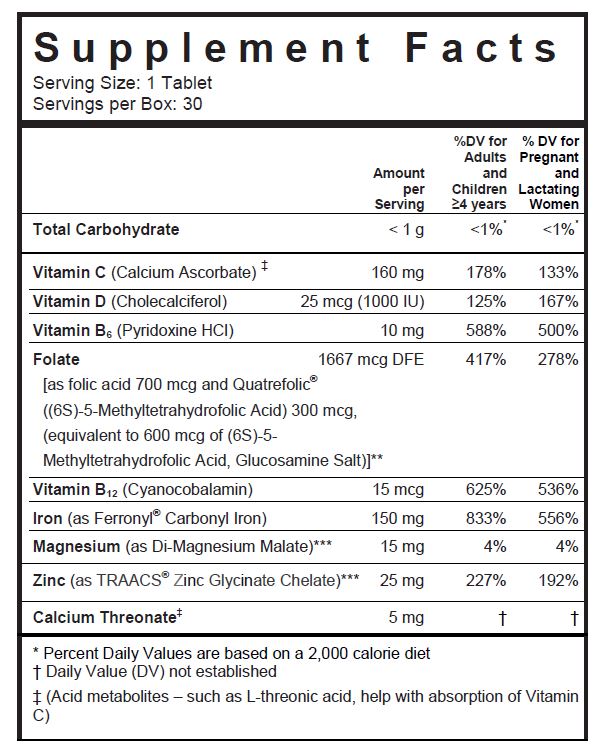
SUPPLEMENT FACTS
Other Ingredients: Microcrystalline Cellulose, di-Calcium Phosphate, Pregelatinized Starch, Red Color Coating (Hydroxypropylmethyl Cellulose, Polyvinyl Alcohol, FD&C Red #40 Lake, Glycerol Triacetate, Titanium Dioxide, Talc, FD&C Yellow #6 Lake, FD&C Blue #2 Lake), Croscarmellose Sodium, Magnesium Stearate, Silicon Dioxide and Fumed Silica.
VEGETARIAN FRIENDLY
GLUTEN, LACTOSE AND SUGAR FREE
CORVITE® FE is a professionally prescribed hematinic multivitamin/multimineral dietary supplement used to improve the nutritional status of patients with iron deficiency; this includes women prior to conception, throughout pregnancy, and in the postnatal period for both lactating and non-lactating mothers.
Other Ingredients: Microcrystalline Cellulose, DiCalcium Phosphate, Croscarmellose Sodium, Acacia, Isopropyl Alcohol, Povidone K30, Purified Water, Magnesium Stearate, TriPotassium Citrate, Citric Acid, Fumed Silica, Hypromellose, Ethyl Cellulose, Titanium Dioxide, Polyethylene Glycol, FD&C Red #40 Lake, Talc, Polysorbate 80, Hydroxypropyl Cellulose, Black Iron Oxide and Vegetable Oil.
CONTRAINDICATIONS
CORVITE®FE should not be used by patients with a known hypersensitivity to any of the listed ingredients. All iron compounds are contraindicated in patients with hemochromatosis, hemosiderosis, or hemolytic anemias.
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. KEEP THIS PRODUCT OUT OF REACH OF CHILDREN. In case of accidental overdose, call a doctor or poison control center immediately. |
PRECAUTIONS
General: Do not exceed recommended dose. The type of anemia and the underlying cause or causes should be determined before starting therapy with CORVITE® FE. Since the anemia may be a result of a systemic disturbance, such as recurrent blood loss, the underlying cause or causes should be corrected, if possible.
Folic Acid: Folic acid alone is an improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
The patient’s medical conditions and consumption of other drugs, herbs, and/or supplements should be considered.
Pediatric Use: Safety and effectiveness in pediatric population have not been established.
Geriatric Use: Safety and effectiveness in elderly population have not been established.
DRUG INTERACTIONS
CORVITE® FE is not recommended for and should not be given to patients receiving levodopa because the action of levodopa is antagonized by pyridoxine. There is a possibility of increased bleeding due to pyridoxine interaction with anticoagulants (e.g., Aspirin, Heparin, Clopidogrel).
ADVERSE REACTIONS
Adverse reactions with iron therapy may include constipation, diarrhea, nausea, vomiting, dark stools and abdominal pain which are usually transient.
OVERDOSAGE
The clinical course of acute iron overdosage can be variable. Initial symptoms may include abdominal pain, nausea, vomiting, diarrhea, tarry stools, melena, hematemesis, hypotension, tachycardia, metabolic acidosis, hyperglycemia, dehydration, drowsiness, pallor, cyanosis, lassitude, seizures, shock and coma.
STORAGE
Store at controlled room temperature 15°-30°C (59°-86°F) [See USP]. Protect from light, moisture and avoid excessive heat. Dispense in a tight, light resistant container as defined by the USP using a child-resistant closure.
KEEP OUT OF REACH OF CHILDREN.
** Quatrefolic® is a registered trademark of Gnosis, SpA.
U.S. Patent No. 7,947,662.
***TRAACS® is a registered trademark of Albion International, Inc.
Malate covered by U.S. Patent No, 6,706,904;
Chelate covered by U.S. Patent No. 7,838,042.
Call your doctor about side effects. To report side effects, call Vertical Pharmaceuticals at 1-770-509-4500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
PLR-CFE-00003-1 Rev. 12/2021
Distributed by:
Vertical Pharmaceuticals, LLC
Alpharetta, GA 30005
www.verticalpharma.com

