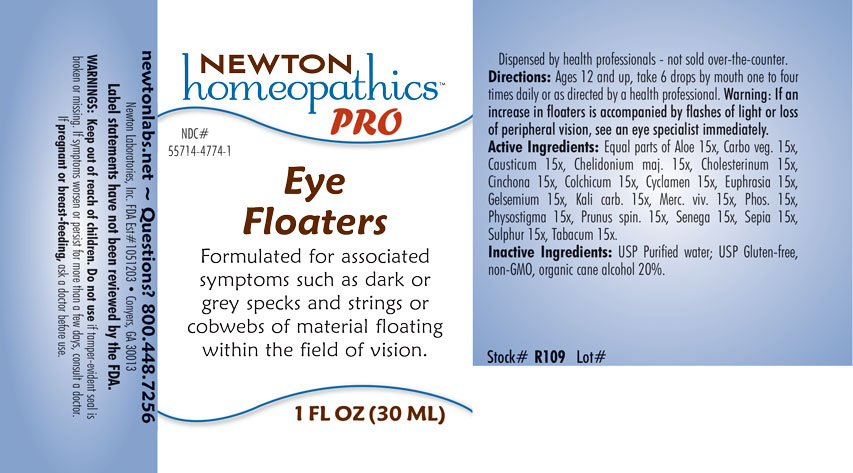
INDICATIONS & USAGE SECTION
Formulated for associated symptoms such as dark or grey specks and strings or cobwebs of material floating within the field of vision.
DOSAGE & ADMINISTRATION SECTION
Directions: Ages 12 and up, take 6 drops by mouth one to four times daily or as directed by a health professional. Warning: If an increase in floaters is accompanied by flashes of light or loss of peripheral vision, see and eye specialist immediately.
OTC - ACTIVE INGREDIENT SECTION
Equal parts of Aloe 15x, Carbo vegetabilis 15x, Causticum 15x, Chelidonium majus 15x, Cholesterinum 15x, Cinchona officinalis 15x, Colchicum autumnale 15x, Cyclamen europaeum 15x, Euphrasia, Gelsemium sempervirens 15x, Kali carbonicum 15x, Mercurius vivus 15x, Phosphorus 15x, Physostigma venenosum 15x, Prunus spinosa 15x, Senega officinalis 15x, Sepia 15x, Sulphur 15x, Tabacum 15x.
OTC - PURPOSE SECTION
Formulated for associated symptoms such as dark or grey specks and strings or cobwebs of material floating within the field of vision.
INACTIVE INGREDIENT SECTION
Inactive Ingredients: USP Purified Water; USP Gluten-free, non-GMO, organic cane alcohol 20%.
QUESTIONS SECTION
newtonlabs.net - Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 Conyers, GA 30013