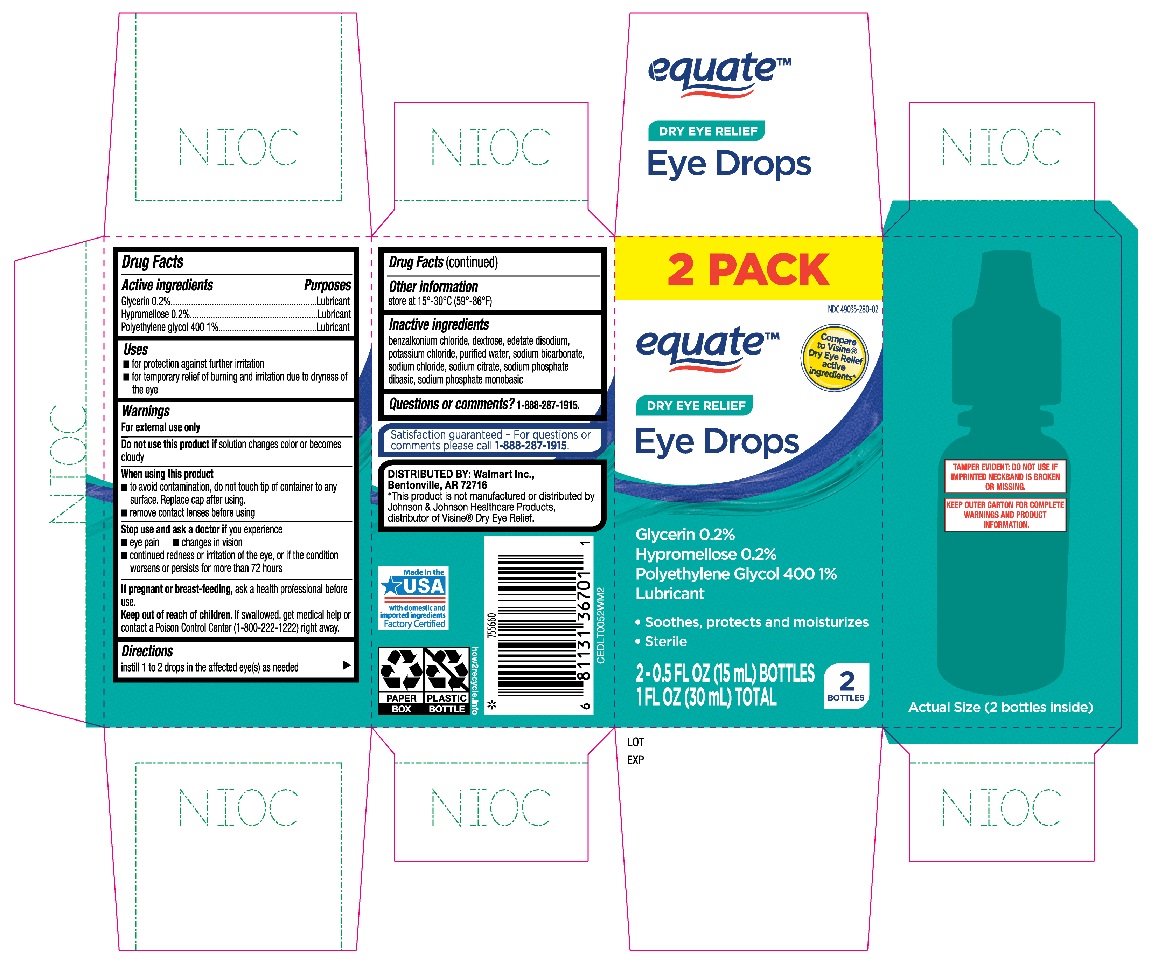
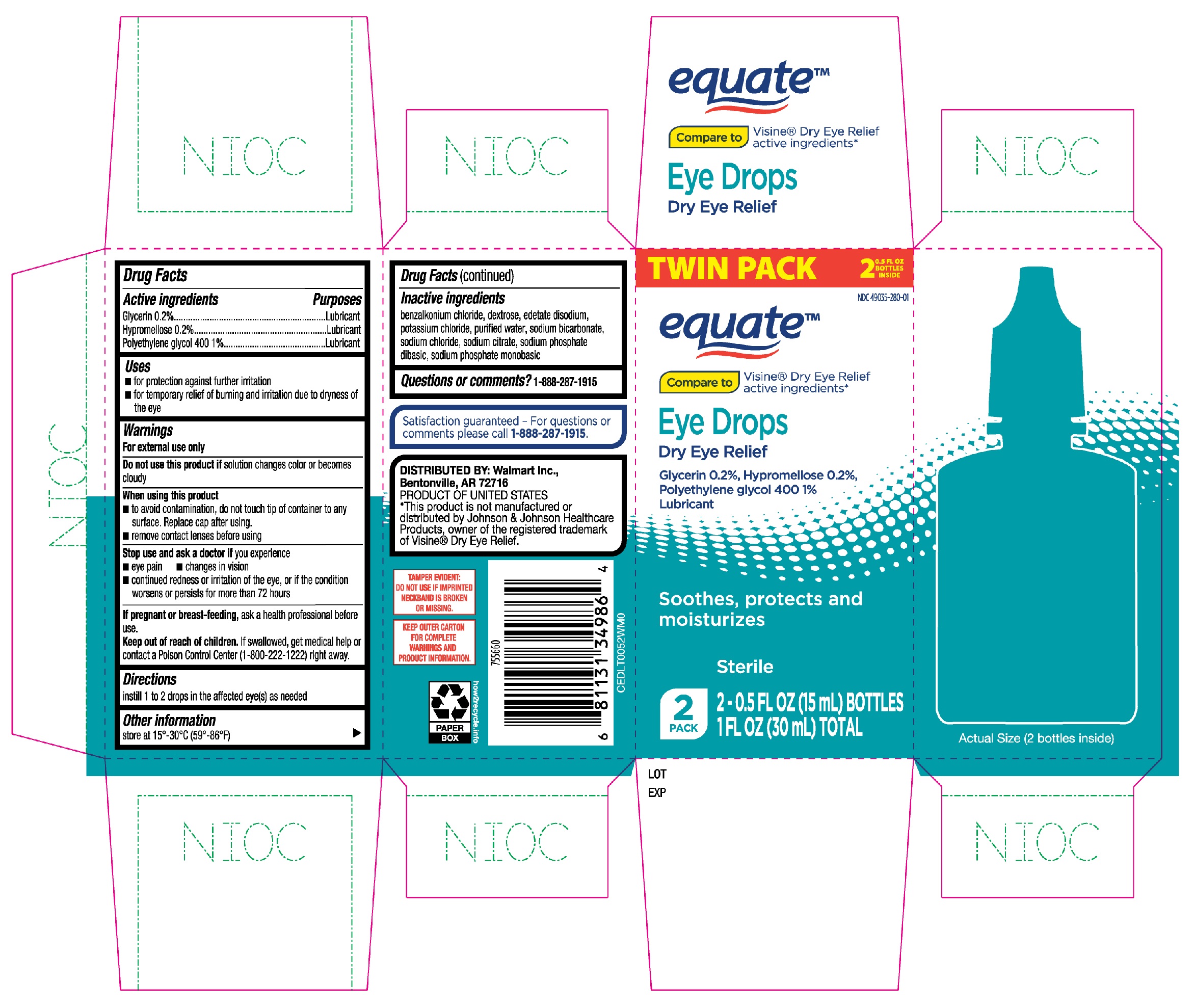
EQUATE EYE DROPS DRY EYE RELIEF- glycerin, hypromellose, polyethylene glycol 400 solution/ drops
Wal-Mart Stores, Inc.
----------
Active ingredients
Glycerin 0.2%
Hypromellose 0.2%
Polyethylene glycol 400 1%
Purposes
Lubricant
Lubricant
Lubricant
Uses
- for protection against further irritation
- for temporary relief of burning and irritation due to dryness of the eye
Warnings
For external use only
Do not use this product if
solution changes color or becomes cloudy
When using this product
- to avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- remove contact lenses before using
Stop use and ask a doctor if you experience
- eye pain
- changes in vision
- continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
instill 1 to 2 drops in the affected eye(s) as needed
Other information
store at 15°-30°C (59°-86°F)
Inactive ingredients
benzalkonium chloride, dextrose, edetate disodium, potassium chloride, purified water, sodium bicarbonate, sodium chloride, sodium citrate, sodium phosphate dibasic, sodium phosphate monobasic
Questions or comments?
1-888-287-1915
Equate Dry Eye Relief Eye Drops 2-0.5oz bottles
Equate Eye Drops Dry Eye Relief 2-0.5oz bottles
Wal-Mart Stores, Inc.

