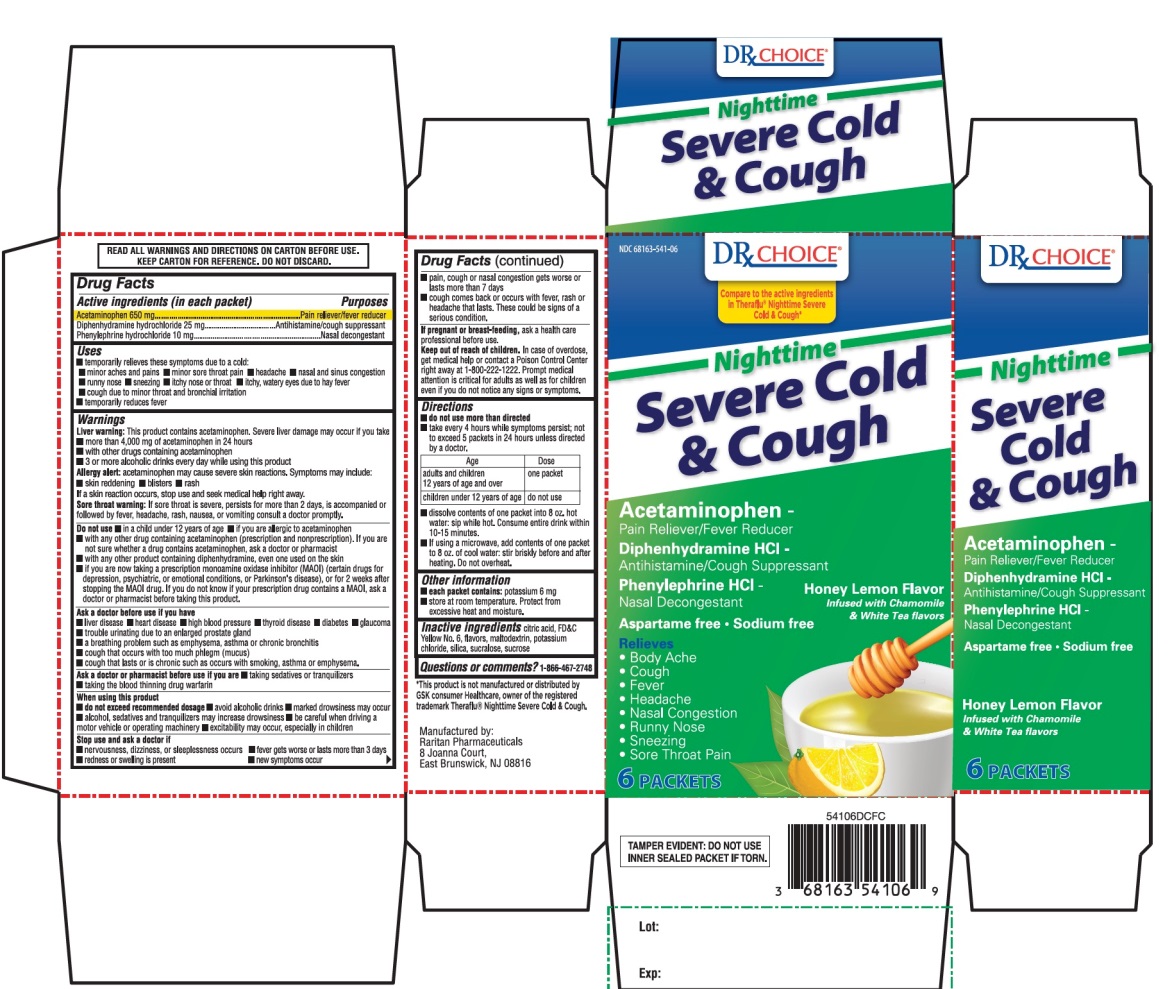
Active ingredients (in each packet)
Acetaminophen 650 mg
Diphenhydramine hydrochloride 25 mg
Phenylephrine hydrochloride 10 mg
Uses
- ▪
- temporarily relieves these symptoms due to a cold:
- ▪
- minor aches and pains
- ▪
- minor sore throat pain
- ▪
- headache
- ▪
- nasal and sinus congestion
- ▪
- runny nose
- ▪
- sneezing
- ▪
- itchy nose or throat
- ▪
- itchy, watery eyes due to hay fever
- ▪
- cough due to minor throat and bronchial irritation
- ▪
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- ▪
- more than 4,000 mg of acetaminophen in 24 hours
- ▪
- with other drugs containing acetaminophen
- ▪
- 3 or more alcoholic drinks every day while using this product
Allergy alert: acetaminophen may cause severe skin reactions, Symptoms may include:
- ▪
- skin reddening
- ▪
- blisters
- ▪
- rash
If a skin reaction occurs, stop use and seek medical help right away
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting consult a doctor promptly.
Do not use
- ▪
- in a child under 12 years of age
- ▪
- if you are allergic to acetaminophen
- ▪
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or a pharmacist.
- ▪
- with any other product containing diphenhydramine, even one used on the skin
- ▪
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains a MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- ▪
- liver disease
- ▪
- heart disease
- ▪
- high blood pressure
- ▪
- thyroid disease
- ▪
- diabetes
- ▪
- glaucoma
- ▪
- trouble urinating due to an enlarged prostate gland
- ▪
- a breathing problem such as emphysema, asthma or chronic bronchitis
- ▪
- cough that occurs with too much phlegm (mucus)
- ▪
- cough that lasts or is chronic such as occurs with smoking, asthma or emphysema
Ask a doctor or pharmacist before use if you are
- ▪
- taking sedatives or tranquilizers
- ▪
- taking the blood thinning drug warfarin
When using this product
- ▪
- do not exceed recommended dosage
- ▪
- avoid alcoholic drinks
- ▪
- marked drowsiness may occur
- ▪
- alcohol, sedatives and tranquilizers may increase drowsiness
- ▪
- be careful when driving a motor vehicle or operating machinery
- ▪
- excitability may occur, especially in children
Stop use and ask a doctor if
- ▪
- nervousness, dizziness, or sleeplessness occurs
- ▪
- fever gets worse or lasts more than 3 days
- ▪
- redness or swelling is present
- ▪
- new symptoms occur
- ▪
- pain, cough or nasal congestion gets worse or lasts more than 7 days
- ▪
- cough comes back or occurs with fever, rash or headache that lasts. There could be signs of a serious condition.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222. Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
- ▪
- do not use more than directed
- ▪
- take every 4 hours while symptoms persist, not to exceed 5 packets in 24 hours unless directed by a doctor
|
Age |
Dose |
|
adults and children 12 years of age and over |
one packet |
|
children under 12 years of age |
do not use |
- ▪
- dissolve contents of one packet into 8 oz. hot water: sip while hot. Consume entire drink within 10 - 15 minutes.
- ▪
- if using a microwave, add contents of one packet to 8 oz. of cool water: stir briskly before and after heating, Do not overheat.
Other information
- ▪
- each packet contains: potassium 6 mg
- ▪
- store at room temperature. Protect from excessive heat and moisture.
Inactive ingredients
citric acid, FD& C Yellow No. 6, flavors, maltodextrin, potassium chloride, silica, sucralose, sucrose,
Principal Display Panels
*Compare to the active ingredients in Theraflu® Nighttime Severe Cold & Cough*
NDC# 68163-541-06
Nighttime
Severe Cold & Cough
ACETAMINOPHEN
PAIN RELIEVER/FEVER REDUER
DIPHENHYDRAMINE HCl
ANTIHISTAMINE/ COUGH SUPPRESSANT
PHENYLEPHRINE HCl
NASAL DECONGESTANT
- •
- Aspartame free
- •
- Sodium Free
Relieves:
Body Ache & Fever
- •
- Cough
- •
- Fever
- •
- Headache
- •
- Nasal Congestion
- •
- Runny Nose
- •
- Sneezing
- •
- Sore Throat Pain
6 PACKETS
Honey Lemon Flavor
Infused with Chamomile & White Tea Flavors
|
TAMPER EVIDENT: DO NOT USE IF INNER SEALED PACKET IS TORN |
Manufactured by:
Raritan Pharmaceuticals
8 Joanna Court
East Brunswick, NJ 08816
*This product is not manufactured or distributed by GSK consumer Healthcare, owner of the registered trademark THERAFLU® Nighttime Severe Cold & Cough.