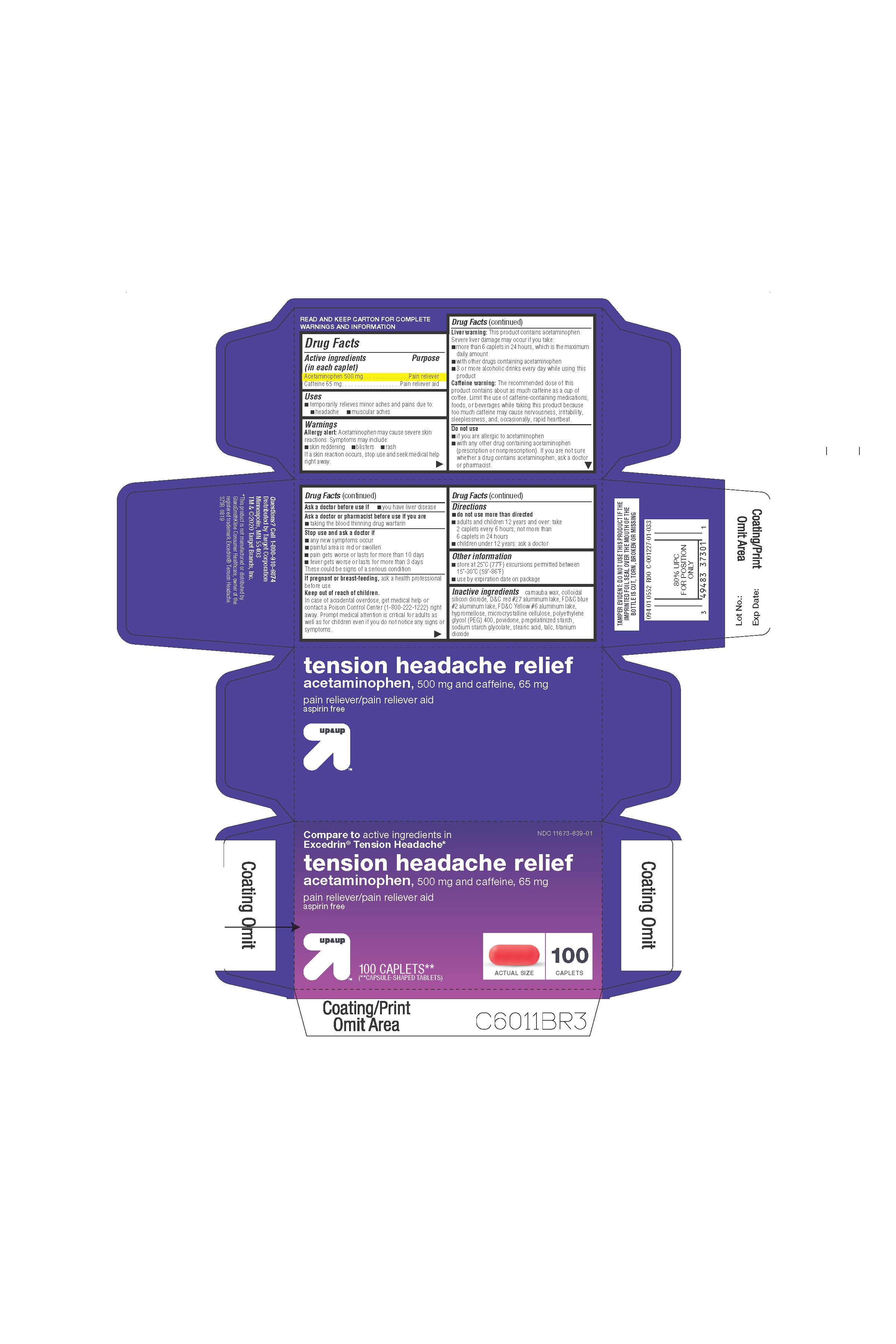
colloidal silicon dioxide, pregelatinized starch, D&C red no. 27 aluminum lake, FD&C blue no. 2 aluminum lake, FD&C yellow no. 6 aluminum lake, hypromellose, microcrystalline cellulose, polyethylene glycol (PEG 400), povidone stearic acid,talc, titanium dioxide
DIRECTIONS
•
do not use more than directed
•
adults and children 12 years and over: take 2 caplets every 6 hours; not more than 6 caplets in 24 hours
•
children under 12 years: ask a doctor
Keep out of reach of children.
In case of accidental overdose, get medical help or contact a Poison Control Center right away (1-800-222-1221). Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
WARNINGS
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
•
skin reddening
•
blisters
•
rash
If a skin reaction occurs, stop use and seek medical help right away.
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
•
more than 6 caplets in 24 hours, which is the maximum daily amount
•
with other drugs containing acetaminophen
•
3 or more alcoholic drinks every day while using this product
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and occasionally, rapid heartbeat.