FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
QINLOCK is indicated for the treatment of adult patients with advanced gastrointestinal stromal tumor (GIST) who have received prior treatment with 3 or more kinase inhibitors, including imatinib.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of QINLOCK is 150 mg orally once daily with or without food until disease progression or unacceptable toxicity.
Instruct patients to swallow tablets whole.
Advise patients to take QINLOCK at the same time each day.
Advise patients to take a missed dose if less than 8 hours have passed since the missed scheduled dose.
Advise patients not to take an additional dose if vomiting occurs after taking QINLOCK and to continue with their next scheduled dose.
2.2 Dosage Modifications for Adverse Reactions
The recommended dose reduction for adverse reactions is:
- QINLOCK 100 mg orally once daily.
Permanently discontinue QINLOCK in patients who are unable to tolerate 100 mg orally once daily.
The recommended dosage modifications of QINLOCK for adverse reactions are provided in Table 1.
| Adverse Reaction | Severitya | QINLOCK Dosage Modifications |
|---|---|---|
|
a Graded per National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03 (NCI CTCAE v4.03). |
||
| Palmar-Plantar Erythrodysesthesia Syndrome (PPES) [see Warnings and Precautions (5.1)] | Grade 2 |
|
| Grade 3 |
|
|
| Hypertension [see Warnings and Precautions (5.3)] | Grade 3 |
|
| Grade 4 | Permanently discontinue QINLOCK. | |
| Left Ventricular Systolic Dysfunction [see Warnings and Precautions (5.4)] | Grade 3 or 4 | Permanently discontinue QINLOCK. |
| Arthralgia or Myalgia [see Adverse Reactions (6.1)] | Grade 2 |
|
| Grade 3 |
|
|
| Other Adverse Reactions [see Adverse Reactions (6.1)] | Grade 3 or 4 |
|
2.3 Dose Modifications for Moderate CYP3A Inducers
Avoid concomitant use of moderate CYP3A inducers during QINLOCK treatment.
If a moderate CYP3A inducer cannot be avoided, increase the QINLOCK dosing frequency from the recommended dose of 150 mg once daily to 150 mg twice daily during the co-administration period. Monitor for clinical response and tolerability. If the concomitant moderate CYP3A inducer is discontinued, resume QINLOCK dosage back to 150 mg once daily 14 days after the discontinuation of the moderate CYP3A inducer [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
For patients concomitantly using a moderate CYP3A inducer with QINLOCK (taking QINLOCK twice daily) who missed a dose:
- If less than 4 hours have passed since the missed scheduled dose, advise the patient to take the missed dose as soon as possible and then take the next dose at the regularly scheduled time.
- If more than 4 hours have passed since the missed scheduled dose, advise the patient to skip the missed dose and then take the next dose at the regularly scheduled time. [see Drug Interactions (7.1)].
3 DOSAGE FORMS AND STRENGTHS
Tablets: 50 mg, white to off-white, oval shaped, debossed with “DC1” on one side.
5 WARNINGS AND PRECAUTIONS
5.1 Palmar-Plantar Erythrodysesthesia Syndrome
In INVICTUS, Grade 1-2 palmar-plantar erythrodysesthesia syndrome (PPES) occurred in 21% of the 85 patients who received QINLOCK [see Adverse Reactions (6.1)]. PPES led to dose discontinuation in 1.2% of patients, dose interruption in 2.4% of patients, and dose reduction in 1.2% of patients.
Based on severity, withhold QINLOCK and then resume at same or reduced dose [see Dosage and Administration (2.2)].
5.2 New Primary Cutaneous Malignancies
In INVICTUS, cutaneous squamous cell carcinoma (cuSCC) occurred in 4.7% of the 85 patients who received QINLOCK, with a median time to event of 4.6 months (range: 3.8 to 6 months). In the pooled safety population, cuSCC and keratoacanthoma occurred in 7% and 1.9% of patients, respectively.
In INVICTUS, melanoma occurred in 2.4% of the 85 of patients who received QINLOCK. In the pooled safety population, melanoma occurred in 0.9% of patients.
Perform dermatologic evaluations when initiating QINLOCK and routinely during treatment. Manage suspicious skin lesions with excision and dermatopathologic evaluation. Continue QINLOCK at the same dose.
5.3 Hypertension
In INVICTUS, Grade 1-3 hypertension occurred in 14% of the 85 patients who received QINLOCK, including Grade 3 hypertension in 7% [see Adverse Reactions (6.1)].
Do not initiate QINLOCK in patients with uncontrolled hypertension. Adequately control blood pressure prior to initiating QINLOCK. Monitor blood pressure as clinically indicated during treatment with QINLOCK, and initiate or adjust antihypertensive therapy as appropriate. Based on severity, withhold QINLOCK and then resume at same or reduced dose or permanently discontinue [see Dosage and Administration (2.2)].
5.4 Cardiac Dysfunction
In INVICTUS, cardiac failure occurred in 1.2% of the 85 patients who received QINLOCK. In the pooled safety population, cardiac dysfunction (including cardiac failure, acute left ventricular failure, diastolic dysfunction, and ventricular hypertrophy) occurred in 1.7% of patients, including Grade 3 adverse reactions in 1.1%.
In INVICTUS, Grade 3 decreased ejection fraction occurred in 2.6% of the 77 patients who received QINLOCK and who had a baseline and at least one post-baseline echocardiogram. In the pooled safety population, Grade 3 decreased ejection fraction occurred in 3.4% of the 263 patients who received QINLOCK and who had a baseline and at least one post-baseline echocardiogram.
In INVICTUS, cardiac dysfunction led to dose discontinuation in 1.2% of the 85 patients who received QINLOCK. The safety of QINLOCK has not been assessed in patients with a baseline ejection fraction below 50%.
Assess ejection fraction by echocardiogram or MUGA scan prior to initiating QINLOCK and during treatment, as clinically indicated. Permanently discontinue QINLOCK for Grade 3 or 4 left ventricular systolic dysfunction [see Dosage and Administration (2.2)].
5.5 Risk of Impaired Wound Healing
Impaired wound healing complications can occur in patients who receive drugs that inhibit the vascular endothelial growth factor (VEGF) signaling pathway. Therefore, QINLOCK has the potential to adversely affect wound healing.
Withhold QINLOCK for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of QINLOCK after resolution of wound healing complications has not been established.
>5.6 Photosensitivity
QINLOCK may cause photosensitivity reactions. In all patients treated with QINLOCK in clinical trials (n=621), photosensitivity reactions occurred in 0.6% of patients.
Advise patients to limit direct ultraviolet exposure during treatment with QINLOCK and for at least one week after discontinuation of treatment.
5.7 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, QINLOCK can cause fetal harm when administered to a pregnant woman. Oral administration of ripretinib to pregnant rats and rabbits during the period of organogenesis resulted in malformations primarily associated with the cardiovascular and skeletal systems, anatomic variations, decreased fetal body weight, and increased post-implantation loss at exposures approximately one half of the recommended dose of 150 mg once daily based on area under the curve (AUC).
Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with QINLOCK and for 1 week after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with QINLOCK and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed elsewhere in the labeling:
- Palmar-Plantar Erythrodysesthesia Syndrome [see Warnings and Precautions (5.1)]
- New Primary Cutaneous Malignancies [see Warnings and Precautions (5.2)]
- Hypertension [see Warnings and Precautions (5.3)]
- Cardiac Dysfunction [see Warnings and Precautions (5.4)]
- Photosensitivity [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Unless otherwise specified, the pooled safety population described in the WARNINGS AND PRECAUTIONS reflect exposure to QINLOCK as a single agent in 351 patients with advanced solid tumors enrolled in either an open-label dose finding with cohort expansion trial or INVICTUS. Among the patients who received QINLOCK in these trials, 52% were exposed for 6 months or longer and 21% were exposed for greater than one year.
Patients Who Received Prior Treatment with Imatinib, Sunitinib and Regorafenib
The safety of QINLOCK was evaluated in INVICTUS [see Clinical Studies (14)]. Patients received QINLOCK 150 mg taken orally once daily (n=85) or placebo (n=43). Among the patients who received QINLOCK, 46% were exposed for 6 months or longer and 3.5% were exposed for greater than one year.
Serious adverse reactions occurred in 31% of patients who received QINLOCK. Serious adverse reactions that occurred in >2% of patients were abdominal pain (4.7%), anemia (3.5%), nausea (2.4%), and vomiting (2.4%).
Permanent discontinuation due to an adverse reaction occurred in 8% of patients who received QINLOCK. Adverse reactions resulting in permanent discontinuation in ≥1% of patients included general physical health deterioration (2.4%), anemia (1.2%), cardiac failure (1.2%), PPES (1.2%), and vomiting (1.2%).
Dosage interruptions due to an adverse reaction occurred in 24% of patients who received QINLOCK. Adverse reactions requiring dosage interruption in >2% of patients included nausea (3.5%), increased blood bilirubin (2.4%), and PPES (2.4%).
Dose reductions due to an adverse reaction occurred in 7% of patients who received QINLOCK. Adverse reactions resulting in a dose reduction in ≥1.2% of patients were abdominal pain, agitation, alopecia, arthritis, dermatosis, gastrointestinal disorder, hyperesthesia, myalgia, PPES, and decreased weight.
The most common adverse reactions (≥20%), were alopecia, fatigue, nausea, abdominal pain, constipation, myalgia, diarrhea, decreased appetite, PPES, and vomiting. The most common Grade 3 or 4 laboratory abnormalities (≥4%) were increased lipase and decreased phosphate.
Table 2 summarizes the adverse reactions in INVICTUS.
| Adverse Reaction | QINLOCK (N=85) | Placebo (N=43) |
||
| Grades 1-4 | Grades 3-4 | Grades 1-4 | Grades 3-4 | |
| Skin and subcutaneous tissue | ||||
| Alopecia | 52 | 0 | 4.7 | 0 |
| Palmar-plantar erythrodysesthesia syndrome | 21 | 0 | 0 | 0 |
| Dry skin | 13 | 0 | 7 | 0 |
| Pruritus | 11 | 0 | 4.7 | 0 |
| General | ||||
| Fatigue | 42 | 3.5 | 23 | 2.3 |
| Peripheral edema | 17 | 1.2 | 7 | 0 |
| Asthenia | 13 | 1.2 | 14 | 4.7 |
| Gastrointestinal | ||||
| Nausea | 39 | 3.5 | 12 | 0 |
| Abdominal pain | 36 | 7 | 30 | 4.7 |
| Constipation | 34 | 1.2 | 19 | 0 |
| Diarrhea | 28 | 1.2 | 14 | 2.3 |
| Vomiting | 21 | 3.5 | 7 | 0 |
| Stomatitis | 11 | 0 | 0 | 0 |
| Musculoskeletal and connective tissue | ||||
| Myalgia | 32 | 1.2 | 12 | 0 |
| Arthralgia | 18 | 0 | 4.7 | 0 |
| Muscle spasms | 15 | 0 | 4.7 | 0 |
| Metabolism and nutrition | ||||
| Decreased appetite | 27 | 1.2 | 21 | 2.3 |
| Investigations | ||||
| Decreased weight | 19 | 0 | 12 | 0 |
| Nervous system | ||||
| Headache | 19 | 0 | 4.7 | 0 |
| Vascular | ||||
| Hypertension | 14 | 7 | 4.7 | 0 |
| Respiratory, thoracic and mediastinal | ||||
| Dyspnea | 13 | 0 | 0 | 0 |
Table 3 summarizes the laboratory abnormalities in INVICTUS.
|
CPK=creatine phosphokinase; INR=international normalized ratio; AST=aspartate aminotransferase; ALT=alanine aminotransferase |
||||
|
a. The denominator used to calculate the rate varied from 82 to 83 for QINLOCK and 34 to 40 for placebo based on the number of patients with a baseline value and at least one post-treatment value. |
||||
|
b. Only includes Grade 3 laboratory abnormalities. |
||||
| Laboratory Abnormality | QINLOCKa
(N=85) | Placeboa
(N=43) |
||
| Grades 1-4 | Grades 3-4b | Grades 1-4 | Grades 3-4 | |
| Hematology | ||||
| Increased activated partial thromboplastin time | 35 | 0 | 9 | 0 |
| Increased INR | 21 | 3.8 | 15 | 0 |
| Decreased neutrophil count | 10 | 0 | 2.5 | 0 |
| Chemistry | ||||
| Increased lipase | 32 | 7 | 13 | 8 |
| Decreased phosphate | 26 | 4.9 | 2.5 | 0 |
| Increased triglycerides | 26 | 2.4 | 23 | 0 |
| Decreased calcium | 23 | 0 | 8 | 0 |
| Increased blood bilirubin | 22 | 0 | 5 | 2.5 |
| Increased CPK | 21 | 1.2 | 10 | 0 |
| Decreased sodium | 17 | 2.4 | 10 | 2.5 |
| Increased creatinine | 16 | 0 | 18 | 0 |
| Increased serum amylase | 13 | 1.2 | 5 | 0 |
| Increased ALT | 12 | 1.2 | 5 | 0 |
Other Adverse Reactions
Clinically relevant adverse reactions that occurred in <10% of QINLOCK-treated patients in INVICTUS included peripheral sensory neuropathy, dermatitis acneiform, and rash.
Clinically relevant adverse reactions that occurred in <10% of patients in the pooled safety population included cardiac ischemic events (including acute coronary syndrome and fatal cardiac arrest or myocardial infarction). Photosensitivity occurred in 0.6% [see Warnings and Precautions (5.6)].
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on QINLOCK
Table 4 includes drug interactions that affect the pharmacokinetics of ripretinib.
| Strong CYP3A Inhibitors | |
| Clinical Impact |
|
| Prevention or Management |
|
| Strong and Moderate CYP3A Inducers | |
| Clinical Impact |
|
| Prevention or Management |
|
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action [see Clinical Pharmacology (12.1)], QINLOCK can cause fetal harm when administered to a pregnant woman. There are no available data on the use of QINLOCK in pregnant women to inform a drug-associated risk. Administration of ripretinib to pregnant rats and rabbits during the period of organogenesis resulted in malformations primarily associated with the cardiovascular and skeletal systems, anatomic variations, reduced fetal body weight, and increased post-implantation loss at maternal exposures that were approximately equal to the human exposure at the recommended dose of 150 mg (see Data). Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In an embryo-fetal development study investigating daily doses of ripretinib administered during the period of organogenesis in rats, ripretinib resulted in malformations primarily associated with the cardiovascular and skeletal systems, including interrupted or retroesophageal aortic arch and retroesophageal subclavian artery, fusion of the exoccipital bone to the first cervical vertebra, branched and fused ribs, anomalies of the cervical, thoracic, caudal, and sacral vertebrae, absent forepaw phalanges, and absent metacarpals at a dose of 20 mg/kg/day (approximately one half of the human exposure at the recommended dose of 150 mg). An increased incidence of anatomic variations were also observed at 20 mg/kg/day. Variations included malpositioned carotid and subclavian artery origins, malpositioned subclavian artery, absent or elongated innominate artery, misshapen and nodulated ribs, bipartite, incompletely ossified, or unossified vertebral centra, small or misshapen vertebral arches, and reductions in ossified forelimb and hindlimb phalanges, hindlimb metatarsals, and caudal vertebrae.
In a preliminary embryo-fetal development study investigating the administration of ripretinib in rabbits during the period of organogenesis, ripretinib resulted in total loss of pregnancy at doses of 150 mg/kg (approximately 3.5 times the human exposure at the recommended dose of 150 mg). At a dose of 40 mg/kg (approximately 2.1 times the human exposure at the recommended dose of 150 mg), toxicities included increased post-implantation loss and decreased fetal body weights.
8.2 Lactation
Risk Summary
There are no data regarding the presence of ripretinib or its metabolites in either human milk or its effects on a breastfed child or on milk production. Because of the potential for serious adverse reactions in the breastfed child, advise women not to breastfeed during treatment with QINLOCK and for 1 week after the last dose.
8.3 Females and Males of Reproductive Potential
QINLOCK can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to the initiation of QINLOCK [see Use in Specific Populations (8.1)].
Contraception
Infertility
Based on findings from animal studies, QINLOCK may impair fertility in males of reproductive potential [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of QINLOCK in pediatric patients have not been established.
Animal Toxicity Data
In 13-week repeat-dose studies in rats there were dose-dependent findings of increased osteoblastic surface and decreased trabeculae of the femur at doses ≥30 mg/kg/day (approximately one half of the human exposure at the recommended dose of 150 mg). There were additional findings of missing or discolored teeth that were accompanied by dose-dependent incisor degeneration at doses ≥30 mg/kg/day.
8.5 Geriatric Use
Of the 85 patients in INVICTUS who received QINLOCK 150 mg orally once daily, 24% were between 65 to 74 years of age and 9% were 75 years of age or older. Clinical studies of QINLOCK did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger patients.
8.6 Hepatic Impairment
No dose adjustment is recommended in patients with hepatic impairment (Child-Pugh A, B, or C) [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
Ripretinib is a kinase inhibitor. The chemical name of ripretinib is 1-(4-bromo-5-[1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl]-2-fluorophenyl)-3-phenylurea. The molecular formula is C24H21BrFN5O2 and the molecular weight is 510.36 g/mol. The chemical structure of ripretinib is shown below:
Ripretinib is a white to off-white crystalline solid. Ripretinib is a lipophilic, weak base, and practically insoluble in aqueous media.
QINLOCK is available as a white to off-white, oval tablets for oral use containing 50 mg of ripretinib. The tablet is debossed with “DC1” on one side. Each tablet contains the following inactive ingredients: crospovidone, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and silicon dioxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ripretinib is a tyrosine kinase inhibitor that inhibits KIT proto-oncogene receptor tyrosine kinase (KIT) and platelet derived growth factor receptor A (PDGFRA) kinase, including wild type, primary, and secondary mutations. Ripretinib also inhibits other kinases in vitro, such as PDGFRB, TIE2, VEGFR2, and BRAF.
12.2 Pharmacodynamics
12.3 Pharmacokinetics
The pharmacokinetics of ripretinib and its equally active metabolite (DP-5439) were characterized in clinical studies. In patients with advanced malignancies, ripretinib AUC0-24h increased proportionally over a dose range of 20-250 mg (0.13 to 1.67 times the recommended dose), but Cmax was less than dose proportional; DP-5439 Cmax and AUC0-24h were less than dose proportional within the dose range of 50-250 mg (0.33 to 1.67 times the recommended dose).
No clinically significant differences in the Cmax and AUC0-24h were observed between administration of QINLOCK with a high-fat meal (150, 250, and 500-600 calories from protein, carbohydrate, and fat, respectively) and under fasted conditions. The pharmacokinetic parameters of ripretinib and DP-5439 are summarized in Table 5.
|
a. Estimated based on cycle 1, day 15 |
|||
|
b. After a single oral dose of 150 mg |
|||
|
CV=coefficient of variation; Cmax=maximum plasma concentration; AUC0-12h=area under the plasma concentration-time curve from time zero to 12 hours; AUC0-24h=area under the plasma concentration-time curve from time zero to 24 hours; Tmax=time to maximum concentration |
|||
| Parameter | Ripretinib | DP-5439 | |
| General Information | |||
| Steady state exposure following QINLOCK 150 mg once daily [Mean (CV%)] | Cmax (ng/mL) | 761 (32) | 804 (46) |
| AUC0-12h (ng•h/mL) | 5678 (32) | 7138 (44) | |
| Time to steady state [Days] | 14 | 14 | |
| Accumulation ratio (AUC0-12h) [Mean (CV%)]a | 1.7 (55) | 5.29 (49) | |
| Absorption | |||
| Tmax [Median in hours]b | 4 | 15.6 | |
| Distribution | |||
| Plasma protein binding (in vitro) | Human serum albumin | 99.8% | 99.7% |
| α-1 acid glycoprotein | 99.4% | >99.8% | |
| Steady state apparent volume of distribution, L [Mean (CV%)]b | 307 (39) | 507 (51) | |
| Elimination | |||
| Apparent clearance, L/hr [Mean (CV%)]b | 15.3 (45) | 17.5 (63) | |
| Half-life, hours [Mean (CV%)]b | 14.8 (30) | 17.8 (23) | |
| Metabolism | |||
| Metabolic pathways | Major | CYP3A4 | CYP3A4 |
| Minor | CYP2C8 and CYP2D6 | CYP2C8, CYP2E1 and CYP2D6 | |
| Excretion | |||
| Excretion pathways | Feces | 34% | 6% |
| Urine | 0.02% | 0.1% | |
Specific Populations
No clinically significant differences in the pharmacokinetics of ripretinib were observed based on age (19 to 87 years), sex, race (White, Black, and Asian), body weight (39 to 138 kg), tumor (GIST or other solid tumors), prior gastrectomy, mild to moderate renal impairment (CLcr 30 to <90 mL/min estimated by Cockcroft-Gault), and mild hepatic impairment (total bilirubin ≤ULN and AST >ULN or total bilirubin 1 to 1.5 × ULN and AST any).
The effects of severe renal impairment (CLcr 15 to 29 mL/min) on the pharmacokinetics of ripretinib have not been studied.
Patients with Hepatic Impairment
In subjects with mild hepatic impairment, there were no clinically significant changes in AUC0-last and Cmax when compared to matched healthy subjects.
In subjects with moderate hepatic impairment, ripretinib AUC0-last increased 2-fold while Cmax was unchanged when compared to matched healthy subjects. The combined AUC0-last of ripretinib and DP-5439 increased 1.5-fold.
In subjects with severe hepatic impairment, ripretinib AUC0-last increased 2.6-fold and Cmax decreased by 24% when compared to matched healthy subjects. The combined AUC0-last of ripretinib and DP-5439 increased by 1.4-fold.
The observed magnitude of increase in ripretinib and the combination of ripretinib and DP-5439 exposures in subjects with hepatic impairment is unlikely to be clinically relevant based on the known safety profile of ripretinib.
Clinical Studies and Model-Informed Approaches
Strong CYP3A Inhibitors: Coadministration of QINLOCK with itraconazole (a strong CYP3A inhibitor and also a P-gp inhibitor) increased ripretinib Cmax by 36% and AUC0-inf by 99% and also increased DP-5439 AUC0-inf by 99% with no change in its Cmax.
Strong CYP3A Inducers: Coadministration of QINLOCK with rifampin (a strong CYP3A inducer) decreased ripretinib Cmax by 18% and AUC0-inf by 61% and also decreased DP-5439 AUC0-inf by 57% with increased Cmax by 37%.
Moderate CYP3A Inducers: Coadministration of QINLOCK with efavirenz (a moderate CYP3A inducer) was predicted to decrease ripretinib Cmax by 24% and decrease AUC0-inf by 56%.
Proton Pump Inhibitors: No clinically significant differences in the plasma exposure to ripretinib and DP-5439 were observed when QINLOCK was coadministered with pantoprazole (a proton pump inhibitor).
In Vitro Studies
CYP Enzymes: Ripretinib and DP-5439 are inhibitors of CYP2C8. Ripretinib and DP-5439 are not inducers of CYP1A2, CYP2B6, or CYP3A4.
Transporter Systems: Ripretinib and DP-5439 are substrates for P-glycoprotein (P-gp) and Breast Cancer Resistance Protein (BCRP). Ripretinib is an inhibitor of P-gp and BCRP. DP-5439 is an inhibitor of BCRP and Multidrug And Toxin Extrusion Protein 1 (MATE1).
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with ripretinib.
Ripretinib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay or clastogenic in either an in vitro human lymphocyte culture micronucleus assay or an in vivo rat bone marrow micronucleus assay.
Dedicated fertility studies in male animals were not conducted with ripretinib. Findings in male reproductive organs occurred in repeat-dose toxicity studies and included degeneration of the testes and cellular debris of the epididymis in males administered ≥30 mg/kg/day (approximately one half of the human exposure at the recommended dose of 150 mg).
14 CLINICAL STUDIES
The efficacy of QINLOCK was evaluated in INVICTUS, an international, multi-center, randomized (2:1), double-blind, placebo-controlled trial (NCT03353753). Eligible patients had unresectable, locally advanced or metastatic gastrointestinal stromal tumor (GIST) and had received prior treatment with imatinib, sunitinib, and regorafenib. Randomization was stratified by prior lines of therapy (3 versus ≥4) and Eastern Cooperative Oncology Group (ECOG) performance status (0 versus 1 or 2). Patients received QINLOCK 150 mg or placebo orally once daily until disease progression or unacceptable toxicity. Tumor response assessments were performed every 28 days through for the first 4 months and then every 56 days thereafter. The major efficacy outcome measure was progression-free survival (PFS) based on disease assessment by blinded independent central review (BICR) using modified RECIST 1.1 criteria, in which lymph nodes and bone lesions were not target lesions and a progressively growing new tumor nodule within a pre-existing tumor mass must meet specific criteria to be considered unequivocal evidence of progression. Additional efficacy outcome measures included objective response rate (ORR) by BICR and overall survival (OS). Patients randomized to receive placebo could be treated with QINLOCK at the time of disease progression.
A total of 129 patients were randomized, 85 to QINLOCK and 44 to placebo.
Patient characteristics of the intent-to-treat (ITT) population in INVICTUS were median age of 60 years (range: 29 to 83 years), with 39% aged ≥65 years; 57% were male; 75% were White; and 92% had an ECOG performance status of 0 or 1. Sixty-three percent (63%) of patients received 3 prior therapies and 37% received 4 or more prior therapies. Sixty-six percent (66%) of patients randomized to placebo switched to QINLOCK after disease progression.
Efficacy results from INVICTUS are summarized in Table 6.
|
BICR=Blinded Independent Central Review; CI=Confidence Interval |
||
|
a. Assessed per BICR. |
||
|
b. p-value is based on 2-sided stratified log-rank test. |
||
|
c. Hazard ratio is based on Cox proportional regression model. This model includes treatment and randomization stratification factors as fixed factors. |
||
|
d. Based on Fisher's exact test. The p-value is not statistically significant. |
||
|
e. Not evaluated for statistical significance as a result of the sequential testing procedure for the secondary endpoints of ORR and OS. |
||
| QINLOCK (N=85) | Placebo (N=44) |
|
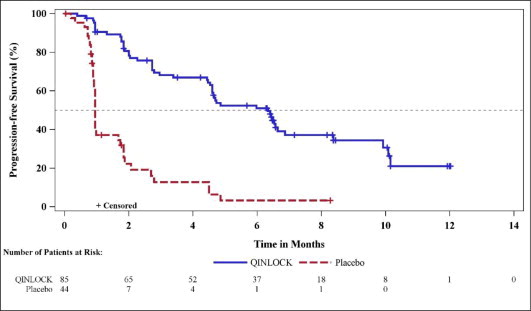
| Progression-Free Survivala | ||
| Number of events (%) | 51 (60) | 37 (84) |
| Progressive disease | 46 (54) | 32 (73) |
| Deaths | 5 (6) | 5 (11) |
| Median PFS (months) (95% CI) | 6.3 (4.6, 6.9) | 1.0 (0.9, 1.7) |
| Hazard ratio (95% CI)c | 0.15 (0.09, 0.25) | |
| p-valueb | < 0.0001 | |
| Overall Response Ratea | ||
| Overall Response Rate (%) | 9 | 0 |
| (95% CI) | (4.2, 18) | (0, 8) |
| p-valued | 0.0504 | |
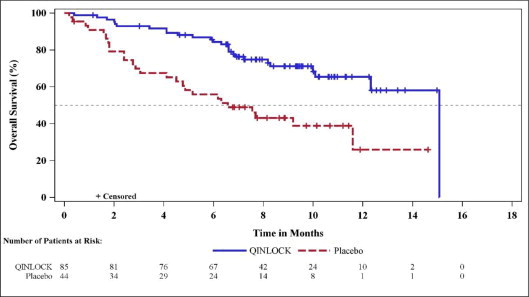
| Overall Survival e | ||
| Number of deaths (%) | 26 (31) | 26 (59) |
| Median OS (months) (95% CI) | 15.1 (12.3, 15.1) | 6.6 (4.1, 11.6) |
| Hazard ratio (95% CI)c | 0.36 (0.21, 0.62) | |
Figure 1: Kaplan-Meier Curve of Progression-Free Survival in INVICTUS
Figure 2: Kaplan-Meier Curve of Overall Survival in INVICTUS
16 HOW SUPPLIED/STORAGE AND HANDLING
QINLOCK 50 mg tablets are white to off-white, oval shaped, and debossed with “DC1” on one side.
- 90-count bottles NDC 73207-101-30
Dispense in original container only. Store in the original container with the desiccant to protect from moisture and light. Replace cap securely each time after opening. Do not discard desiccant.
Store at 20°C to 25°C (68°F to 77°F); excursion permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Palmar-Plantar Erythrodysesthesia Syndrome
Advise patients to contact their healthcare provider immediately if they experience severe skin changes [see Warnings and Precautions (5.1)].
New Primary Cutaneous Malignancies
Advise patients to contact their healthcare provider immediately for change in or development of new skin lesions [see Warnings and Precautions (5.2)].
Hypertension
Advise patients that hypertension may develop during treatment with QINLOCK and that blood pressure should be monitored regularly during treatment [see Warnings and Precautions (5.3)].
Cardiac Dysfunction
Advise patients that cardiac failure may develop during treatment with QINLOCK and that signs or symptoms of cardiac failure should be regularly monitored during treatment. Advise patients to contact their healthcare provider immediately for signs or symptoms of cardiac dysfunction [see Warnings and Precautions (5.4)].
Risk of Impaired Wound Healing
Advise patients that QINLOCK may impair wound healing. Advise patients to inform their healthcare provider of any planned surgical procedure [see Warnings and Precautions (5.5)].
Photosensitivity
Inform patients that there is a potential risk of photosensitivity reactions with QINLOCK. Advise patients to limit direct ultraviolet exposure by using sunscreen and protective clothing during treatment with QINLOCK [see Warnings and Precautions (5.6)].
Embryo-Fetal Toxicity
- Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.7), Use in Specific Populations (8.1)].
- Advise females of reproductive potential to use effective contraception during treatment with QINLOCK and for 1 week after the last dose [see Use in Specific Populations (8.3)].
- Advise males with female partners of reproductive potential to use effective contraception during treatment with QINLOCK and for 1 week after the last dose [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)].
Lactation
Advise females not to breastfeed during treatment with QINLOCK and for 1 week after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise males of reproductive potential that QINLOCK may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)].
Drug Interactions
Advise patients to inform their healthcare provider of all concomitant medications, including prescription medicines, over-the-counter drugs, vitamins, and herbal products [see Drug Interactions (7.1)].
Dosage and Administration
Instruct patients to take QINLOCK at the same time each day (once daily) with or without food. Advise patients to swallow tablets whole. Inform patients about what to do in the event they miss a dose or vomit after taking a dose of QINLOCK [see Dosage and Administration (2.1)].
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 10/2023 |
|
| Patient Information
QINLOCK® (kin-lok) (ripretinib) tablets |
||
| What is QINLOCK?
QINLOCK is a prescription medicine used to treat adults with advanced gastrointestinal stromal tumor (GIST) who have received prior treatment with 3 or more kinase inhibitor medicines, including imatinib, for their GIST. It is not known if QINLOCK is safe and effective in children. |
||
Before taking QINLOCK, tell your healthcare provider about all of your medical conditions, including if you:
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. QINLOCK and certain other medicines can affect each other causing side effects or affect how QINLOCK works. Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. |
||
| What should I avoid while taking QINLOCK?
Your skin may be sensitive to the sun or other forms of light (photosensitivity) during treatment with QINLOCK. Avoid or limit exposure to direct sunlight, sunlamps, and other sources of ultraviolet radiation during treatment and for at least 1 week after stopping treatment with QINLOCK. Use sunscreen and wear clothes that cover your skin if you need to be out in the sun. |
||
How should I take QINLOCK?
|
||
| What are the possible side effects of QINLOCK?
QINLOCK may cause serious side effects, including:
|
||
|
|
|
|
||
| The most common side effects of QINLOCK include: | ||
|
|
|
| QINLOCK may affect fertility in males which may affect the ability to have children. Talk to your healthcare provider if this is a concern for you. These are not all the possible side effects of QINLOCK. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store QINLOCK?
|
||
| Keep QINLOCK and all medicines out of the reach of children. | ||
| General information about the safe and effective use of QINLOCK.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use QINLOCK for a condition for which it was not prescribed. Do not give QINLOCK to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about QINLOCK that is written for health professionals. |
||
| What are the ingredients in QINLOCK?
Active ingredient: ripretinib Inactive ingredients: crospovidone, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and silicon dioxide Manufactured for: Deciphera Pharmaceuticals, LLC, 200 Smith Street, Waltham, MA 02451 QINLOCK is a trademark of Deciphera, LLC. All other trademarks referenced herein are the property of their respective owners. For more information, call 1-888-724-3274 or go to www.QINLOCK.com |
||