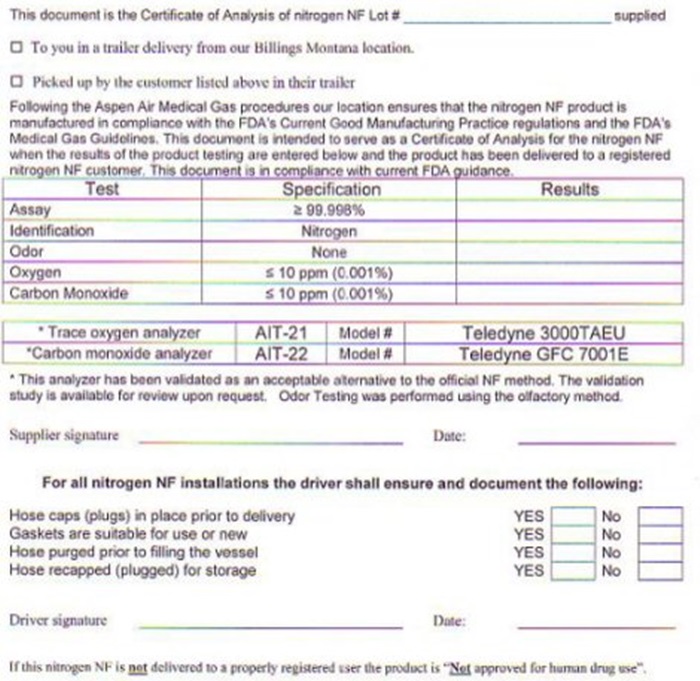
NITROGEN CERTIFICATE OF ANALYSIS
Customer
_______ Aspen Air
________ 1524 Lockwood Road
________ Billings, MT
This document is the Certificate of Analysis of nitrogen NF Lot # ______________ supplied
___ To you is a trailer delivery from our Billings Montana location.
___ Picked up by the customer listed above in their trailer
Following the Aspen Air Medical Gas procedures our location ensures that the nitrogen NF product is manufactured in compliance with the FDA’s Current Good Manufacturing Practice regulations and the FDA’s Medical Gas Guidelines. This document is intended to serve as a Certificate of Analysis for the nitrogen NF when the results of the product testing are entered below and the product has been delivered to a registered nitrogen NF customer. This document is in compliance with current FDA guidance.
Test Specification Results
Assay ≥ 99.998% ____
Identification Nitrogen ____
Odor None ____
Oxygen ≤ 10 ppm (0.001%) ____
Carbon Monoxide ≤ 10 ppm (0.001%) ____
*Trace oxygen analyzer AIT-21 Model# Teledyne 3000TAEU
*Carbon monoxide analyzer AIT-22 Model# Teledyne GFC 7001E
*This analyzer has been validated as an acceptable alternative to the official NF method. The validation study is available for review upon request. Odor Testing was performed using the olfactory method.
Supplier signature: __________________________ Date: ____________
For all nitrogen NF installations, the driver shall ensure and document the following:
Hose caps (plugs) in place prior to delivery Yes ___ No ___
Gaskets are suitable for use or new Yes ___ No ___
Hose purged prior to filling the vessel Yes ___ No ___
Hose recapped (plugged) for storage Yes ___ No ___
Driver signature: ___________________ Date: ________________
If this nitrogen NF is not delivered to a properly registered user the product is “Not approved for human drug use”.