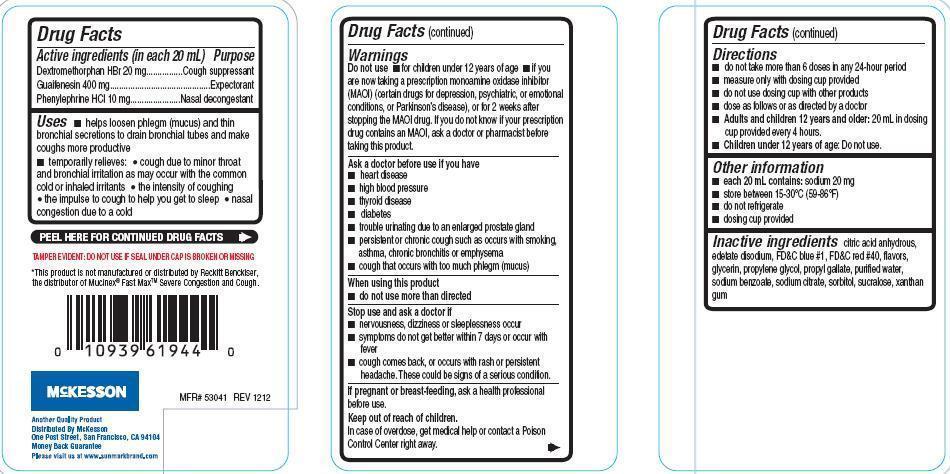
Active ingredients (in each 20 mL)
Dextromethorphan HBr 20 mg
Guaifenesin 400 mg
Phenylephrine HCl 10 mg
Uses
- hepls loosen phelgm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help you get to sleep
- nasal congestion due to a cold
Warnings
Do not use
- for children under 12 years of age
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
- cough that occurs with too much phlegm (mucus)
Directions
- do not take more than 6 doses in 24-hour period
- measure only with dosing coup provided
- do not use dosing cup with other products
- dose as follows or as directed by a doctor
- Adults and children 12 years and older: 20 mL in dosing cup provided every 4 hours
- Children under 12 years of age: Do not use
Other information
- each 20 mL contains: sodium 20 mg
- store between 15-30oC(59-86oF)
- do not refrigerate
- dosing cup provided
Inactive ingredients
citric acid anhydrous, edetate disodium, FDand C blue 1, FD and C red 40, flavors, glycerin, propylene glycol, propyl gallate, purified water, sodium benzoate, sodium citrate, sorbitol, sucralose, xanthan gum
Principal Display Panel
COMPARE TO MUCINEX FAST-MAX SEVERE CONGESTION AND COUGH ACTIVE INGREDIENTS
adult severe congestion and cough
multi-symptom
Relieves nasal and chest congestion,
Soothes Cough,
Thins and loosens mucus
DEXTROMETHORPHAN HBr
Cough suppressant
GUAIFENESIN
Expectorent
PHENYLEPHRINE HCl
Nasal decongestant,
For Ages 12 and Over
MAXIMUM STRENGTH
6 fl oz (177 mL)