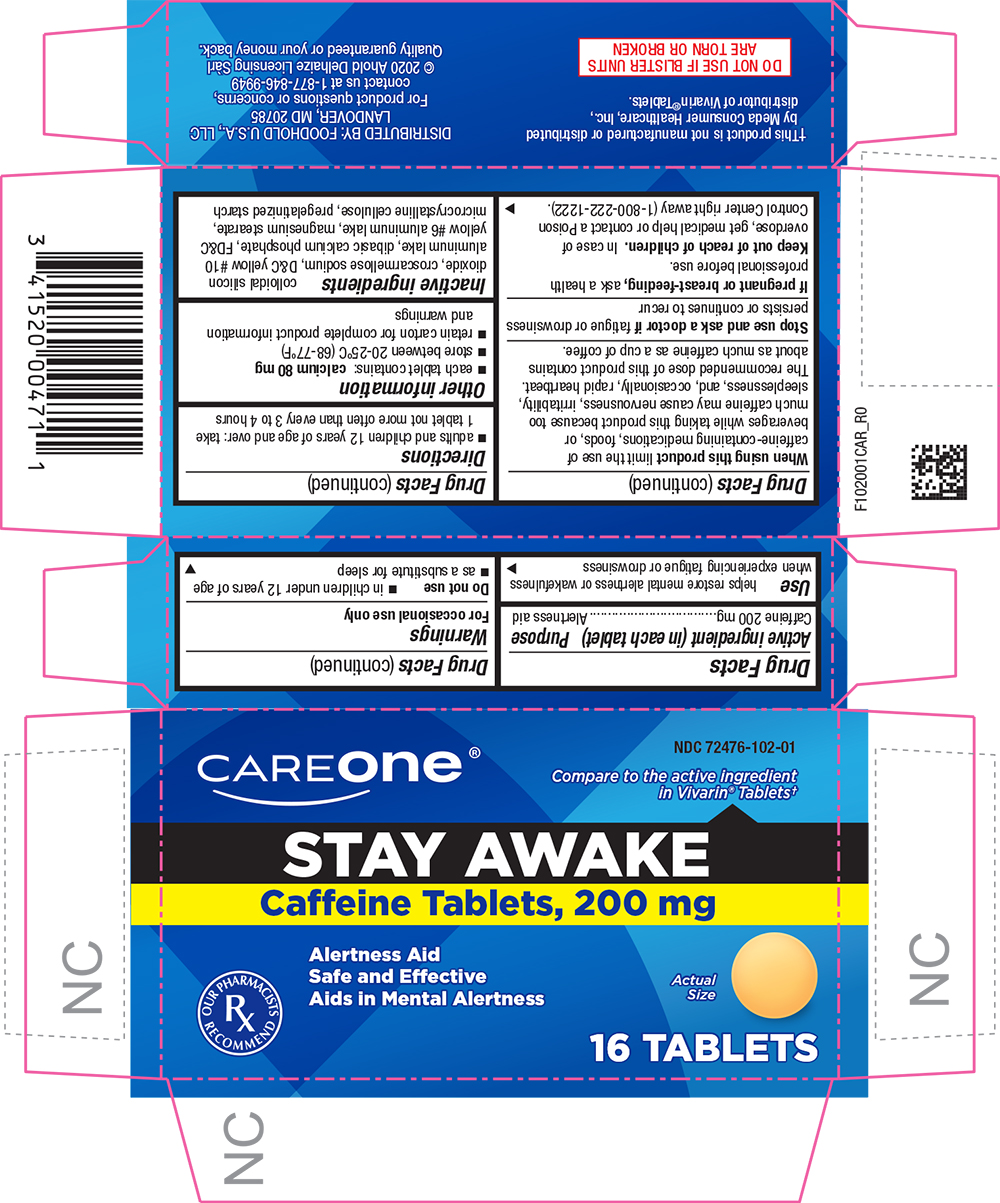
STAY AWAKE- caffeine tablet
RETAIL BUSINESS SERVICES , LLC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active ingredient (in each tablet)
Caffeine 200 mg
Use
helps restore mental alertness or wakefulness when experiencing fatigue or drowsiness
Warnings
For occasional use only
Do not use
- in children under 12 years of age
- as a substitute for sleep
When using this product limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heartbeat. The recommended dose of this product contains about as much caffeine as a cup of coffee.
Stop use and ask a doctor if fatigue or drowsiness persists or continues to recur
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Directions
- adults and children 12 years of age and over: take 1 tablet not more often than every 3 to 4 hours
Other information
- each tablet contains:
calcium 80 mg
- store between 20-25°C (68-77°F)
- retain carton for complete product information and warnings
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, D&C yellow #10 aluminum lake, dibasic calcium phosphate, FD&C yellow #6 aluminum lake, magnesium stearate, microcrystalline cellulose, pregelatinized starch
PRINCIPAL DISPLAY PANEL
NDC 72476-102-01
CAREONE®
Compare to the active ingredient in Vivarin® Tablets†
STAY AWAKE
Caffeine Tablets, 200 mg
Alertness Aid
Safe and Effective
Aids in Mental Alertness
Actual Size
16 TABLETS
RETAIL BUSINESS SERVICES , LLC.
