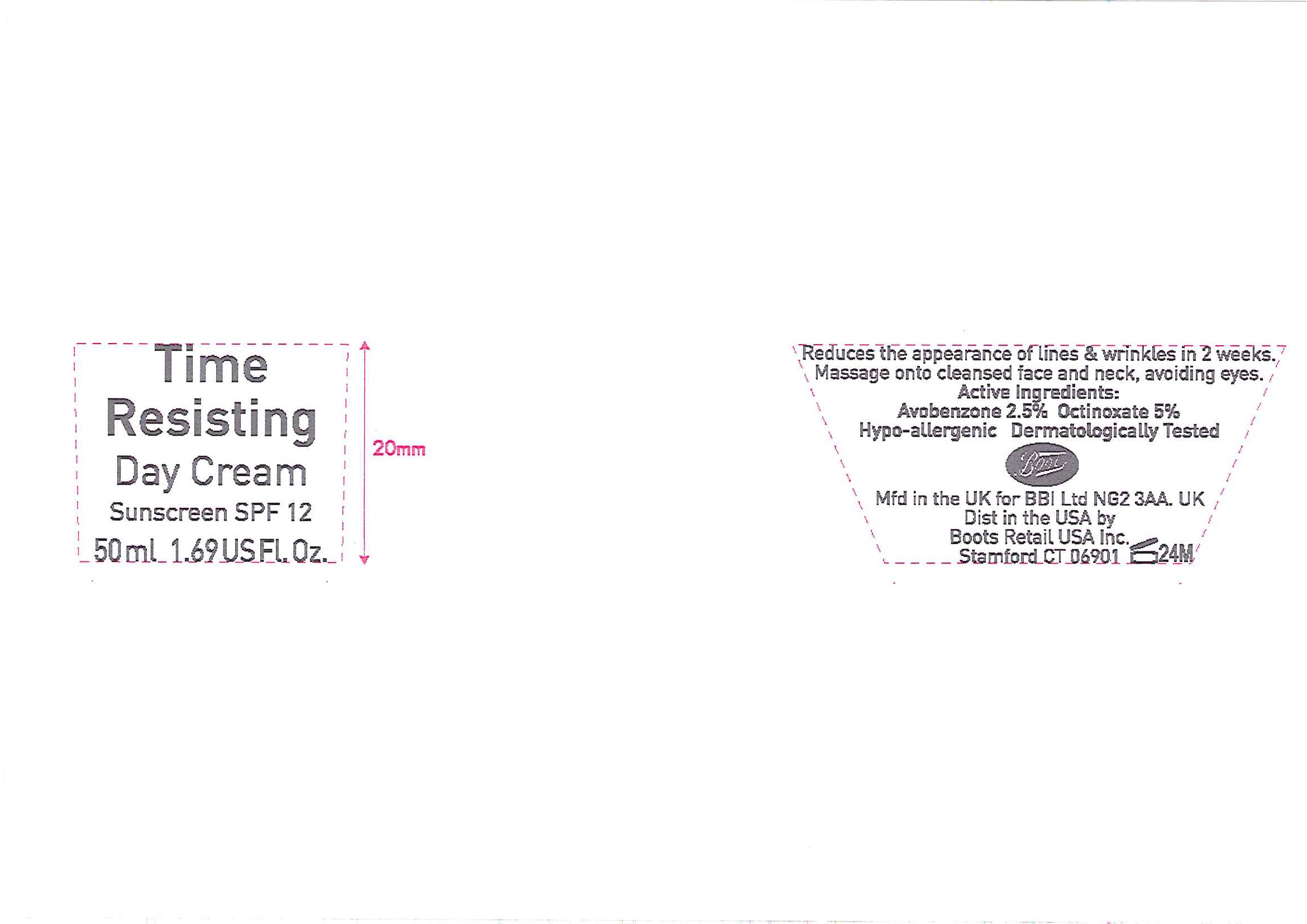
No7 Time Resisting Day Cream Sunscreen SPF 12
Smooth your cares away ...
Rewind time and reveal younger looking skin with this velvety soft day cream. In just 2 weeks it reduces the appearance of fine lines and wrinkles, replenishing lost moisture to leave your skin soft, supple and radiant.
Dermatologically Tested
www.boots.com
- Provides moderate protection against adverse effects of the sun
- Higher SPF gives more protection
Warnings
For external use only
When using this product
Avoid contact with eyes. If contact occurs rinse immediately with cold water.
Inactive ingredients
Aqua (Water), Butylene glycol, C12-15 alkyl benzoate, Sorbitan stearate, Dimethicone, Isohexadecane, Cetyl alcohol, Prunus amygdalus dulcis (Sweet almond oil), Mica, Phenoxyethanol, Polyacrylamide, Glyceryl stearate, C18-36 acid glycol ester, Tocopheryl acetate, PEG-100 stearate, C13-14 isoparaffin, Parfum (Fragrance), Dimethiconol, Sucrose cocoate, Methylparaben, Sodium ascorbyl phosphate, Sodium stearoyl lactylate, Carbomer, Propylparaben, Laureth-7, Tetrasodium EDTA, Potassium hydroxide, Propylene glycol, Glycerin, Xanthan gum, Retinyl palmitate, Panax ginseng root extract, BHT, Morus alba leaf extract, Sodium hydroxide, Tocopherol, CI 77891 (Titanium dixoide).
Reduces the appearance of lines and wrinkles in 2 weeks.
Massage onto cleansed face and neck, avoiding eyes.
Hypo-allergenic.
Dermatologically Tested
Boots
Mfd in the UK for BBI Ltd NG2 3AA. UK
Dist in the USA by Boots Retail USA Inc. Stamford CT 06901