WARNINGS
IF ANY OF THESE CONDITIONS PERSIST, CONSULT A DOCTOR
- DIZZINESS
- DIARHEA
- ABDOMINAL CRAMPS
- VOMITING
- NAUSEA
- HEADACHE
IF PREGNANT OR IF YOU HAVE LIVER DISEASE, ASK A DOCTOR BEFORE USE. CONSULT YOUR DOCTOR IF AN ALLERGIC REACTION OCCURS.
KEEP OUT OF REACH OF CHILDREN. IN CASE OF ACCIDENTAL OVERDOSE, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
OTHER INFORMATION
- PHENYLKETONURICS: CONTAIN PHENYLALANINE 11.8 MG PER TABLET
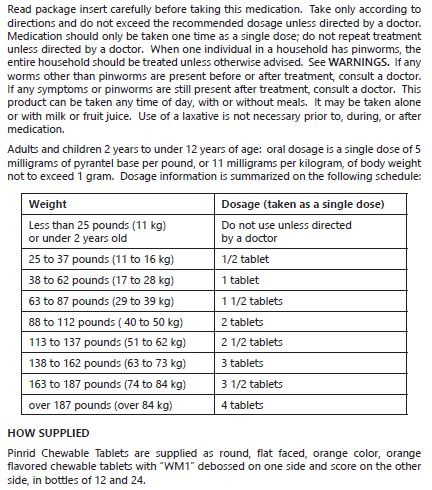
- READ PACKAGE INSERT CAREFULLY BEFORE USING
- IF SIGNS OF PINWORMS PERSIST, SEE A DOCTOR
- STORE AT 15° - 30°c (59° - 86°f)