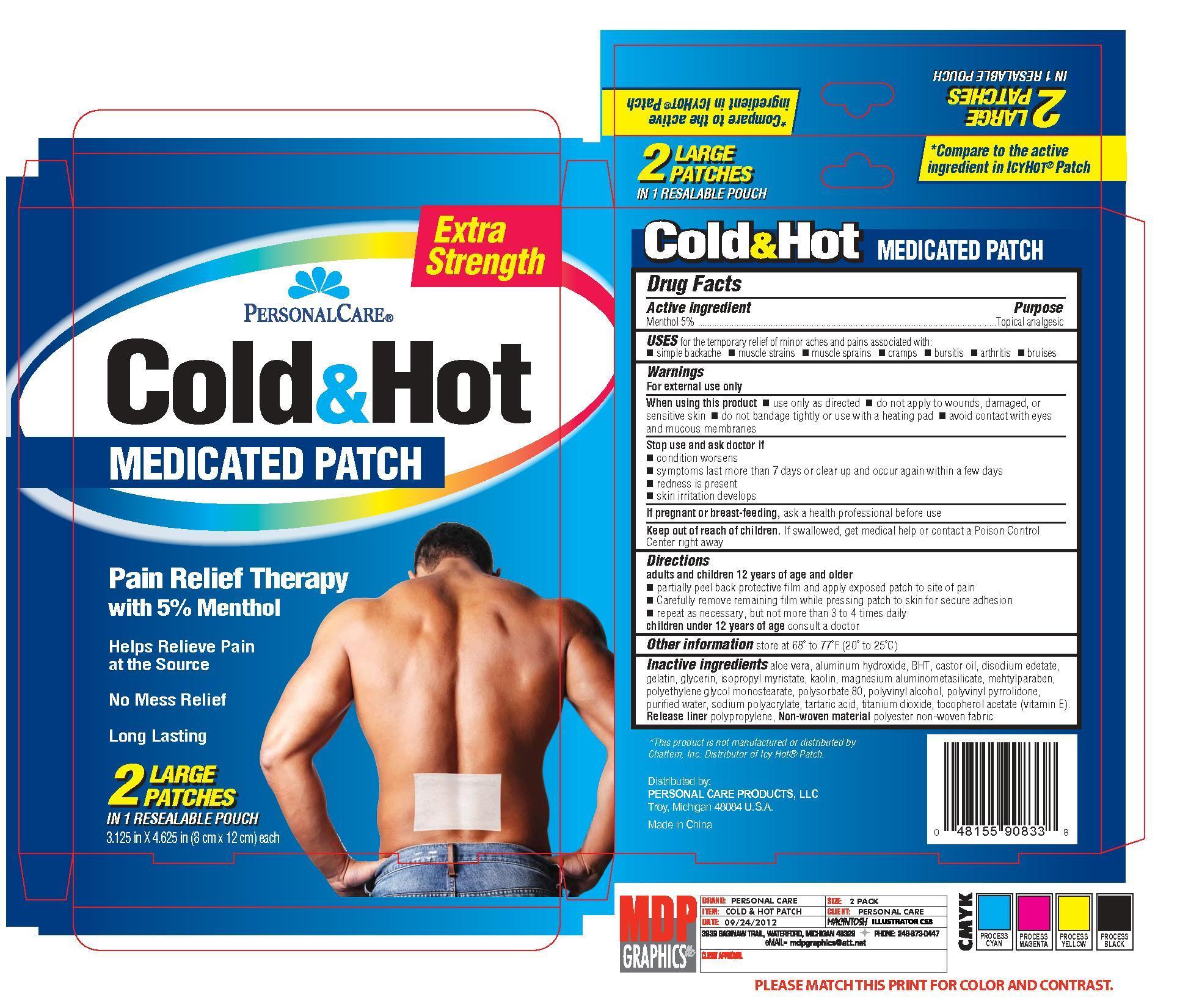
Active ingredient Purpose
Menthol 5% ..........................................................................Topical analgesic
Uses for the temporary relief of minor aches and pains associated with:
- simple backache
- muscle strains
- muscle sprains
- cramps
- bursitis
- arthritis
- bruises
When using this product
- use only as directed
- do not apply to wounds, damaged, or sensitive skin
- do not bandage tightly or use with a heating pad
- avoid contact with eyes and mucous membranes
Stop use and ask doctor if
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
- redness is present
- skin irritation develops
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Directions
adults and children 12 years of age and older
- partially peel back protective film and apply exposed patch to site of pain
- carefully remove remaining film while pressing patch to skin for secure adhesion
- repeat as necessary, but not more than 3 to 4 times daily
Inactive ingredients
aloe vera, aluminum hydroxide, BHT, castor oil, disodium edetate, gelatin, glycerin, isopropyl myristate, kaolin, magnesium aluminometasilicate, methylparaben, polyethylene glycol monsterarate, polysorbate 80,
polyvinyl alcohol, polyvinyl pyrrolidone, purified water, sodium polyacrylate, tartaric acid, titanium dioxide, tocopherol acetate (Vitamin E).
 Enter section text here
Enter section text here