DESCRIPTION
Acetaminophen and codeine is supplied in tablet form for oral administration.
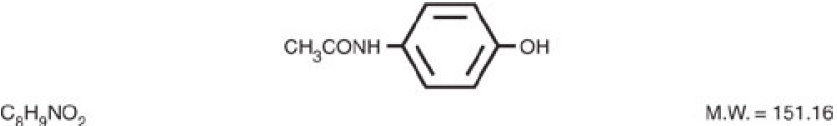
Acetaminophen, 4'-hydroxyacetanilide, a slightly bitter, white, odorless, crystalline powder, is a non-opiate, non-salicylate analgesic and antipyretic. It has the following structural formula:
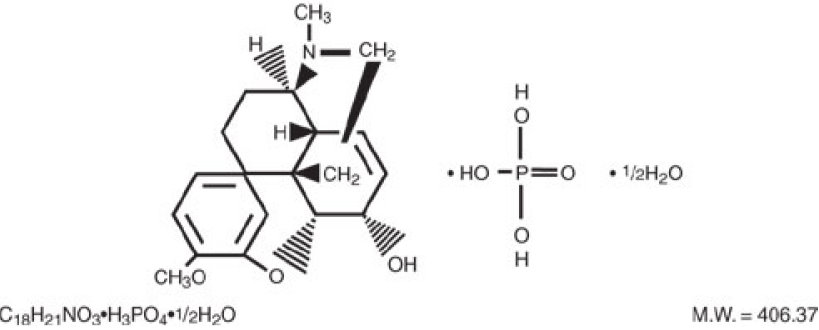
Codeine phosphate, 7,8-didehydro-4,5a-epoxy-3-methoxy-17methylmorphinan-6a-ol phosphate (1:1) (salt) hemihydrate, a white crystalline powder, is a narcotic analgesic and antitussive. It has the following structural formula:
Each 300 mg/30 mg Acetaminophen and Codeine Phosphate Tablet contains:
Acetaminophen ......................................................................................................300 mg
Codeine Phosphate ..................................................................................................30 mg
Each 300 mg/60 mg Acetaminophen and Codeine Phosphate Tablet contains:
Acetaminophen ......................................................................................................300 mg
Codeine Phosphate ..................................................................................................60 mg
In addition each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, povidone, pregelatinized corn starch, sodium metabisulfite, sodium starch glycolate and stearic acid.
CLINICAL PHARMACOLOGY
This product combines the analgesic effects of a centrally acting analgesic, codeine, with a peripherally acting analgesic, acetaminophen.
The behavior on the individual components is described below.
Codeine:
Codeine is readily absorbed from the gastrointestinal tract. It is rapidly distributed from the intravascular spaces to the various body
tissues, with preferential uptake by parenchymatous organs such as the liver, spleen and kidney. Codeine crosses the blood-brain
barrier, and is found in fetal tissue and breast milk. The plasma concentration does not correlate with brain concentration or relief of
pain; however, codeine is not bound to plasma proteins and does not accumulate in body tissues.
The plasma half-life is about 2.9 hours. The elimination of codeine is primarily via the kidneys, and about 90% of an oral dose is
excreted by the kidneys within 24 hours of dosing. The urinary secretion products consist of free and glucuronide conjugated codeine
(about 70%), free and conjugated norcodeine (about 10%), free and conjugated morphine (about 10%), normorphine (4%), and
hydrocodone (1%). The remainder of the dose is excreted in the feces.
At therapeutic doses, the analgesic effect reaches a peak within 2 hours and persists between 4 and 6 hours.
See OVERDOSAGE for toxicity information.
Acetaminophen:
Acetaminophen is rapidly absorbed from the gastrointestinal tract and is distributed throughout most body tissues. The plasma half-life
is 1.25 to 3 hours, but may be increased by liver damage and following overdosage. Elimination of acetaminophen is principally by
liver metabolism (conjugation) and subsequent renal excretion of metabolites. Approximately 85% of an oral dose appears in the urine
within 24 hours of administration, most as the glucuronide conjugate, with small amounts of other conjugates and unchanged drug.
See OVERDOSAGE for toxicity information.
INDICATIONS AND USAGE
Acetaminophen and codeine phosphate tablets are indicated for the relief of mild to moderately severe pain.
CONTRAINDICATIONS
This product should not be administered to patients who have previously exhibited hypersensitivity to codeine or acetaminophen.
WARNINGS
In the presence of head injury or other intracranial lesions, the respiratory depressant effects of codeine and other narcotics may be markedly enhanced, as well as their capacity for elevating cerebrospinal fluid pressure. Narcotics also produce other CNS depressant effects such as drowsiness, that may further obscure the clinical course of the patients with head injuries.
Codeine or other narcotics may obscure signs on which to judge the diagnosis or clinical course of patients with acute abdominal conditions.
Codeine is habit-forming and potentially abusable. Consequently, the extended use of this product is not recommended.
Acetaminophen and codeine phosphate tablets contain sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
PRECAUTIONS
General:
Acetaminophen and codeine phosphate tablets should be prescribed with caution in certain special-risk patients, such as the elderly or debilitated, and those with severe impairment of renal or hepatic function, head injuries, elevated intracranial pressure, acuted abdominal conditions, hypothyroidism, urethral stricture, Addison's disease, or prostatic hypertrophy.
Ultra-Rapid Metabolizers of Codeine
Some individuals may be ultra-rapid metabolizers due to a specific CYP2D6*2x2 genotype. These individuals convert codeine into its active metabolite, morphine, more rapidly and completely than other people. This rapid conversion results in higher-than-expected serum morphine levels. Even at labeled dosage regimens, individuals who are ultra-rapid metabolizers may experience overdose symptoms such as extreme sleepiness, confusion, or shallow breathing.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1–10% in Caucasians, 3% in African Americans, and 16–28% in North Africans, Ethiopians and Arabs. Data is not available for other ethnic groups.
When physicians prescribe codeine-containing drugs, they should choose the lowest effective dose for the shortest period of time and should inform their patients about these risks and the signs of morphine overdose (see PRECAUTIONS - Nursing Mothers).
Information for Patients:
Codeine may impair mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery. Such tasks should be avoided while taking this product.
Alcohol and other CNS depressants may produce an additive CNS depression, when taken with this combination product, and should be avoided.
Codeine may be habit-forming. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.
Caution patients that some people have a variation in a liver enzyme and change codeine into morphine more rapidly and completely than other people. These people are ultra-rapid metabolizers and are more likely to have higher-than-normal levels of morphine in their blood after taking codeine which can result in overdose symptoms such as extreme sleepiness, confusion, or shallow breathing. In most cases, it is unknown if someone is an ultra-rapid codeine metabolizer.
Nursing mothers taking codeine can also have higher morphine levels in their breast milk if they are ultra-rapid metabolizers. These higher levels of morphine in breast milk may lead to life-threatening or fatal side effects in nursing babies. Instruct nursing mothers to watch for signs of morphine toxicity in their infants including increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties, or limpness. Instruct nursing mothers to talk to the baby's doctor immediately if they notice these signs and, if they cannot reach the doctor right away, to take the baby to an emergency room or call 911 (or local emergency services).
Laboratory Tests:
In patients with severe hepatic or renal disease, effects of therapy should be monitored with serial liver and/or renal function tests.
Drug Interactions:
This drug may enhance the effects of: other narcotic analgesics, alcohol, general anesthetics, tranquilizers such as chlordiazepoxide, sedative-hypnotics, or other CNS depressants, causing increased CNS depression.
Drug/Laboratory Test Interactions:
Codeine may increase serum amylase levels.
Acetaminophen may produce false-positive test results for urinary 5-hydroxyindoleacetic acid.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
No adequate studies have been conducted in animals to determine whether acetaminophen and codeine have a potential for carcinogenesis or mutagenesis. No adequate studies have been conducted in animals to determine whether acetaminophen has a potential for impairment of fertility.
Acetaminophen and codeine have been found to have no mutagenic potential using the Ames Salmonella-Microsomal Activation test, the Basc test on Drosophila germ cells, and Micronucleus test on mouse bone marrow.
Pregnancy:
Teratogenic Effects: Pregnancy Category C:
Codeine:
A study in rats and rabbits reported no teratogenic effect of codeine administered during the period of organogeneses in doses ranging from 5 to 120 mg/kg. In the rat, doses at the 120 mg/kg level, in the toxic range for the adult animal, were associated with an increase in embryo resorption at the time of implantation. In another study a single 100 mg/kg dose of codeine administered to pregnant mice reportedly resulted in delayed ossification in the offspring.
There are no adequate and well-controlled studies in pregnant women. Acetaminophen and codeine phosphate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects:
Dependence has been reported in newborns whose mothers took opiates regularly during pregnancy. Withdrawal signs include irritability, excessive crying, tremors, hyperreflexia, fever, vomiting, and diarrhea. These signs usually appear during the first few days
of life.
Labor and Delivery:
Narcotic analgesics cross the placental barrier. The closer to delivery and the larger the dose used, the greater the possibility of respiratory depression in the newborn. Narcotic analgesics should be avoided during labor if delivery of a premature infant is anticipated. If the mother has received narcotic analgesics during labor, newborn infants should be observed closely for signs of respiratory depression. Resuscitation may be required (see OVERDOSAGE). The effect of codeine, if any, on the later growth, development, and functional maturation of the child is unknown.
Nursing Mothers:
Acetaminophen is excreted in breast milk in small amounts, but the significance of its effects on nursing infants is not known. Because of the potential for serious adverse reactions in nursing infants from acetaminophen, a decision should be made whether to discontinue the drug, taking into account the importance of the drug to the mother.
Codeine is secreted into human milk. In women with normal codeine metabolism (normal CYP2D6 activity), the amount of codeine secreted into human milk is low and dose-dependent. Despite the common use of codeine products to manage postpartum pain, reports of adverse events in infants are rare. However, some women are ultra-rapid metabolizers of codeine. These women achieve higher-than-expected serum levels of codeine's active metabolite, morphine, leading to higher-than-expected levels of morphine in breast milk and potentially dangerously high serum morphine levels in their breastfed infants. Therefore, maternal use of codeine can potentially lead to serious adverse reactions, including death, in nursing infants.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1–10% in Caucasians, 3% in African Americans, and 16–28% in North Africans, Ethiopians and Arabs. Data is not available for other ethnic groups.
The risk of infant exposure to codeine and morphine through breast milk should be weighed against the benefits of breastfeeding for both the mother and baby. Caution should be exercised when codeine is administered to a nursing woman. If a codeine-containing product is selected, the lowest dose should be prescribed for the shortest period of time to achieve the desired clinical effect. Mothers using codeine should be informed about when to seek immediate medical care and how to identify the signs and symptoms of neonatal toxicity, such as drowsiness or sedation, difficulty breastfeeding, breathing difficulties, and decreased tone, in their baby. Nursing mothers who are ultra-rapid metabolizers may also experience overdose symptoms such as extreme sleepiness, confusion, or shallow breathing. Prescribers should closely monitor mother-infant pairs and notify treating pediatricians about the use of codeine during breastfeeding (see PRECAUTIONS - General - Ultra-Rapid Metabolizers of Codeine).
ADVERSE REACTIONS
The most frequently reported adverse reactions are drowsiness, lightheadedness, dizziness, sedation, shortness of breath, nausea and vomiting. These effects seem to be more prominent in ambulatory than in non-ambulatory patients, and some of these adverse reactions may be alleviated if the patient lies down.
Other adverse reactions include allergic reactions, euphoria, dysphoria, constipation, abdominal pain, pruritus, rash, thrombocytopenia, agranulocytosis.
At higher doses codeine has most of the disadvantages of morphine including respiratory depression.
DRUG ABUSE AND DEPENDENCE
Controlled Substance:
Acetaminophen and Codeine Phosphate tablets are classified as a Schedule III controlled substance.
Abuse and Dependence:
Codeine can produce drug dependence of the morphine type and, therefore, has the potential for being abused. Psychological dependence, physical dependence, and tolerance may develop upon repeated administration, and it should be prescribed and administered with the same degree of caution appropriate to the use of other oral narcotic medications.
OVERDOSAGE
Following an acute overdosage, toxicity may result from codeine or acetaminophen. Signs and Symptoms:
Codeine:
Toxicity from codeine poisoning includes the opioid triad of: pinpoint pupils, depression of respiration, and loss of consciousness. Convulsions may occur.
Acetaminophen:
In acetaminophen overdosage: dose-dependent, potentially fatal hepatic necrosis is the most serious adverse effect. Renal tubular necrosis, hypoglycemic coma and thrombocytopenia may also occur.
Early symptoms following a potentially hepatotoxic overdose may include: nausea, vomiting, diaphoresis and general malaise. Clinical and laboratory evidence of hepatic toxicity may not be apparent until 48 to 72 hours post-ingestion.
In adults hepatic toxicity has rarely been reported with acute overdoses of less than 10 grams, or fatalities with less than 15 grams.
Treatment:
A single or multiple overdose with acetaminophen and codeine is a potentially lethal polydrug overdose, and consultation with a regional poison control center is recommended.
Immediate treatment includes support of cardiorespiratory function and measures to reduce drug absorption. Vomiting should be induced mechanically, or with syrup of ipecac, if the patient is alert (adequate pharyngeal and laryngeal reflexes). Oral activated charcoal (1 g/kg) should follow gastric emptying. The first dose should be accompanied by an appropriate cathartic. If repeated doses are used, the cathartic might be included with alternate doses as required. Hypotension is usually hypovolemic and should respond to fluids. Vasopressors and other supportive measures should be employed as indicated. A cuffed endo-tracheal tube should be inserted before gastric lavage of the unconscious patient and, when necessary, to provide assisted respiration.
Meticulous attention should be given to maintaining adequate pulmonary ventilation. In severe cases of intoxication, peritoneal dialysis or preferably hemodialysis may be considered. If hypoprothrombinemia occurs due to acetaminophen overdose, vitamin K should be administered intravenously.
Naloxone, a narcotic antagonist, can reverse respiratory depression and coma associated with opioid overdose. Naloxone hydrochloride 0.4 mg to 2 mg is given parenterally. Since the duration of action of codeine may exceed that of the naloxone, the patient should be kept under continuous surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. A narcotic antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
If the dose of acetaminophen may have exceeded 140 mg/kg, acetylcysteine should be administered as early as possible. Serum acetaminophen levels should be obtained, since levels four or more hours following ingestion help predict acetaminophen toxicity. Do not await acetaminophen assay results before initiating treatment. Hepatic enzymes should be obtained initially, and repeated at 24-hour intervals.
Methemoglobinemia over 30% should be treated with methylene blue by slow intravenous administration.
Toxic Doses (for adults):
Acetaminophen: toxic dose 10 g
Codeine: toxic dose 240 mg
DOSAGE AND ADMINISTRATION
Dosage should be adjusted according to severity of pain and response of the patient. The usual adult dosage is:
Single Doses (range) Maximum 24 Hour Dose
Codeine Phosphate 15 mg to 60 mg 360 mg
Acetaminophen 300 mg to 1000 mg 4000 mg
The usual dose of codeine phosphate in children is 0.5 mg/kg.
Doses may be repeated up to every 4 hours.
The prescriber must determine the number of tablets per dose, and the maximum number of tablets per 24 hours based upon the above dosage guidance. This information should be conveyed in the prescription.
It should be kept in mind, however, that tolerance to codeine can develop with continued use and that the incidence of untoward effects is dose related. Adult doses of codeine higher than 60 mg fail to give commensurate relief of pain but merely prolong analgesia and are associated with an appreciably increased incidence of undesirable side effects. Equivalently high doses in children would haveDOSAGE AND ADMINISTRATION Dosage should be adjusted according to severity of pain and response of the patient. The usual adult dosage is: Single Doses (range) Maximum 24 Hour Dose Codeine Phosphate 15 mg to 60 mg 360 mg Acetaminophen 300 mg to 1000 mg 4000 mg The usual dose of codeine phosphate in children is 0.5 mg/kg. Doses may be repeated up to every 4 hours. The prescriber must determine the number of tablets per dose, and the maximum number of tablets per 24 hours based upon the above dosage guidance. This information should be conveyed in the prescription. It should be kept in mind, however, that tolerance to codeine can develop with continued use and that the incidence of untoward effects is dose related. Adult doses of codeine higher than 60 mg fail to give commensurate relief of pain but merely prolong analgesia and are associated with an appreciably increased incidence of undesirable side effects. Equivalently high doses in children would have similar effects.
HOW SUPPLIED
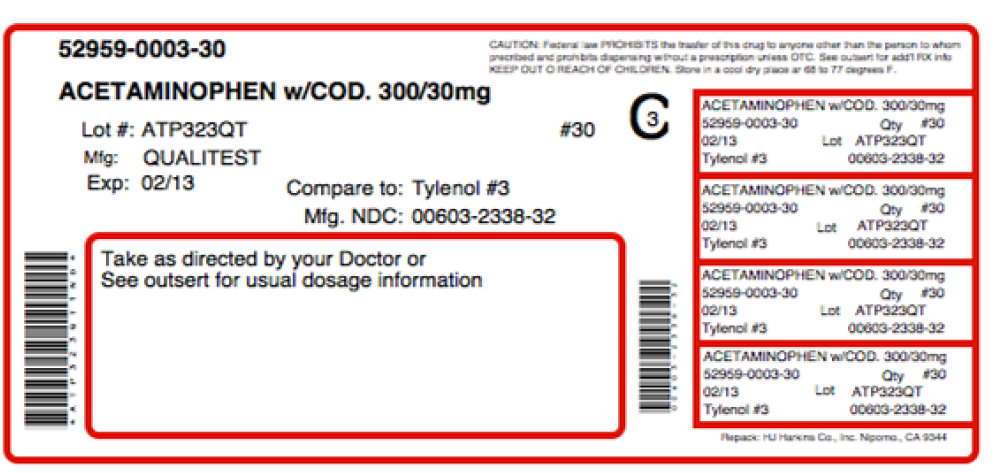
Acetaminophen and Codeine Phosphate Tablets 300 mg/30 mg are white, round, flat-faced, beveled edge, scored (bisect bar) tablets, debossed "2064" and "V" on one side and debossed "3" on the reverse side. They are supplied in bottles of 30, 50, 60, 90, 100, 120, 180, 500 and 1000.
Acetaminophen and Codeine Phosphate Tablets 300 mg/60 mg are white, round, flat-faced, beveled edge, scored (bisect bar) tablets, debossed "2065" and "V" on one side and debossed "4" on the reverse side. They are supplied in bottles of 100, 500 and 1000.
Dispense in a tight, light resistant container as defined in the USP/NF.
Store at 20 degree to 25 degree C (68 degree to 77 degree F) [see USP Controlled Room Temperature].
Manufactured for:
QUALITEST PHARMACEUTICALS
Huntsville, AL 35811
8182436
R11/07-R2
EACH TABLET CONTAINS:
Acetaminophen .................................... 300 mg
Codeine Phosphate ............................. 30 mg
* WARNING: MAY BE HABIT FORMING. USUAL DOSAGE: See package insert for complete dosage recommendations.
DISPENSE in a tight, light-resistant container as defined in the USP/NF.
STORE at 20 degree to 25 degree C (68 degree to 77 degree F) [see USP Controlled Room Temperature].
NDC 0603-2338-21
ACETAMINOPHEN and CODEINE * PHOSPHATE TABLETS, USP
CIII
300 mg/30 mg
*WARNING: MAY BE HABIT FORMING.
Rx only 100 TABLETS
Qualitest
Manufactured for:
QUALITEST PHARMACEUTICALS
Huntsville, AL 35811
Rev. 11/07 R2
8081508 2064
non varn
Theramine™ PRODUCT INFORMATION
Theramine (U.S. patent pending) capsules by oral administration. A specially formulated Medical Food product, consisting of a proprietary blend of amino acids and polyphenol ingredients in specific proportions, for the dietary management of the metabolic processes associated with pain disorders and inflammatory conditions. (PD) (IC). Must be administered under physician supervision.
Medical Foods
Medical Food products are often used in hospitals (e.g., for burn victims or kidney dialysis patients) and outside of a hospital setting under a physician’s care for the dietary management of diseases in patients with particular medical or metabolic needs due to their disease or condition. Congress defined "Medical Food" in the Orphan Drug Act and Amendments of 1988 as "a food which is formulated to be consumed or administered enterally [or orally] under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation." Medical Foods are complex formulated products, requiring sophisticated and exacting technology. Theramine has been developed, manufactured, and labeled in accordance with both the statutory and the FDA regulatory definition of a Medical Food. Theramine must be used while the patient is under the ongoing care of a physician.
PAIN DISORDERS (PD) INFLAMMATORY CONDITIONS (IC)
PD and IC as a Metabolic Deficiency Disease
A critical component of the definition of a Medical Food is the requirement for a distinctive nutritional deficiency. FDA scientists have proposed a physiologic definition of a distinctive nutritional deficiency as follows: “the dietary management of patients with specific diseases requires, in some instances, the ability to meet nutritional requirements that differ substantially from the needs of healthy persons. For example, in establishing the recommended dietary allowances for general, healthy population, the Food and Nutrition Board of the Institute of Medicine National Academy of Sciences, recognized that different or distinctive physiologic requirements may exist for certain persons with "special nutritional needs arising from metabolic disorders, chronic diseases, injuries, premature birth, other medical conditions and drug therapies. Thus, the distinctive nutritional needs associated with a disease reflects the total amount needed by a healthy person to support life or maintain homeostasis, adjusted for the distinctive changes in the nutritional needs of the patient as a result of the effects of the disease process on absorption, metabolism and excretion.” It was also proposed that in patients with certain disease states who respond to nutritional therapies, a physiologic deficiency of the nutrient is assumed to exist. For example, if a patient with pain disorders responds to a tryptophan formulation by decreasing perceived pain, a deficiency of tryptophan is assumed to exist. Patients with pain disorders and inflammatory conditions are known to have nutritional deficiencies of tryptophan, choline, arginine, GABA, flavonoids, and certain antioxidants. Patients with pain disorders and inflammatory conditions frequently exhibit reduced plasma levels of tryptophan and GABA and have been shown to respond to oral administration of GABA, arginine, tryptophan, or a 5-hydoxytryptophan formulation. Research has shown that tryptophan, arginine or GABA reduced diets result in a fall of circulating tryptophan, arginine, and/or GABA.
Patients with pain disorders frequently exhibit activation of the degradation pathways that increases the turnover of GABA, arginine and/or tryptophan leading to a reduced level of production of serotonin, GABA or nitric oxide for a given precursor blood level. Research has also shown that a genetic predisposition to accelerated degradation can lead to increased precursor requirements in certain patients with pain disorders and inflammatory conditions.
Choline is required to fully potentiate acetylcholine synthesis by brain neurons. A deficiency of choline leads to reduced acetylcholine production by the neurons. Flavonoids potentiate the production of acetylcholine by the neurons thereby reducing delta pain. Diets deficient in flavonoid rich foods and choline result in inadequate flavonoid concentrations, impeding acetylcholine production in certain patients with pain disorders and/or inflammatory conditions. Acetylcholine in pre-synaptic ganglia is necessary for the production of serotonin and nitric oxide in post-synaptic ganglia. Provision of tryptophan, arginine, GABA, choline and flavonoids with antioxidants, in specific proportions can restore the production of beneficial serotonin, nitric oxide, and acetylcholine, thereby reducing the perception of pain and reducing inflammation. L-Histidine is known to produce brain histamine that stimulates production of ACTH
Primary Ingredients Theramine consists of a proprietary blend of amino acids, cocoa, caffeine, cinnamon, and flavonoids in specific proportions. These ingredients fall into the category of Generally Regarded as Safe” (GRAS) as defined by the Food and Drug Administration (FDA) (Sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act). A GRAS substance is distinguished from a food additive on the basis of the common knowledge about the safety of the substance for its intended use. The standard for an ingredient to achieve GRAS status requires not only technical demonstration of non-toxicity and safety, but also general recognition of safety through widespread usage and agreement of that safety by experts in the field. Many ingredients have been determined by the U.S. Food and Drug Administration (FDA) to be GRAS, and are listed as such by regulation, in Volume 21 Code of Federal Regulations (CFR) Sections 182, 184, and 186.
Amino Acids
Amino Acids are the building blocks of protein. All amino acids are GRAS listed as they have been ingested by humans for thousands of years. The doses of the amino acids in Theramine are equivalent to those found in the usual human diet. Patients with pain disorders may require an increased amount of certain amino acids that cannot be obtained from normal diet alone. Tryptophan, for example, is an obligatory amino acid. The body cannot make tryptophan and must obtain tryptophan from the diet. Tryptophan is needed to produce serotonin. Serotonin is required to reduce pain. Patients with pain disorders and inflammatory conditions have altered serotonin metabolism. Some patients with pain disorders and inflammatory conditions have a resistance to the use of tryptophan that is similar to the mechanism found in insulin resistance. Patients with pain disorders and inflammatory conditions cannot acquire sufficient tryptophan from the diet to alter the perception of pain and the inflammatory process without ingesting a prohibitively large amount of calories, particularly calories from protein.
Flavonoids
Flavonoids are a group of phytochemical compounds found in all vascular plants including fruits and vegetables. They are a part of a larger class of compounds known as polyphenols. Many of the therapeutic or health benefits of colored fruits and vegetables, cocoa, red wine, and green tea are directly related to their flavonoid content. The specially formulated flavonoids found in Theramine cannot be obtained from conventional foods in the necessary proportions to elicit a therapeutic response.
Other Ingredients
Theramine contains the following inactive or other ingredients, as fillers, excipients, and colorings: magnesium stearate, microcrystalline cellulose, Maltodextrin NF, gelatin (as the capsule material).
Physical Description
Theramine is a yellow to light brown powder. Theramine contains L-Glutamine, L-Arginine, L-Histidine, and L-Serine, 5-Hydroxytryptophan as Griffonia Seed Extract, GABA, Choline Bitartrate, Cinnamon, Cocoa, Hydrolyzed Whey Protein, and Grape Seed Extract.
CLINICAL PHARMACOLOGY
Mechanism of Action
Theramine acts by restoring and maintaining the balance of the neurotransmitters; GABA, nitric oxide, serotonin, and acetylcholine that are associated with pain disorders and inflammatory conditions. Theramine stimulates the production ACTH to reduce inflammation.
Metabolism
The amino acids in Theramine are primarily absorbed by the stomach and small intestines. All cells metabolize the amino acids in Theramine. Circulating tryptophan, arginine and choline blood levels determine the production of serotonin, nitric oxide, and acetylcholine.
Excretion
Theramine is not an inhibitor of cytochrome P450 1A2, 2C9, 2C19, 2D6, or 3A4. These isoenzymes are principally responsible for 95% of all detoxification of drugs, with CYP3A4 being responsible for detoxification of roughly 50% of drugs. Amino acids do not appear to have an effect on drug metabolizing enzymes.
INDICATIONS FOR USE
Theramine is intended for the clinical dietary management of the metabolic processes of pain disorders and inflammatory conditions.
CLINICAL EXPERIENCE
Administration of Theramine has demonstrated significant reduction in symptoms of pain and inflammation in patients with acute and chronic pain when used for the dietary management of the metabolic processes associated with pain disorders and inflammatory conditions. Administration of Theramine results in the induction and maintenance of pain relief in patients with pain disorders and inflammatory conditions.
PRECAUTIONS AND CONTRAINDICATIONS
Theramine is contraindicated in an extremely small number of patients with hypersensitivity to any of the nutritional components of Theramine.
ADVERSE REACTIONS
Oral supplementation with L-tryptophan, L-arginine or choline at high doses up to 15 grams daily is generally well tolerated. The most common adverse reactions of higher doses — from 15 to 30 grams daily — are nausea, abdominal cramps, and diarrhea. Some patients may experience these symptoms at lower doses. The total combined amount of amino acids in each Theramine capsule does not exceed 400 mg.
DRUG INTERACTIONS
Theramine does not directly influence the pharmacokinetics of prescription drugs. Clinical experience has shown that administration of Theramine may allow for lowering the dose of co-administered drugs under physician supervision.
OVERDOSE
There is a negligible risk of overdose with Theramine as the total dosage of amino acids in a one month supply (90 capsules) is less than 36 grams. Overdose symptoms may include diarrhea, weakness, and nausea.
POST-MARKETING SURVEILLANCE
Post-marketing surveillance has shown no serious adverse reactions. Reported cases of mild rash and itching may have been associated with allergies to Theramine flavonoid ingredients, including cinnamon, cocoa, and chocolate. These reactions were transient in nature and subsided within 24 hours.
DOSAGE AND ADMINISTRATION
Recommended Administration For the dietary management of the metabolic processes associated with pain disorders and inflammatory conditions. Take (2) capsules one to three times daily or as directed by physician. As with most amino acid formulations Theramine should be taken without food to increase the absorption of key ingredients.
How Supplied
Theramine is supplied in purple and white, size 0 capsules in bottles of 60 or 90 capsules.
Physician Supervision
Theramine is a Medical Food product available by prescription only and must be used while the patient is under ongoing physician supervision.
U.S. patent pending.
Manufactured by Arizona Nutritional Supplements, Inc. Chandler AZ 85225
Distributed by Physician Therapeutics LLC, Los Angeles, CA 90077. www.ptlcentral.com
Copyright 2003-2006, Physician Therapeutics LLC, all rights reserved
NDC: 68405-1008-02
NDC: 68405-1008-03
Storage
Store at room temperature, 59-86OF (15-30OC) Protect from light and moisture. Theramine is supplied to physicians in a recyclable plastic bottle with a child-resistant cap.