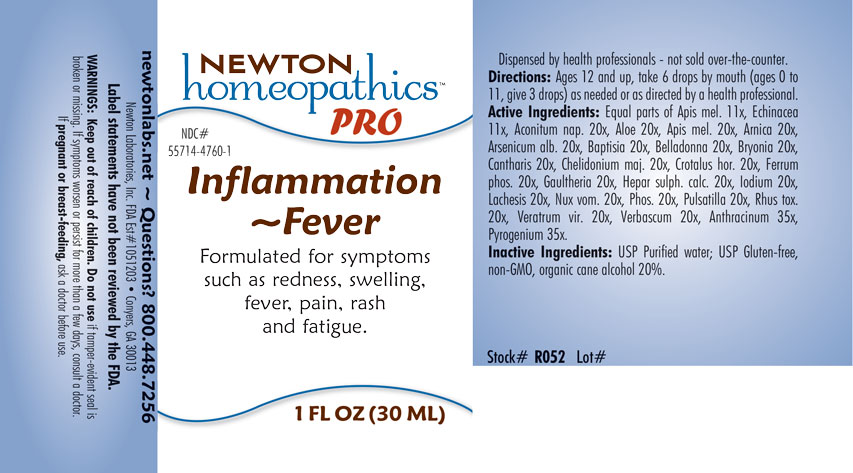
INDICATIONS & USAGE SECTION
Formulated for symptoms such as redness, swelling, fever, pain, rash and fatigue.
DOSAGE & ADMINISTRATION SECTION
Directions: Ages 12 and up, take 6 drops by mouth (ages 0 to 11, give 3 drops) as needed or as directed by a health professional.
OTC - ACTIVE INGREDIENT SECTION
Equal parts of Apis mellifica 11x, Echinacea 11x, Aconitum napellus 20x, Aloe 20x, Apis mellifica 20x, Arnica montana 20x, Arsenicum album 20x, Baptisia tinctoria 20x, Belladonna 20x, Bryonia 20x, Cantharis 20x, Chelidonium majus 20x, Crotalus horridus 20x, Ferrum phosphoricum 20x, Gaultheria procumbens 20x, Hepar sulphuris calcareum 20x, Iodium 20x, Lachesis mutus 20x, Nux vomica 20x, Phosphorus 20x, Pulsatilla 20x, Rhus toxicodendron 20x, Veratrum viride 20x, Verbascum thapsus 20x, Anthracinum 35x, Pyrogenium 35x.
OTC - PURPOSE SECTION
Formulated for symptoms such as redness, swelling, fever, pain, rash and fatigue.
INACTIVE INGREDIENT SECTION
Inactive Ingredients: USP Purified Water; USP Gluten-free, non-GMO, organic cane alcohol 20%.
QUESTIONS SECTION
newtonlabs.net – Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013