FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
AK-FLUOR® 10% (100 mg/mL) and 25% (250 mg/mL) is indicated in diagnostic fluorescein angiography or angioscopy of the retina and iris vasculature.
2 DOSAGE AND ADMINISTRATION
2.1 Dosing
2.2 Preparation for Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not mix or dilute with other solutions or drugs.
2.3 Administration
Inject the dose (over 5-10 seconds is normally recommended) into the antecubital vein, after taking precautions to avoid extravasation. A syringe, filled with AK-FLUOR®, may be attached to transparent tubing and a 23 gauge butterfly needle for injection. Insert the needle and draw the patient's blood to the hub of the syringe so that a small air bubble separates the patient's blood in the tubing from the fluorescein. With the room lights on, slowly inject the blood back into the vein while watching the skin over the needle tip. If the needle has extravasated, the patient's blood will be seen to bulge the skin and the injection should be stopped before any fluorescein is injected. When assured that extravasation has not occurred, the room light may be turned off and the fluorescein injection completed. Luminescence usually appears in the retina and choroidal vessels in 7 to 14 seconds and can be observed by standard viewing equipment.
Reduction in dose from 500 mg to 200 mg of AK-FLUOR® 10% may be appropriate in cases when a highly sensitive imaging system e.g., scanning laser ophthalmoscope is used.
3 DOSAGE FORMS AND STRENGTHS
AK-FLUOR® (fluorescein injection, USP) 10%, 100 mg/mL in a 5 mL single-dose vial.
AK-FLUOR® (fluorescein injection, USP) 25%, 250 mg/mL in a 2 mL single-dose vial.
4 CONTRAINDICATIONS
4.1 Hypersensitivity
AK-FLUOR® is contraindicated in patients with known hypersensitivity to fluorescein sodium or any other ingredients in this product. Rare cases of death due to anaphylaxis have been reported [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Respiratory Reactions
Caution should be exercised in patients with a history of allergy or bronchial asthma. An emergency tray should always be available.
If a potential allergy is suspected, an intradermal skin test may be performed prior to intravenous administration, i.e., 0.05 mL injected intradermally to be evaluated 30 to 60 minutes following injection. Given the sensitivity and specificity of skin testing, a negative skin test is not proof that a patient is not allergic to fluorescein.
5.2 Severe local tissue damage
Extravasation during injection can result in severe local tissue damage due to high pH of fluorescein solution. The following complications resulting from extravasation of fluorescein have been noted to occur: Sloughing of the skin, superficial phlebitis, subcutaneous granuloma, and toxic neuritis along the median nerve in the antecubital area. Complications resulting from extravasation can cause severe pain in the arm for up to several hours. When extravasation occurs, the injection should be discontinued and conservative measures to treat damaged tissue and to relieve pain should be implemented. [see Administration (2.3) and Adverse Reactions (6.6)].
6 ADVERSE REACTIONS
6.1 Skin and urine discoloration
The most common reaction is discoloration of the skin and urine. Skin will attain a temporary yellowish discoloration. Urine attains a bright yellow color. Discoloration of the skin usually fades in 6 to 12 hours and usually fades in urine in 24 to 36 hours.
6.2 Gastrointestinal Reaction
The next most common adverse reaction is nausea. Vomiting, and gastrointestinal distress have also occurred. A strong taste may develop after injection.
6.3 Hypersensitivity Reactions
Symptoms and signs of hypersensitivity have occurred. Generalized hives and itching, bronchospasm and anaphylaxis have been reported. [see Contraindications (4.1) and Warnings and Precautions (5.1)]
6.4 Cardiopulmonary Reactions
Syncope and hypotension may occur. Cardiac arrest, basilar artery ischemia, severe shock and death may occur rarely. [see Warnings and Precautions (5.1)]
6.6 Thrombophlebitis
Thrombophlebitis at the injection site has been reported. Extravasation of the solution at the injection site causes intense pain at the site and a dull aching pain in the injected arm. [see Administration (2.3) and Warnings and Precautions (5.2)].
8 USE IN SPECIFIC POPULATIONS
8.3 Nursing Mothers
Fluorescein sodium has been demonstrated to be excreted in human milk. Caution should be exercised when fluorescein sodium is administered to a nursing woman.
11 DESCRIPTION
AK-FLUOR® (fluorescein injection, USP) is a sterile solution for use intravenously as a diagnostic aid. It is a dark reddish orange solution with a pH of 8.3 to 9.8 and an osmolality of 572 to 858 mOsm/kg for the 10% and 1800 to 2200 mOsm/kg for the 25%. Its chemical name is spiro[isobenzofuran-1 (3H),9'-[9H]xanthene]-3-one,3'6'-dihydroxy, disodium salt. The active ingredient is represented by the chemical structure:
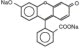
MW = 376.27
AK-FLUOR® 10% contains:
Active: fluorescein sodium (equivalent to fluorescein 10 % w/v, 100 mg/mL)
Inactives: Sodium Hydroxide and/or Hydrochloric Acid may be used to adjust pH (8.3 to 9.8), and Water for Injection.
AK-FLUOR® 25% contains:
Active: fluorescein sodium (equivalent to fluorescein 25% w/v, 250 mg/mL)
Inactives: Sodium Hydroxide and/or Hydrochloric Acid may be used to adjust pH (8.3 to 9.8), and Water for Injection.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Fluorescein sodium responds to electromagnetic radiation and light between the wavelengths of 465 to 490 nm and fluoresces, i.e., emits light at wavelengths of 520 to 530 nm. Thus, the hydrocarbon is excited by blue light and emits light that appears yellowish green. Following intravenous injection of fluorescein sodium in an aqueous solution, the unbound fraction of the fluorescein can be excited with a blue light flash from a fundus camera as it circulates through the ocular vasculature, and the yellowish green fluorescence of the dye is captured by the camera. In the fundus, the fluorescence of the dye demarcates the retinal and/or choroidal vasculature under observation, distinguishing it from adjacent areas/structures.
12.3 Pharmacokinetics
Distribution.
Within 7 to 14 seconds after IV administration into the antecubital vein, fluorescein usually appears in the central retinal artery of the eye. Within a few minutes of IV administration of fluorescein sodium, a yellowish discoloration of the skin occurs, which begins to fade 6 to 12 hours after dosing. Various estimates of volume of distribution indicate that fluorescein distributes into interstitial space (0.5 L/kg).
Metabolism.
Fluorescein is metabolized to fluorescein monoglucuronide. After IV administration of fluorescein sodium (14 mg/kg) to 7 healthy subjects, approximately 80% of fluorescein in plasma was converted to glucuronide conjugate after a period of 1 hour post dose.
Excretion.
Fluorescein and its metabolite are mainly eliminated via renal excretion. After IV administration, the urine remains slightly fluorescent for 24 to 36 hours. A renal clearance of 1.75 mL/min/kg and a hepatic clearance (due to conjugation) of 1.50 mL/min/kg have been estimated. The systemic clearance of fluorescein was essentially complete by 48 to 72 hours after administration of 500 mg fluorescein.
16 HOW SUPPLIED/STORAGE AND HANDLING
AK-FLUOR® (fluorescein injection, USP) 10% is supplied in a single-dose 5 mL glass vial with a gray bromobutyl serum siliconized stopper and orange flip-off cap. It contains a sterile dark reddish orange solution of fluorescein sodium.
(NDC 17478-253-10) 5 mL, single dose vials in a package of 12.
AK-FLUOR® (fluorescein injection, USP) 25% is supplied in a single-dose 2 mL glass vial with a gray bromobutyl serum siliconized stopper and orange flip-off cap. It contains a sterile dark reddish orange solution of fluorescein sodium.
(NDC 17478-250-20) 2 mL, single dose vials in a package of 12.
17 PATIENT COUNSELING INFORMATION
After administration of fluorescein sodium, skin will attain a temporary yellowish discoloration. Urine attains a bright yellow color. Discoloration of the skin usually fades in 6 to 12 hours and usually fades in urine in 24 to 36 hours. [see Warnings and Precautions (6.1)].
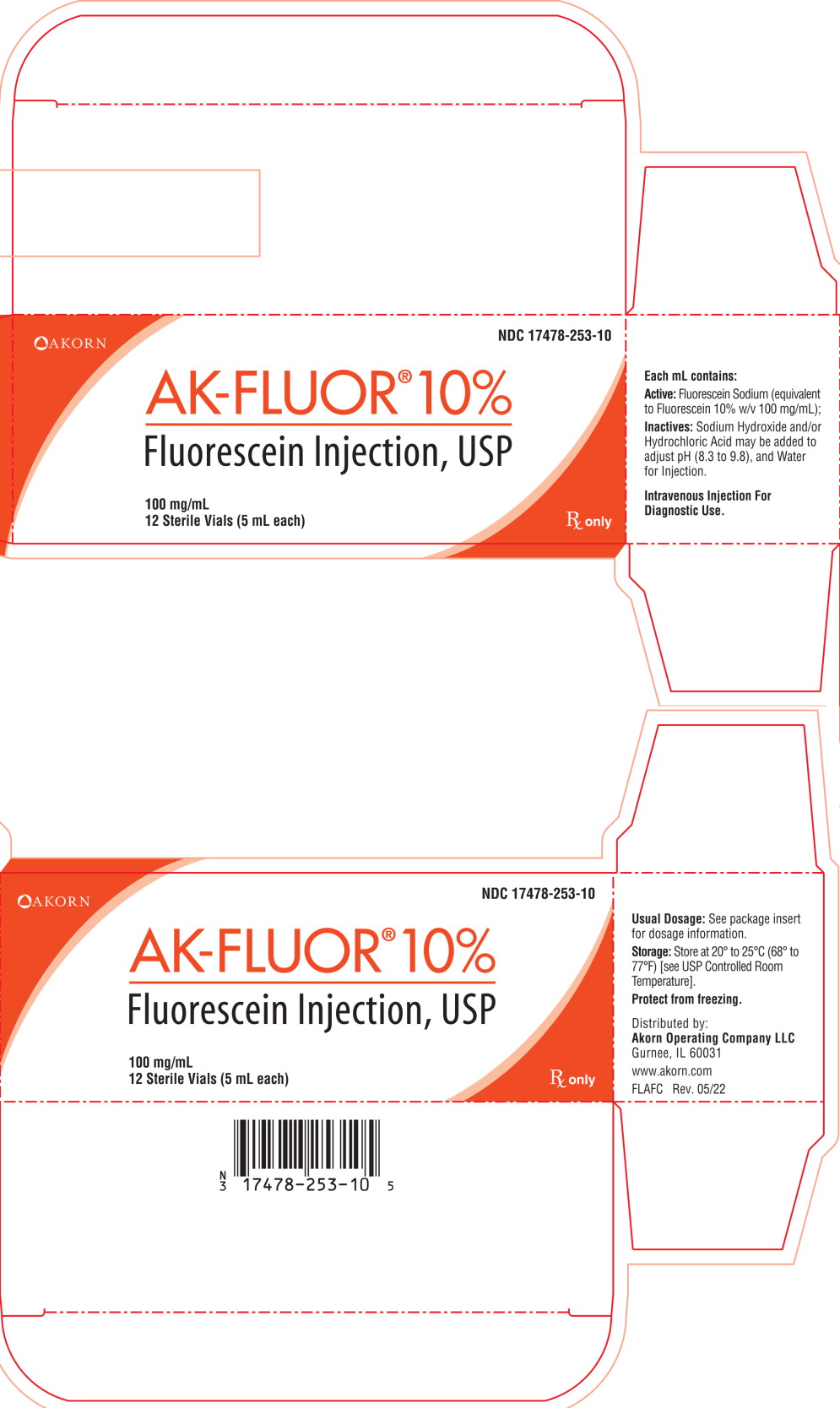
Principal Display Panel Text for Container Label:
NDC 17478-253-10
AK-FLUOR® 10%
Fluorescein Injection, USP
100 mg/mL
5mL Sterile Vial
For IV Injection. Rx only
Principal Display Panel Text for Carton Label:
Akorn Logo NDC 17478-253-10
AK-FLUOR® 10%
Fluorescein Injection, USP
100 mg/mL
12 Sterile Vials (5 mL each) Rx only