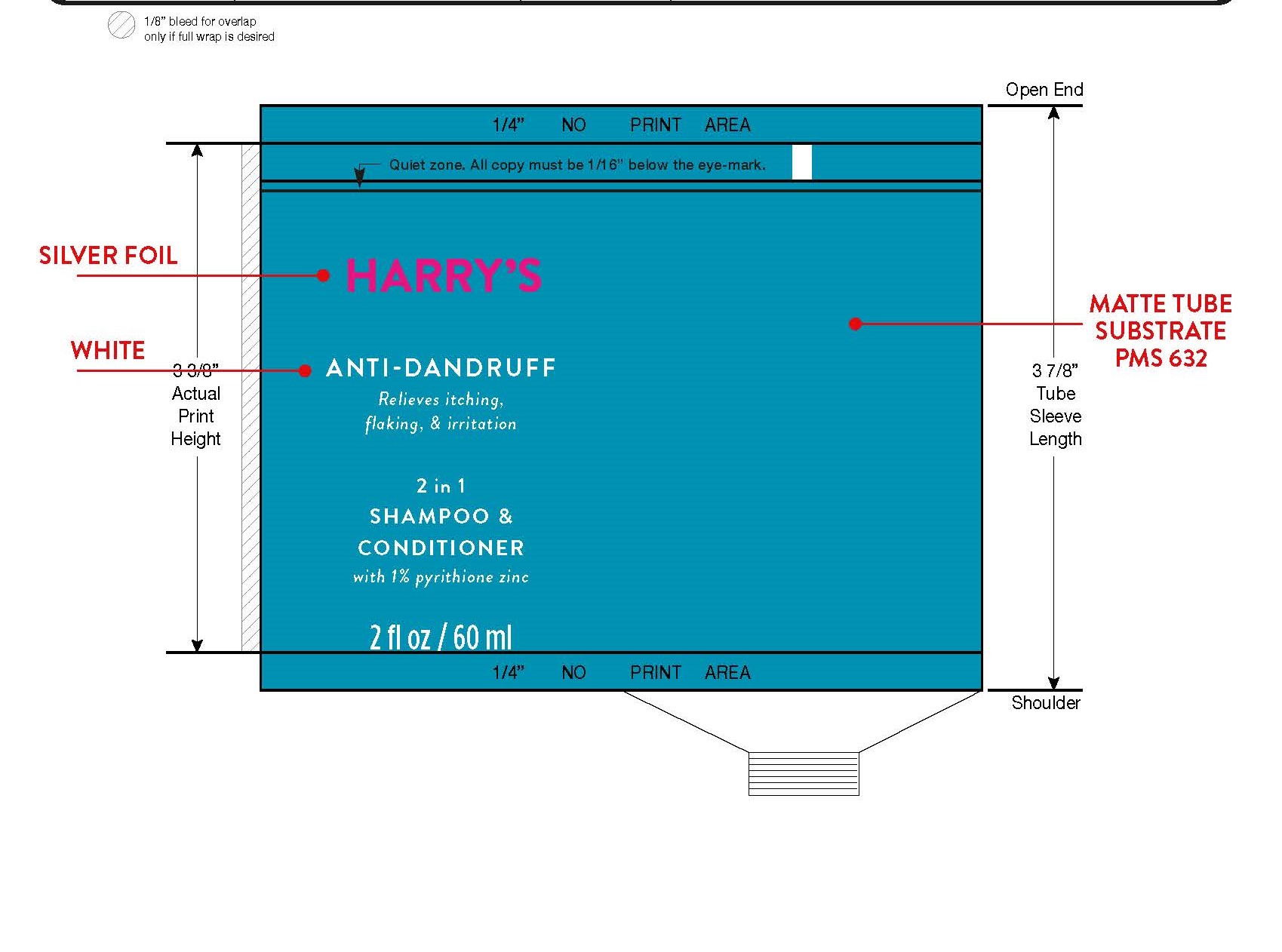
Warnings
For external use only.
When using this product avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water
Directions
• for best results use at least twice a week or as
directed by a doctor.
• shake before use.
• wet hair, massage onto scalp, rinse, repeat if desired.
Inactive ingredients
Water/Aqua/Eau, Aloe Barbadensis Leaf Juice, Cocamidopropyl Betaine, Lauric Acid, Disodium Cocoamphodiacetate, Decyl Glucoside, Polysorbate 20, Sodium Chloride, Citric Acid, Acrylates Copolymer, Glycerin, Phenoxyethanol, Polyquaternium-6, Coconut Acid, Mentha Piperita (Peppermint) Oil, PEG-120 Methyl Glucose Dioleate, Polyquaternium-67, Coco-Glucoside, Glyceryl Oleate, Eucalyptus Globulus Leaf Oil, Guar Hydroxypropyltrimonium Chloride, Menthol, Butylene Glycol, Lavandula Hybrida Oil, Carica Papaya (Papaya) Fruit Extract, Chlorphenesin, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Raphanus Sativus (Radish)
Root Extract, Potassium Hydroxide , Sodium Lauroyl Lactylate, Lavandula Angustifolia (Lavender) Oil, Sodium Caproyl Lactylate,
Propylene Glycol, Ferula Galbaniflua (Galbanum) Resin Oil, Sodium Polynaphthalenesulfonate, Sodium Acetate, Piper Nigrum (Pepper) Fruit Extract, Xanthan Gum, Isopropyl Alcohol, Benzoic Acid, Dehydroacetic Acid, Inga Alba Bark Extract, Tocopherol, Hydrogenated Palm Glycerides, Lecithin, Ascorbyl Palmitate, Linalool, Limonene.