PANEL - 67 mg Capsule Bottle Label
NDC 35561-345-12
Fenofibrate Capsules, USP
67 mg
Rx Only 100 Capsules

PRINCIPAL DISPLAY PANEL - 67 mg Capsule Bottle Label
NDC 35561-345-13
Fenofibrate Capsules, USP
67 mg
Rx Only 100 Capsules

PRINCIPAL DISPLAY PANEL - 134 mg Capsule Bottle Label
NDC 35561-346-11
Fenofibrate Capsules, USP
134 mg
Rx Only 90 Capsules

PRINCIPAL DISPLAY PANEL - 134 mg Capsule Bottle Label
NDC 35561-346-12
Fenofibrate Capsules, USP
134 mg
Rx Only 100 Capsules

PRINCIPAL DISPLAY PANEL - 134 mg Capsule Bottle Label
NDC 35561-346-13
Fenofibrate Capsules, USP
134 mg
Rx Only 500 Capsules

PRINCIPAL DISPLAY PANEL - 200 mg Capsule Bottle Label
NDC 35561-347-11
Fenofibrate Capsules, USP
200 mg
Rx Only 90 Capsules


