FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Tekturna HCT is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including hydrochlorothiazide (HCTZ). There are no controlled trials demonstrating risk reduction with Tekturna HCT.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than 1 drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality have also been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (e.g., patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Add-On Therapy
A patient whose blood pressure is not adequately controlled with aliskiren alone or HCTZ alone may be switched to combination therapy with Tekturna HCT.
A patient whose blood pressure is controlled with HCTZ alone but who experiences hypokalemia may be switched to combination therapy with Tekturna HCT.
A patient who experiences dose-limiting adverse reactions on either component alone may be switched to Tekturna HCT containing a lower dose of that component in combination with the other to achieve similar blood pressure reductions.
Initial Therapy
Tekturna HCT may be used as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals.
The choice of Tekturna HCT as initial therapy should be based on an assessment of potential benefits and risks. Patients with Stage 2 hypertension are at a relatively high risk for cardiovascular events (such as strokes, heart attacks, and heart failure), kidney failure, and vision problems, so prompt treatment is clinically relevant. The decision to use a combination as initial therapy should be individualized and should be shaped by considerations such as baseline blood pressure, the target goal, and the incremental likelihood of achieving goal with a combination compared to monotherapy. Individual blood pressure goals may vary based upon the patient's risk.
Data from the high-dose multifactorial study
[see Clinical Studies (
14)]
provide estimates of the probability of reaching a target blood pressure with Tekturna HCT compared to aliskiren or HCTZ monotherapy.
Figures 1–
4 provide estimates of the likelihood of achieving systolic or diastolic blood pressure control with Tekturna HCT 300/25 mg, based upon baseline systolic or diastolic blood pressure. The curve of each treatment group was estimated by logistic regression modeling. The estimated likelihood at the right tail of each curve is less reliable because of small numbers of subjects with high baseline blood pressures.
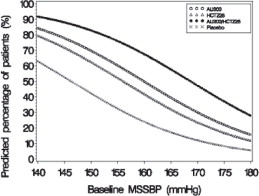
Figure 1: Probability of Achieving Systolic Blood Pressure (SBP) Less Than 140 mmHg
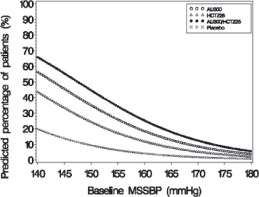
Figure 2: Probability of Achieving Systolic Blood Pressure (SBP) Less Than 130 mmHg
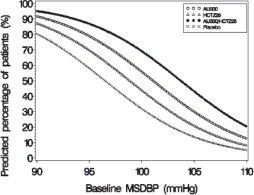
Figure 3: Probability of Achieving Diastolic Blood Pressure (DBP) Less Than 90 mmHg
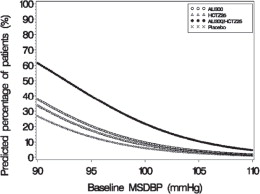
Figure 4: Probability of Achieving Diastolic Blood Pressure (DBP) Less Than 80 mmHg
At all levels of baseline blood pressure, the probability of achieving any given diastolic or systolic goal is greater with the combination than for either monotherapy. For example, the mean baseline msSBP/msDBP for patients participating in this multifactorial study was 154/99 mmHg. A patient with a baseline blood pressure of 154/99 mmHg has about a 62% chance of achieving a goal of less than 140 mmHg (systolic) and 61% chance of achieving less than 90 mmHg (diastolic) on aliskiren alone, and the chance of achieving these goals on HCTZ alone is about 54% (systolic) and 49% (diastolic). The chance of achieving these goals on Tekturna HCT rises to about 77% (systolic) and 74% (diastolic). The chance of achieving these goals on placebo is about 34% (systolic) and 37% (diastolic) [see Dosage and Administration ( 2) and Clinical Studies ( 14)].
2 DOSAGE AND ADMINISTRATION
2.1 Dose Selection
The recommended once-daily doses of Tekturna HCT in order of increasing mean effect are 150/12.5 mg, 150/25 mg or 300/12.5 mg, and 300/25 mg.
2.2 Dose Titration
The antihypertensive effect of Tekturna HCT is largely manifested within 1 week, with maximal effects generally seen at around 4 weeks. If blood pressure remains uncontrolled after 2 to 4 weeks of therapy, the dose may be titrated up to a maximum of aliskiren 300 mg/HCTZ 25 mg.
2.3 Add-On Therapy
A patient whose blood pressure is not adequately controlled with aliskiren alone or hydrochlorothiazide alone may be switched to combination therapy with Tekturna HCT. The usual recommended starting dose is 150/12.5 mg once daily as needed to control blood pressure. The dose may be titrated up to a maximum of aliskiren 300 mg/hydrochlorothiazide 25 mg once daily.
2.5 Initial Therapy
The usual recommended starting dose is 150/12.5 mg once daily as needed to control blood pressure. The dose may be titrated up to a maximum of aliskiren 300 mg/HCTZ 25 mg once daily.
Tekturna HCT is not recommended for use as initial therapy in patients with intravascular volume depletion [ see Warnings and Precautions ( 5.4) ].
2.6 Relationship to Meals
Patients should establish a routine pattern for taking Tekturna HCT with regard to meals. High-fat meals decrease absorption substantially [see Clinical Pharmacology ( 12.3)].
3 DOSAGE FORMS AND STRENGTHS
- 150 mg/12.5 mg tablets: white, biconvex ovaloid, film-coated tablets imprinted with NVR/LCI
- 150 mg/25 mg tablets: pale yellow, biconvex ovaloid, film-coated tablets imprinted with NVR/CLL
- 300 mg/12.5 mg tablets: violet white, biconvex ovaloid, film-coated tablets imprinted with NVR/CVI
- 300 mg/25 mg tablets: light yellow, biconvex ovaloid, film-coated tablets imprinted with NVR/CVV
4 CONTRAINDICATIONS
Do not use aliskiren with ARBs or ACEIs in patients with diabetes [see Warnings and Precautions ( 5.2) and Clinical Studies ( 14.4)].
Tekturna HCT is contraindicated in patients with known anuria or hypersensitivity to sulfonamide-derived drugs like HCTZ or to any of the components [see Warnings and Precautions ( 5.6) and Adverse Reactions ( 6.1)] . Hypersensitivity reactions may range from urticaria to anaphylaxis.
5 WARNINGS AND PRECAUTIONS
5.1 Fetal Toxicity
Aliskiren
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue Tekturna HCT as soon as possible [see Use in Specific Populations ( 8.1)].
Hydrochlorothiazide
Thiazides cross the placenta, and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possible other adverse reactions that have occurred in adults [see Use in Specific Populations ( 8.1)].
5.2 Renal Impairment/Hyperkalemia/Hypotension when Tekturna HCT is Given in Combination with ARBs or ACEIs
Tekturna HCT is contraindicated in patients with diabetes who are receiving ARBs or ACEIs because of the increased risk of renal impairment, hyperkalemia, and hypotension. In general, avoid combined use of aliskiren with ACE inhibitors or ARBs, particularly in patients with creatinine clearance (CrCl) less than 60 mL/min [see Contraindications ( 4), Drug Interactions ( 7) and Clinical Studies ( 14.3)] .
5.3 Anaphylactic Reactions and Head and Neck Angioedema
Aliskiren
Hypersensitivity reactions such as anaphylactic reactions and angioedema of the face, extremities, lips, tongue, glottis and/or larynx have been reported in patients treated with Tekturna and has necessitated hospitalization and intubation. This may occur at any time during treatment and has occurred in patients with and without a history of angioedema with ACEIs or angiotensin receptor antagonists. Anaphylactic reactions have been reported from postmarketing experience with unknown frequency. If angioedema involves the throat, tongue, glottis or larynx, or if the patient has a history of upper respiratory surgery, airway obstruction may occur and be fatal. Patients who experience these effects, even without respiratory distress, require prolonged observation and appropriate monitoring measures since treatment with antihistamines and corticosteroids may not be sufficient to prevent respiratory involvement. Prompt administration of subcutaneous epinephrine solution 1:1000 (0.3 mL to 0.5 mL) and measures to ensure a patent airway may be necessary.
Discontinue Tekturna HCT immediately in patients who develop anaphylactic reactions or angioedema, and do not readminister [see Contraindications ( 4)] .
5.4 Hypotension
Symptomatic hypotension may occur after initiation of treatment with Tekturna HCT in patients with marked volume depletion, patients with salt depletion, or with combined use of aliskiren and other agents acting on the renin-angiotensin-aldosterone system (RAAS). The volume or salt depletion should be corrected prior to administration of Tekturna HCT, or the treatment should start under close medical supervision.
A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
5.5 Impaired Renal Function
Monitor renal function periodically in patients treated with Tekturna HCT. Changes in renal function, including acute renal failure, can be caused by drugs that affect the RAAS and by diuretics. Patients whose renal function may depend in part on the activity of the RAAS (e.g., patients with renal artery stenosis, severe heart failure, post-myocardial infarction or volume depletion) or patients receiving ARB, ACEI or nonsteroidal anti- inflammatory drug (NSAID), including selective Cyclooxygenase-2 inhibitors (COX-2 inhibitors), therapy may be at particular risk of developing acute renal failure on Tekturna HCT [see Warnings and Precautions ( 5.2), Drug Interactions ( 7), and Clinical Studies ( 14.4)] . Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on Tekturna HCT.
5.8 Serum Electrolyte Abnormalities
Tekturna HCT
In the short-term controlled trials of various doses of Tekturna HCT, in patients with hypertension not concomitantly treated with an ARB or ACEI, the incidence of hypertensive patients who developed hypokalemia (serum potassium less than 3.5 mEq/L) was 2.2%; the incidence of hyperkalemia (serum potassium greater than 5.5 mEq/L) was 0.8%. No patients discontinued due to increase or decrease of serum potassium.
Aliskiren
Monitor serum potassium periodically in patients receiving aliskiren. Drugs that affect the RAAS can cause hyperkalemia. Risk factors for the development of hyperkalemia include renal insufficiency, diabetes, combination use with ARBs or ACEIs [see Contraindications ( 4), Warnings and Precautions ( 5.2), and Clinical Studies ( 14.4)], NSAIDs, including selective Cyclooxygenase-2 inhibitors (COX-2 inhibitors), or potassium supplements or potassium-sparing diuretics.
Hydrochlorothiazide (HCTZ)
HCTZ can cause hypokalemia and hyponatremia. Hypomagnesemia can result in hypokalemia which appears difficult to treat despite potassium repletion.
If hypokalemia is accompanied by clinical signs (e.g., muscular weakness, paresis, or ECG alterations), Tekturna HCT should be discontinued. Correction of hypokalemia and any coexisting hypomagnesemia is recommended prior to the initiation of thiazides.
5.9 Cyclosporine or Itraconazole
Aliskiren
When aliskiren was given with cyclosporine or itraconazole, the blood concentrations of aliskiren were significantly increased. Avoid concomitant use of aliskiren with cyclosporine or itraconazole [see Drug Interactions ( 7)] .
5.10 Acute Myopia and Secondary Angle-Closure Glaucoma
HCTZ, a sulfonamide, can cause an idiosyncratic reaction, resulting in acute transient myopia and acute angle- closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue HCTZ as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
5.11 Metabolic Disturbances
Hydrochlorothiazide (HCTZ)
HCTZ may alter glucose tolerance and raise serum levels of cholesterol and triglycerides.
HCTZ may raise the serum uric acid level due to reduced clearance of uric acid and may cause or exacerbate hyperuricemia and precipitate gout in susceptible patients.
HCTZ decreases urinary calcium excretion and may cause elevations of serum calcium. Monitor calcium levels in patients with hypercalcemia receiving Tekturna HCT.
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- Fetal Toxicity [see Warnings and Precautions ( 5.1)].
- Anaphylactic Reactions and Head and Neck Angioedema [see Warnings and Precautions ( 5.3)].
- Hypotension [see Warnings and Precautions ( 5.4)].
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
Tekturna HCT
Tekturna HCT has been evaluated for safety in more than 2,700 patients, including over 700 treated for 6 months and 190 for over 1 year. In placebo-controlled clinical trials, discontinuation of therapy due to a clinical adverse event (including uncontrolled hypertension) occurred in 2.7% of patients treated with Tekturna HCT versus 3.6% of patients given placebo.
Adverse events in placebo-controlled trials that occurred in at least 1% of patients treated with Tekturna HCT and at a higher incidence than placebo included dizziness (2.3% versus 1%), influenza (2.3% versus 1.6%), diarrhea (1.6% versus 0.5%), cough (1.3% versus 0.5%), vertigo (1.2% versus 0.5%), asthenia (1.2% versus 0%), and arthralgia (1% versus 0.5%).
Aliskiren
Aliskiren has been evaluated for safety in 6,460 patients, including 1,740 treated for longer than 6 months, and 1,250 for longer than 1 year. In placebo-controlled clinical trials, discontinuation of therapy due to a clinical adverse event, including uncontrolled hypertension occurred in 2.2% of patients treated with aliskiren, versus 3.5% of patients given placebo. These data do not include information from the ALTITUDE study which evaluated the use of aliskiren in combination with ARBs or ACEIs [see Contraindications ( 4), Warnings and Precautions ( 5.2), and Clinical Studies ( 14.4)].
Two cases of angioedema with respiratory symptoms were reported with aliskiren use in the clinical studies. Two other cases of periorbital edema without respiratory symptoms were reported as possible angioedema and resulted in discontinuation. The rate of these angioedema cases in the completed studies was 0.06%.
In addition, 26 other cases of edema involving the face, hands, or whole body were reported with aliskiren use, including 4 leading to discontinuation.
In the placebo-controlled studies, however, the incidence of edema involving the face, hands, or whole body was 0.4% with aliskiren compared with 0.5% with placebo. In a long-term active-controlled study with aliskiren and HCTZ arms, the incidence of edema involving the face, hands, or whole body was 0.4% in both treatment arms.
Aliskiren produces dose-related gastrointestinal (GI) adverse reactions. Diarrhea was reported by 2.3% of patients at 300 mg, compared to 1.2% in placebo patients. In women and the elderly (age 65 years and older) increases in diarrhea rates were evident starting at a dose of 150 mg daily, with rates for these subgroups at 150 mg comparable to those seen at 300 mg for men or younger patients (all rates about 2% to 2.3%). Other GI symptoms included abdominal pain, dyspepsia, and gastroesophageal reflux, although increased rates for abdominal pain and dyspepsia were distinguished from placebo only at 600 mg daily. Diarrhea and other GI symptoms were typically mild and rarely led to discontinuation.
Aliskiren was associated with a slight increase in cough in the placebo-controlled studies (1.1% for any aliskiren use versus 0.6% for placebo). In active-controlled trials with ACE inhibitor (ramipril, lisinopril) arms, the rates of cough for the aliskiren arms were about one-third to one-half the rates in the ACE inhibitor arms.
Other adverse reactions with increased rates for aliskiren compared to placebo included rash (1% versus 0.3%) and renal stones (0.2% versus 0%). Single episodes of tonic-clonic seizures with loss of consciousness were reported in 2 patients treated with aliskiren in the clinical trials. One patient had predisposing causes for seizures and had a negative electroencephalogram (EEG) and cerebral imaging following the seizures; for the other patient, EEG and imaging results were not reported. Aliskiren was discontinued and there was no rechallenge in either case.
No clinically meaningful changes in vital signs or in ECG (including QTc interval) were observed in patients treated with aliskiren.
Hydrochlorothiazide (HCTZ)
Other adverse reactions that have been reported with HCTZ, without regard to causality, are listed below:
Body As A Whole: weakness
Digestive: pancreatitis, jaundice (intrahepatic cholestatic jaundice), sialadenitis, cramping, gastric irritation
Hematologic: aplastic anemia, agranulocytosis, leukopenia, hemolytic anemia, thrombocytopenia;
Hypersensitivity: purpura, photosensitivity, urticaria, necrotizing angiitis (vasculitis and cutaneous vasculitis), fever, respiratory distress including pneumonitis and pulmonary edema, anaphylactic reactions
Metabolic: hyperglycemia, glycosuria, hyperuricemia
Musculoskeletal: muscle spasm
Nervous System/Psychiatric: restlessness
Renal: renal failure, renal dysfunction, interstitial nephritis
Skin: erythema multiforme including Stevens-Johnson syndrome, exfoliative dermatitis including toxic epidermal necrolysis
Special Senses: transient blurred vision, xanthopsia
Clinical Laboratory Test Abnormalities
In controlled clinical trials, clinically important changes in standard laboratory parameters were rarely associated with administration of Tekturna HCT in patients with hypertension not concomitantly treated with an ARB or ACEI.
Blood Urea Nitrogen (BUN)/Creatinine: In patients with hypertension not concomitantly treated with an ARB or ACEI, elevations (greater than 50% increase) in BUN and creatinine occurred in 11.8% and 0.9%, respectively, of patients taking Tekturna HCT, and 7% and 1.1%, respectively, of patients given placebo in short-term controlled clinical trials. No patients were discontinued due to an increase in either BUN or creatinine.
6.2 Post-Marketing Experience
The following adverse reactions have been reported in aliskiren or hydrochlorothiazide post-marketing experience. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
Aliskiren
Hypersensitivity: anaphylactic reactions and angioedema requiring airway management and hospitalization, urticaria, peripheral edema, hepatic enzyme increase with clinical symptoms of hepatic dysfunction, severe cutaneous adverse reactions, including Stevens-Johnson syndrome and toxic epidermal necrolysis, pruritus, erythema, hyponatremia , nausea, vomiting.
Hydrochlorothiazide
Acute renal failure, renal disorder, aplastic anemia, erythema mutliforme, pyrexia, muscle spasm, asthenia, acute angle-closure glaucoma, bone marrow failure, worsening of diabetes control, hypokalemia, blood lipids increased, hyponatremia, hypomagnesemia, hypercalcemia, hyperchloremic alkalosis, impotence, visual impairment.
Pathological changes in the parathyroid gland of patients with hypercalcemia and hypophosphatemia have been observed in a few patients on prolonged thiazide therapy. If hypercalcemia occurs, further diagnostic evaluation is necessary.
Non-melanoma Skin Cancer: Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
7 DRUG INTERACTIONS
No drug interaction studies have been conducted with Tekturna HCT and other drugs, although studies with the individual aliskiren and HCTZ components are described below.
Aliskiren
Cyclosporine: Avoid coadministration of cyclosporine with aliskiren [see Warnings and Precautions ( 5.9) and Clinical Pharmacology ( 12.3)] .
Itraconazole: Avoid coadministration of itraconazole with aliskiren [see Warnings and Precautions ( 5.7) and Clinical Pharmacology ( 12.3)].
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) including selective Cyclooxygenase-2 inhibitors (COX-2 inhibitors): In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors with agents that affect the RAAS, including aliskiren, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving aliskiren and NSAID therapy.
The antihypertensive effect of aliskiren may be attenuated by NSAIDs.
Dual Blockade of the Renin-Angiotensin-Aldosterone System (RAAS): The concomitant use of aliskiren with other agents acting on the RAAS such as ACEIs or ARBs is associated with an increased risk of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Most patients receiving the combination of two drugs that inhibit the renin-angiotensin system do not obtain any additional benefit compared to monotherapy. In general, avoid combined use of aliskiren with ACE inhibitors or ARBs, particularly in patients with CrCl less than 60 mL/min. Monitor blood pressure, renal function, and electrolytes in patients on aliskiren and other agents that affect the RAAS [see Warnings and Precautions ( 5.4, 5.5, 5.8)].
The concomitant use of aliskiren with an ARB or an ACEI in diabetic patients is contraindicated [see Contraindications ( 4)].
Furosemide: Oral coadministration of aliskiren and furosemide reduced exposure to furosemide. Monitor diuretic effects when furosemide is coadministered with aliskiren.
Hydrochlorothiazide (HCTZ)
When administered concurrently, the following drugs may interact with thiazide diuretics.
Antidiabetic drugs (oral agents and insulin): Dosage adjustment of the antidiabetic drug may be required.
Lithium: Diuretic agents increase the risk of lithium toxicity. Increases in serum lithium concentrations and lithium toxicity have been reported during concomitant administration of lithium with angiotensin II receptor antagonists. Monitoring of serum lithium levels is recommended during concomitant use.
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) and COX-2 selective agents: When Tekturna HCT and NSAIDs are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
Ion-exchange Resins: Staggering the dosage of HCTZ and ion exchange resins (e.g., cholestyramine, colestipol) such that HCTZ is administered at least 4 hours before or 4 to 6 hours after the administration of resins would potentially minimize the interaction [see Clinical Pharmacology ( 12.3)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Tekturna HCT can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin- angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin- angiotensin system from other antihypertensive agents. When pregnancy is detected, discontinue Tekturna HCT as soon as possible.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major malformations and miscarriage in clinically recognized pregnancies is 2-4%, and 15-20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Hypertension in pregnancy increases the maternal risk for pre-eclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section, and post-partum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Aliskiren
Use of drugs that act on the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: reduced fetal renal function leading to anuria and renal failure, oligohydramnios, fetal lung hypoplasia and skeletal deformations, including skull hypoplasia, hypotension, and death. In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus.
In patients taking Tekturna HCT during pregnancy, perform serial ultrasound examinations to assess the intra-amniotic environment. Fetal testing may be appropriate, based on the week of gestation. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to Tekturna HCT for hypotension, oliguria, and hyperkalemia. If oliguria or hypotension occur in neonates with a history of in utero exposure to Tekturna HCT, support blood pressure and renal perfusion Exchange transfusions or dialysis may be required as a means of reversing hypotension and substituting for disordered renal function.
Hydrochlorothiazide
Thiazides cross the placenta, and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possible other adverse reactions that have occurred in adults.
Aliskiren
In developmental toxicity studies, pregnant rats and rabbits received oral aliskiren hemifumarate during organogenesis at doses up to 20 and 7 times the maximum recommended human dose (MRHD) based on body surface area (mg/m 2), respectively, in rats and rabbits. (Actual animal doses were up to 600 mg/kg/day in rats and up to 100 mg/kg/day in rabbits.) No teratogenicity was observed; however, fetal birth weight was decreased in rabbits at doses 3.2 times the MRHD based on body surface area (mg/m 2). Aliskiren was present in placentas, amniotic fluid and fetuses of pregnant rabbits.
HCTZ
When pregnant mice and rats were given HCTZ at doses up to 3000 and 1000 mg/kg/day, respectively (about 600 and 400 times the MRHD), during their respective periods of major organogenesis, there was no evidence of fetal harm.
Thiazides can cross the placenta, and concentrations reached in the umbilical vein approach those in the maternal plasma. HCTZ, like other diuretics, can cause placental hypoperfusion. It accumulates in the amniotic fluid, with reported concentrations up to 19 times higher than in umbilical vein plasma. Use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice or thrombocytopenia. Since they do not prevent or alter the course of EPH (Edema, Proteinuria, Hypertension) gestosis (pre eclampsia), these drugs should not be used to treat hypertension in pregnant women. The use of HCTZ for other indications (e.g., heart disease) in pregnancy should be avoided.
These animal studies were conducted according to the standards of the time.
8.2 Lactation
Risk Summary
There is no information regarding the presence of Tekturna HCT or aliskiren in human milk, the effects on the breastfed infant, or the effects on milk production. Limited published studies report that hydrochlorothiazide is present in human milk. However, there is insufficient information to determine the effects hydrochlorothiazide on the breastfed infant or the effects of hydrochlorothiazide on milk production. Because of the potential for serious adverse reactions, including hypotension, electrolyte imbalances and renal impairment in nursing infants, advise a nursing woman that breastfeeding is not recommended during treatment with Tekturna HCT.
8.4 Pediatric Use
Safety and effectiveness of Tekturna HCT in pediatric patients have not been established.
Preclinical studies indicate a potential for substantial increase in exposure to aliskiren in pediatric patients [see Nonclinical Toxicology ( 13.2)] .
Neonates with a history of in utero exposure to Tekturna HCT:
If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function.
8.5 Geriatric Use
In the short-term controlled clinical trials of Tekturna HCT, 325 (19.6%) patients treated with Tekturna HCT were 65 years and older and 53 (3.2%) were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
Safety and effectiveness of Tekturna HCT in patients with severe renal impairment [creatinine clearance (CrCl) less than 30 mL/min] have not been established [see Warnings and Precautions ( 5.6), Clinical Pharmacology ( 12.3) and Clinical Studies ( 14)] .
No dose adjustment is required in patients with mild (CrCl 60 to 90 mL/min) or moderate (CrCl 30 to 60 mL/min) renal impairment.
8.7 Hepatic Impairment
Aliskiren
No dose adjustment is necessary for patients with mild-to-severe liver disease [see Clinical Pharmacology ( 12.3)].
10 OVERDOSAGE
Aliskiren
Limited data are available related to overdosage in humans. The most likely manifestation of overdosage would be hypotension. If symptomatic hypotension should occur, supportive treatment should be initiated.
Aliskiren is poorly dialyzed. Therefore, hemodialysis is not adequate to treat aliskiren overexposure [see Clinical Pharmacology ( 12.3)] .
Hydrochlorothiazide (HCTZ)
The most common signs and symptoms of overdose observed in humans are those caused by electrolyte depletion (hypokalemia, hypochloremia, hyponatremia) and dehydration resulting from excessive diuresis. If digitalis has also been administered, hypokalemia may accentuate cardiac arrhythmias. The degree to which HCTZ is removed by hemodialysis has not been established. The oral LD 50 of HCTZ is greater than 10 g/kg in both mice and rats.
11 DESCRIPTION
Tekturna HCT is a fixed combination of aliskiren hemifumarate, an orally active, nonpeptide, direct renin inhibitor, and HCTZ, a thiazide diuretic that is provided as tablets for oral administration.
Aliskiren
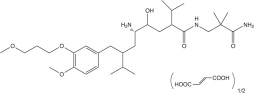
Aliskiren hemifumarate is chemically described as (2S,4S,5S,7S)-N-(2-carbamoyl-2-methylpropyl)-5-amino-4- hydroxy-2,7-diisopropyl-8-[4-methoxy-3-(3-methoxypropoxy)phenyl]-octanamide hemifumarate and its structural formula is:
Molecular formula: C 30H 53N 3O 6 • 0.5 C 4H 4O 4
Aliskiren hemifumarate is a white to slightly yellowish crystalline powder with a molecular weight of 609.8 (free base- 551.8). It is soluble in phosphate buffer, n-octanol, and highly soluble in water.
Hydrochlorothiazide (HCTZ)
Hydrochlorothiazide USP is a white, or practically white, practically odorless, crystalline powder. It is slightly soluble in water; freely soluble in sodium hydroxide solution, in n-butylamine, and in dimethylformamide; sparingly soluble in methanol; and insoluble in ether, in chloroform, and in dilute mineral acids.
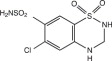
Hydrochlorothiazide is chemically described as 6-chloro-3,4-dihydro-2 H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide.
Hydrochlorothiazide is a thiazide diuretic. Its empirical formula is C 7H 8ClN 3O 4S 2, its molecular weight is 297.73, and its structural formula is:
Tekturna HCT tablets are formulated for oral administration to contain aliskiren and hydrochlorothiazide, USP 150/12.5 mg, 150/25 mg, 300/12.5 mg, and 300/25 mg. The inactive ingredients for all strengths of the tablets are colloidal silicon dioxide, crospovidone, hydroxypropyl methylcellulose, iron oxide colorants, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, talc, titanium dioxide, and wheat starch.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Aliskiren
Renin is secreted by the kidney in response to decreases in blood volume and renal perfusion. Renin cleaves angiotensinogen to form the inactive decapeptide angiotensin I (Ang I). Ang I is converted to the active octapeptide angiotensin II (Ang II) by angiotensin-converting enzyme (ACE) and non-ACE pathways. Ang II is a powerful vasoconstrictor and leads to the release of catecholamines from the adrenal medulla and prejunctional nerve endings. It also promotes aldosterone secretion and sodium reabsorption. Together, these effects increase blood pressure. Ang II also inhibits renin release, thus providing a negative feedback to the system. This cycle, from renin through angiotensin to aldosterone and its associated negative feedback loop, is known as the renin-angiotensin-aldosterone system (RAAS). Aliskiren is a direct renin inhibitor, decreasing plasma renin activity (PRA) and inhibiting the conversion of angiotensinogen to Ang I. Whether aliskiren affects other RAAS components, e.g., ACE or non-ACE pathways, is not known.
All agents that inhibit the RAAS, including renin inhibitors, suppress the negative feedback loop, leading to a compensatory rise in plasma renin concentration. When this rise occurs during treatment with ACE inhibitors and ARBs, the result is increased levels of PRA. During treatment with aliskiren, however, the effect of increased renin levels is blocked, so that PRA, Ang I and Ang II are all reduced, whether aliskiren is used as monotherapy or in combination with other antihypertensive agents.
Hydrochlorothiazide (HCTZ)
HCTZ is a thiazide diuretic. Thiazides affect the renal tubular mechanisms of electrolyte reabsorption, directly increasing excretion of sodium and chloride in approximately equivalent amounts. Indirectly, the diuretic action of HCTZ reduces plasma volume, with consequent increases in plasma renin activity, increases in aldosterone secretion, increases in urinary potassium loss, and decreases in serum potassium. The renin-aldosterone link is mediated by angiotensin II, so coadministration of agents that block the production or function of angiotensin II tends to reverse the potassium loss associated with these diuretics.
The mechanism of action of the antihypertensive effect of thiazides is unknown.
12.2 Pharmacodynamics
Tekturna HCT
In placebo-controlled clinical trials, PRA was decreased with aliskiren monotherapy (ranging from 54% to 65%) and increased with hydrochlorothiazide monotherapy (ranging from 4% to 72%). Treatment with Tekturna HCT resulted in PRA reductions ranging from approximately 46% to 63% in various doses despite the increase in PRA with hydrochlorothiazide treatment. The clinical implications of the differences in effect on PRA are not known.
Aliskiren
PRA reductions in clinical trials ranged from approximately 50% to 80%, were not dose-related and did not correlate with blood pressure reductions. The clinical implications of the differences in effect on PRA are not known.
Hydrochlorothiazide (HCTZ)
After oral administration of HCTZ, diuresis begins within 2 hours, peaks in about 4 hours, and lasts about 6 to 12 hours.
Drug Interactions
Hydrochlorothiazide (HCTZ)
Alcohol, barbiturates, or narcotics: Potentiation of orthostatic hypotension may occur.
Skeletal muscle relaxants: Possible increased responsiveness to muscle relaxants such as curare derivatives.
Digitalis glycosides: Thiazide-induced hypokalemia or hypomagnesemia may predispose the patient to digoxin toxicity.
12.3 Pharmacokinetics
Absorption and Distribution
Tekturna HCT
Following oral administration of Tekturna HCT combination tablets, the median peak plasma concentration time is within 1 hour for aliskiren and 2.5 hours for HCTZ. When taken with food, mean AUC and C max of aliskiren are decreased by 60% and 82%, respectively; mean AUC and C max of HCTZ increased by 13% and 10%, respectively. As a result, patients should establish a routine pattern for taking Tekturna HCT with regard to meals.
Aliskiren
Aliskiren is poorly absorbed (bioavailability about 2.5%). Following oral administration, peak plasma concentrations of aliskiren are reached within 1 to 3 hours. When taken with a high fat meal, mean AUC and C max of aliskiren are decreased by 71% and 85% respectively. In the clinical trials of aliskiren, it was administered without requiring a fixed relation of administration to meals.
Hydrochlorothiazide (HCTZ)
The estimated absolute bioavailability of HCTZ after oral administration is about 70%. Peak plasma HCTZ concentrations (C max) are reached within 2 to 5 hours after oral administration. There is no clinically significant effect of food on the bioavailability of HCTZ.
HCTZ binds to albumin (40% to 70%) and distributes into erythrocytes. Following oral administration, plasma HCTZ concentrations decline bi-exponentially, with a mean distribution half-life of about 2 hours and an elimination half-life of about 10 hours.
Metabolism and Elimination
Aliskiren
The effective half-life for aliskiren is 24 hours. Steady state blood levels are reached in about 7 to 8 days. About one-fourth of the absorbed dose appears in the urine as parent drug. How much of the absorbed dose is metabolized is unknown. Based on the in vitro studies, the major enzyme responsible for aliskiren metabolism appears to be CYP3A4. Aliskiren does not inhibit the CYP450 isoenzymes (CYP 1A2, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A) or induce CYP3A4.
Transporters: Pgp (MDR1/Mdr1a/1b) was found to be the major efflux system involved in intestinal absorption and elimination via biliary excretion of aliskiren in preclinical studies. The potential for drug interactions at the Pgp site will likely depend on the degree of inhibition of this transporter.
Hydrochlorothiazide (HCTZ)
About 70% of an orally administered dose of HCTZ is eliminated in the urine as unchanged drug.
Drug Interactions:
Aliskiren
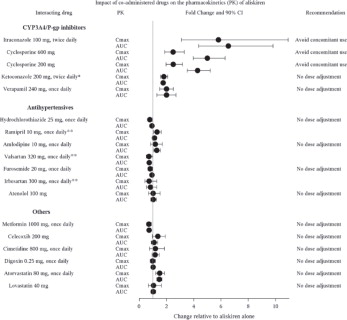
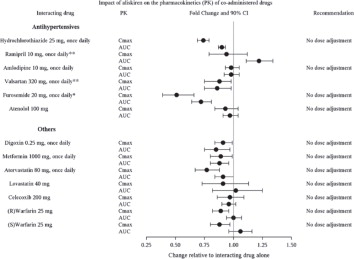
The effect of coadministered drugs on the pharmacokinetics of aliskiren and vice versa, were studied in several single and multiple dose studies. Pharmacokinetic measures indicating the magnitude of these interactions are presented in
Figure 5 (impact of coadministered drugs on aliskiren) and
Figure 6 (impact on coadministered drugs).
Figure 5: The Impact of Coadministered Drugs on the Pharmacokinetics of Aliskiren
*Ketoconazole: A 400 mg once daily dose was not studied, but would be expected to increase aliskiren blood levels further.
**Ramipril, valsartan, irbesartan: In general, avoid combined use of aliskiren with ACE inhibitors or ARBs, particularly in patients with CrCl less than 60 mL/min [see Drug Interactions ( 7)].
Warfarin: There was no clinically significant effect of a single dose of warfarin 25 mg on the pharmacokinetics of aliskiren.
Figure 6: The Impact of Aliskiren on the Pharmacokinetics of Coadministered Drugs
* Furosemide: Patients receiving furosemide may find its effects diminished after starting aliskiren. In patients with heart failure, co-administration of aliskiren (300 mg/day) reduced plasma AUC and C max of oral furosemide (60 mg/day) by 17% and 27%, respectively, and reduced 24 hour urinary furosemide excretion by 29%. This change in exposure did not result in statistically significant difference in total urine volume and urinary sodium excretion over 24 hours. However, a transient decrease in urinary sodium excretion and urine volume effects up to 12 hours were observed when furosemide was co-administered with aliskiren 300 mg/day.
**Ramipril, valsartan: In general, avoid combined use of aliskiren with ACE inhibitors or ARBs, particularly in patients with CrCl less than 60 mL/min [see Drug Interactions ( 7)].
Drugs that alter gastrointestinal motility: The bioavailability of thiazide-type diuretics may be increased by anticholinergic agents (e.g., atropine, biperiden), apparently due to a decrease in gastrointestinal motility and the stomach emptying rate. Conversely, pro-kinetic drugs may decrease the bioavailability of thiazide diuretics.
Cholestyramine: In a dedicated drug interaction study, administration of cholestyramine 2 hours before HCTZ resulted in a 70% reduction in exposure to HCTZ. Further, administration of HCTZ 2 hours before cholestyramine, resulted in 35% reduction in exposure to HCTZ.
Antineoplastic agents (e.g., cyclophosphamide, methotrexate): Concomitant use of thiazide diuretics may reduce renal excretion of cytotoxic agents and enhance their myelosuppressive effects.
Special Populations
Pediatric Patients
The pharmacokinetics of aliskiren have not been investigated in patients less than 18 years of age.
Aliskiren
The pharmacokinetics of aliskiren were studied in the elderly (65 years and older). Exposure (measured by AUC) is increased in elderly patients.
Hydrochlorothiazide (HCTZ)
A limited amount of data suggest that the systemic clearance of HCTZ is reduced in both healthy and hypertensive elderly subjects compared to young healthy volunteers.
Race
Too few non-Caucasians have been studied with Tekturna HCT to assess pharmacokinetic differences among races. The pharmacokinetic differences among blacks, Caucasians, and Japanese are minimal with aliskiren therapy.
Aliskiren
The pharmacokinetics of aliskiren were evaluated in patients with varying degrees of renal impairment. Rate and extent of exposure (AUC and C max) of aliskiren in subjects with renal impairment did not show a consistent correlation with the severity of renal impairment [see Use in Specific Populations ( 8.6)].
The pharmacokinetics of aliskiren following administration of a single oral dose of 300 mg was evaluated in patients with End Stage Renal Disease (ESRD) undergoing hemodialysis. When compared to matched healthy subjects, changes in the rate and extent of aliskiren exposure (C max and AUC) in ESRD patients undergoing hemodialysis were not clinically significant.
Timing of hemodialysis did not significantly alter the pharmacokinetics of aliskiren in ESRD patients. Therefore, no dose adjustment is warranted in ESRD patients receiving hemodialysis.
Hydrochlorothiazide (HCTZ)
In a study in individuals with impaired renal function, the mean elimination half-life of HCTZ was doubled in individuals with mild/moderate renal impairment (30 < CrCl < 90 mL/min) and tripled in severe renal impairment (CrCl less than or equal to 30 mL/min), compared to individuals with normal renal function (CrCl greater than 90 mL/min) [see Use in Specific Populations ( 8.6)] .
Aliskiren
The pharmacokinetics of aliskiren were not significantly affected in patients with mild-to-severe liver disease [see Use in Specific Populations ( 8.7)] .
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Tekturna HCT
No carcinogenicity, mutagenicity or fertility studies have been conducted with Tekturna HCT. However, these studies have been conducted for aliskiren as well as HCTZ alone.
Aliskiren
Carcinogenic potential was assessed in a 2-year rat study and a 6-month transgenic (rasH2) mouse study with aliskiren hemifumarate at oral doses of up to 1500 mg aliskiren/kg/day. Although there were no statistically significant increases in tumor incidence associated with exposure to aliskiren, mucosal epithelial hyperplasia with or without erosion/ulceration) was observed in the lower gastrointestinal tract at doses of 750 or more mg/kg/day in both species, with a colonic adenoma identified in 1 rat and a cecal adenocarcinoma identified in another, rare tumors in the strain of rat studied. On a systemic exposure (AUC 0-24hr) basis, 1500 mg/kg/day in the rat was about 4 times and in the mouse about 1.5 times the MRHD (300 mg aliskiren/day). Mucosal hyperplasia in the cecum or colon of rats was also observed at doses of 250 mg/kg/day (the lowest tested dose) as well as at higher doses in 4- and 13-week studies.
Aliskiren hemifumarate was devoid of genotoxic potential in the Ames reverse mutation assay with S. typhimurium and E. coli, the in vitro Chinese hamster ovary cell chromosomal aberration assay, the in vitro Chinese hamster V79 cell gene mutation test and the in vivo mouse bone marrow micronucleus assay.
Fertility of male and female rats was unaffected at doses of up to 250 mg aliskiren/kg/day (8 times the MRHD of 300 mg Tekturna/60 kg on a mg/m 2 basis).
Hydrochlorothiazide (HCTZ)
Two-year feeding studies in mice and rats conducted under the auspices of the National Toxicology Program (NTP) uncovered no evidence of a carcinogenic potential of HCTZ in female mice (at doses of up to approximately 600 mg/kg/day) or in male and female rats (at doses of up to approximately 100 mg/kg/day). The NTP, however, found equivocal evidence for hepatocarcinogenicity in male mice.
HCTZ was not genotoxic in vitro in the Ames mutagenicity assay of S. typhimurium strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 and in the Chinese Hamster Ovary (CHO) test for chromosomal aberrations, or in vivo in assays using mouse germinal cell chromosomes, Chinese hamster bone marrow chromosomes, and the Drosophila sex-linked recessive lethal trait gene. Positive test results were obtained only in the in vitro CHO Sister Chromatid Exchange (clastogenicity) and in the Mouse Lymphoma Cell (mutagenicity) assays, using concentrations of HCTZ from 43 to 1300 mcg/mL, and in the Aspergillums Nidulans nondisjunction assay at an unspecified concentration.
HCTZ was not teratogenic and had no adverse effects on the fertility of mice and rats of either sex in studies wherein these species were exposed, via their diet, to doses of up to 100 and 4 mg/kg, respectively, prior to mating and throughout gestation. These doses of HCTZ in mice and rats represent 19 and 1.5 times, respectively, the MRHD on a mg/m 2 basis. (Calculations assume an oral dose of 25 mg/day and a 60-kg patient.)
13.2 Animal Toxicology and/or Pharmacology
Juvenile Animal Studies
Juvenile toxicity studies indicated increased systemic exposure to aliskiren 85- to 385-fold in 14 day and 8 day old rats respectively, compared with adult rats. The mdr1 gene expression in juvenile rats was also significantly lower when compared to adult rats. The increased aliskiren exposure in juvenile rats appears to be mainly attributed to lack of maturation of P-gp. The overexposure in juvenile rats was associated with high mortality [see Use in Specific Populations ( 8.4)] .
14 CLINICAL STUDIES
14.1 Tekturna HCT
In all clinical trials including over 6,200 patients, more than 2,700 patients were exposed to combinations of aliskiren and HCTZ. The safety and efficacy of Tekturna HCT were evaluated in patients with mild-to-moderate hypertension in an 8-week, randomized, double-blind, placebo-controlled, parallel-group, 15-arm factorial trial (n=2762). Patients were randomized to receive various combinations of aliskiren (75 mg to 300 mg) plus HCTZ (6.25 mg to 25 mg) once daily (without titrating up from monotherapy) and followed for blood pressure response. The combination of aliskiren and HCTZ resulted in additive placebo-adjusted decreases in systolic and diastolic blood pressure at trough of 10–14/5–7 mmHg at doses of 150–300 mg/12.5–25 mg, compared to 5–8/2–3 mmHg for aliskiren 150 mg to 300 mg and 6–7/2–3 mmHg for HCTZ 12.5 mg to 25 mg, alone. Blood pressure reductions with the combinations were greater than the reductions with the monotherapies as shown in Table 1.
| Hydrochlorothiazide, mg | |||||
| 0 | 6.25 | 12.5 | 25 | ||
| Aliskiren, mg | Placebo Mean Change | Placebo- subtracted | Placebo- subtracted | Placebo- subtracted | Placebo- subtracted |
| 0 | 7.5/6.9 | -- | 3.5/2.1 | 6.4/3.2 | 6.8/2.4 |
| 75 | -- | 1.9/1.8 | 6.8/3.8 | 8.2/4.2 | 9.8/4.5 |
| 150 | -- | 4.8/2 | 7.8/3.4 | 10.1/5 | 12/5.7 |
| 300 | -- | 8.3/3.3 | -- | 12.3/7 | 13.7/7.3 |
The safety and efficacy of Tekturna HCT as initial therapy was evaluated in this trial. All patients randomized to the combination groups received the combination treatment of Tekturna HCT at assigned doses as initial therapy without titration from monotherapy. The figures [see Indications and Usage ( 1)] display the probability that a patient will achieve systolic or diastolic blood pressure goal with Tekturna HCT 300/25 mg, based upon their baseline systolic or diastolic blood pressure. At all levels of baseline blood pressure, the probability of achieving any given diastolic or systolic goal is greater with the combination than for either monotherapy.
The antihypertensive effect of Tekturna HCT was largely manifested within 1 week. The maximum antihypertensive effect was generally attained after about 4 weeks of therapy.
One active-controlled trial investigated the addition of 300 mg aliskiren in obese hypertensive patients who did not respond adequately to HCTZ 25 mg, and showed incremental decreases of systolic and diastolic blood pressure of approximately 7/4 mmHg.
In long-term follow-up studies (without placebo control) the effect of the combination of aliskiren and HCTZ was maintained for over 1 year.
The antihypertensive effect was independent of age and gender. There were too few non-Caucasians to assess differences in blood pressure effects by race.
14.2 Aliskiren Monotherapy
The antihypertensive effects of aliskiren have been demonstrated in 6 randomized, double-blind, placebo- controlled, 8-week clinical trials in patients with mild-to-moderate hypertension. The placebo response and placebo-subtracted changes from baseline in seated trough cuff blood pressure are shown in Table 2.
|
*p value less than 0.05 versus placebo by ANCOVA with Dunnett's procedure for multiple comparisons. |
|||||
|
†p value less than 0.05 versus placebo by ANCOVA for the pairwise comparison. |
|||||
| Aliskiren Daily Dose, mg | |||||
| 75 | 150 | 300 | 600 | ||
|
Study | Placebo Mean Change |
Placebo-subtracted |
Placebo-subtracted |
Placebo-subtracted |
Placebo-subtracted |
| 1 | 2.9/3.3 | 5.7/4* | 5.9/4.5* | 11.2/7.5* | -- |
| 2 | 5.3/6.3 | -- | 6.1/2.9* | 10.5/5.4* | 10.4/5.2* |
| 3 | 10/8.6 | 2.2/1.7 | 2.1/1.7 | 5.1/3.7* | -- |
| 4 | 7.5/6.9 | 1.9/1.8 | 4.8/2* | 8.3/3.3* | -- |
| 5 | 3.8/4.9 | -- | 9.3/5.4* | 10.9/6.2* | 12.1/7.6* |
| 6 | 4.6/4.1 | -- | -- | 8.4/4.9 † | -- |
The studies included approximately 2,730 patients given doses of 75 mg to 600 mg of aliskiren and 1,231 patients given placebo. As shown in Table 2, there is some increase in response with administered dose in all studies, with reasonable effects seen at 150 mg to 300 mg, and no clear further increase at 600 mg. A substantial proportion (85% to 90%) of the blood pressure lowering effect was observed within 2 weeks of treatment.
Studies with ambulatory blood pressure monitoring showed reasonable control throughout the interdosing interval, e.g., the ratios of mean daytime to mean nighttime ambulatory BP ranged from 0.6 to 0.9.
Patients in the placebo-controlled trials continued open-label aliskiren for up to 1 year. A persistent blood pressure lowering effect was demonstrated by a randomized withdrawal study (patients randomized to continued drug or placebo), which showed a statistically significant difference between patients kept on aliskiren and those randomized to placebo. With cessation of treatment, blood pressure gradually returned toward baseline levels over a period of several weeks. There was no evidence of rebound hypertension after abrupt cessation of therapy.
The effectiveness of aliskiren was demonstrated across all demographic subgroups, although black patients tended to have smaller reductions in blood pressure than Caucasians and Asians, as has been seen with ACE inhibitors and ARBs.
14.3 Aliskiren in Combination with Other Antihypertensives
Valsartan
Aliskiren 150 mg and 300 mg and valsartan 160 mg and 320 mg were studied alone and in combination in an 8- week, 1,797-patient, randomized, double-blind, placebo-controlled, parallel-group, 4-arm, dose-escalation study. The dosages of aliskiren and valsartan were started at 150 mg and 160 mg, respectively, and increased at 4 weeks to 300 mg and 320 mg, respectively. Seated trough cuff blood pressure was measured at baseline, 4, and 8 weeks. Blood pressure reductions with the combinations were greater than the reductions with the monotherapies as shown in Table 3. In general, avoid use of aliskiren in combination with other drugs that affect the RAAS [see Contraindications ( 4), Warnings and Precautions ( 5), and Drug Interactions ( 7)] .
|
* The placebo change is 5.2/4.8 for Week 4 endpoint which was used for the dose groups containing aliskiren 150 mg or valsartan 160 mg. |
||||
| Valsartan, mg | ||||
| Aliskiren, mg | Placebo Mean Change | 0 | 160 | 320 |
| 0 | 4.6/4.1* | -- | 5.6/3.9 | 8.2/5.6 |
| 150 | -- | 5.4/2.7 | 10.0/5.7 | -- |
| 300 | -- | 8.4/4.9 | -- | 12.6/8.1 |
Amlodipine
Aliskiren 150 mg and 300 mg and amlodipine besylate 5 mg and 10 mg were studied alone and in combination in an 8- week, 1,685-patient, randomized, double-blind, placebo-controlled, multifactorial study. Treatment with aliskiren and amlodipine resulted overall in significantly greater reductions in diastolic and systolic blood pressure compared to the respective monotherapy components as shown in Table 4.
|
Aliskiren, mg |
Placebo mean change | Amlodipine, mg | ||
| 0 | 5 | 10 | ||
| 0 | 5.4/6.8 | -- | 5.6/9.0 | 8.5/14.3 |
| 150 | -- | 2.6/3.9 | 8.6/13.9 | 10.8/17.1 |
| 300 | -- | 4.9/8.6 | 9.6/15.0 | 11.1/16.4 |
ACEIs
Aliskiren has not been studied when added to maximal doses of ACEIs to determine whether aliskiren produces additional blood pressure reduction.
There are no trials of the Tekturna HCT combination tablet demonstrating reductions in cardiovascular risk in patients with hypertension, but the HCTZ component has demonstrated such benefits.
14.4 Aliskiren in Patients with Diabetes Treated with an ARB or ACEI (ALTITUDE study)
Patients with diabetes with renal disease (defined either by the presence of albuminuria or reduced GFR) were randomized to aliskiren 300 mg daily (n=4296) or placebo (n=4310). All patients were receiving background therapy with an ARB or ACEI. The primary efficacy outcome was the time to the first event of the primary composite endpoint consisting of cardiovascular death, resuscitated sudden death, nonfatal myocardial infarction, nonfatal stroke, unplanned hospitalization for heart failure, onset of end stage renal disease, renal death, and doubling of serum creatinine concentration from baseline sustained for at least 1 month. After a median follow up of about 32 months, the trial was terminated early for lack of efficacy. Higher risk of renal impairment, hypotension and hyperkalemia was observed in aliskiren compared to placebo treated patients, as shown in Table 5.
|
†renal failure, renal failure acute, renal failure chronic, renal impairment |
||||
|
††dizziness, dizziness postural, hypotension, orthostatic hypotension, presyncope, syncope |
||||
|
††† Given the variable baseline potassium levels of patients with renal insufficiency on dual RAAS therapy, the reporting of adverse event of hyperkalemia was at the discretion of the investigator. |
||||
|
* A Serious Adverse Event (SAE) is defined as: an event which is fatal or life-threatening, results in persistent or significant disability/incapacity, constitutes a congenital anomaly/birth defect, requires inpatient hospitalization or prolongation of existing hospitalization, or is medically significant (i.e., defined as an event that jeopardizes the patient or may require medical or surgical intervention to prevent one of the outcomes previously listed). |
||||
| Aliskiren N=4272 | Placebo N=4285 | |||
| Serious Adverse Events* (%) | Adverse Events (%) | Serious Adverse Events* (%) | Adverse Events (%) | |
| Renal impairment † | 5.7 | 14.5 | 4.3 | 12.4 |
| Hypotension †† | 2.3 | 19.9 | 1.9 | 16.3 |
| Hyperkalemia ††† | 1.0 | 38.9 | 0.5 | 28.8 |
The risk of stroke (3.4% aliskiren versus 2.7% placebo) and death (8.4% aliskiren versus 8.0% placebo) were also numerically higher in aliskiren-treated patients.
16 HOW SUPPLIED/STORAGE AND HANDLING
Tekturna HCT is supplied as biconvex, ovaloid film-coated tablets.
All strengths are packaged in bottles and unit-dose blister packages (10 strips of 10 tablets) as described below.
| Tablet | Color | Imprint | Imprint | NDC 70839-XXX-XX | ||
| Aliskiren/HCTZ | Side 1 | Side 2 | Bottle of 30 | Bottle of 90 | Blister Packages of
100 |
|
| 150 mg/12.5 mg | White | NVR | LCI | 112-30 | 112-90 | 112-01
|
| 150 mg/25 mg | Pale Yellow | NVR | CLL | 125-30 | 125-90 | 125-01
|
| 300 mg/12.5 mg | Violet White | NVR | CVI | 312-30 | 312-90 | 312-01
|
| 300 mg/25 mg | Light Yellow | NVR | CVV | 325-30 | 325-90 | 325-01
|
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Pregnancy
Inform female patients of childbearing age about the consequences of exposure to Tekturna HCT during pregnancy. Discuss treatment options with women planning to become pregnant. Advise patients to report pregnancies to their physicians as soon as possible.
Lactation
Advise nursing women that breastfeeding is not recommended during treatment with Tekturna HCT [see Use in Specific Populations ( 8.2)].
Symptomatic Hypotension
Inform patients that lightheadedness can occur, especially during the first days of Tekturna HCT therapy, and that it should be reported to the prescribing physician. Advise patients that if syncope occurs, Tekturna HCT should be discontinued until the physician has been consulted.
Caution patients that inadequate fluid intake, excessive perspiration, diarrhea, or vomiting can lead to an excessive fall in blood pressure, with the same consequences of lightheadedness and possible syncope.
Anaphylactic Reactions and Angioedema
Advise patients to immediately report any signs or symptoms suggesting a severe allergic reaction (difficulty breathing or swallowing, tightness of the chest, hives, general rash, swelling, itching, dizziness, vomiting, or abdominal pain) or angioedema (swelling of face, extremities, eyes, lips, tongue, difficulty in swallowing or breathing) and to take no more drug until they have consulted with the prescribing physician. Angioedema, including laryngeal edema, may occur at any time during treatment with Tekturna HCT.
Potassium Supplements
Advise patients receiving Tekturna HCT not to use potassium supplements or salt substitutes containing potassium without consulting the prescribing physician.
Patient Information
Tekturna
® HCT (tek-turn-a HCT)
(aliskiren and hydrochlorothiazide)
Tablets
Read the Patient Information that comes with Tekturna HCT before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your condition and treatment.
What is the most important information I should know about Tekturna HCT?
Tekturna HCT can cause harm or death to an unborn baby. Talk to your doctor about other ways to lower your blood pressure if you plan to become pregnant. If you get pregnant while taking Tekturna HCT, tell your doctor right away.
What is Tekturna HCT?
Tekturna HCT contains 2 prescription medicines in 1 tablet that work together to lower blood pressure. It contains:
- aliskiren (Tekturna), a direct renin inhibitor (DRI)
- hydrochlorothiazide (HCTZ), a diuretic (water pill)
Aliskiren (Tekturna) reduces the effect of renin, and the harmful process that narrows blood vessels. Aliskiren also helps blood vessels relax and widen so blood pressure is lower. Hydrochlorothiazide reduces the amount of salt and water in your body so your blood pressure is lower.
Tekturna HCT may be used to lower high blood pressure in adults
- when 1 medicine to lower high blood pressure is not enough
- as the first medicine to lower high blood pressure if your doctor decides that you are likely to need more than 1 medicine
Tekturna HCT has not been studied in children under 18 years of age.
Your doctor may prescribe other medicines for you to take along with Tekturna HCT to treat your high blood pressure.
What is high blood pressure (hypertension)?
Blood pressure is the force of blood in your blood vessels when your heart beats and when your heart rests. You have high blood pressure when the force is too much.
High blood pressure makes the heart work harder to pump blood throughout the body and causes damage to blood vessels. Tekturna HCT can help your blood vessels relax so your blood pressure is lower. Medicines that lower your blood pressure may lower your chance of having a stroke or heart attack.
Who should not take Tekturna HCT?
Do not take Tekturna HCT if:
- you get pregnant. Stop taking Tekturna HCT and call your doctor right away. If you plan to become pregnant, talk to your doctor about other treatment options for your high blood pressure.
- you have diabetes and are taking a kind of medicine called an angiotensin receptor blocker (ARB) or angiotensin-converting enzyme inhibitor (ACEI).
- you make very little or no urine due to kidney problems.
- you are allergic to aliskiren, hydrochlorothiazide, any of the other ingredients of Tekturna HCT listed at the end of this leaflet.
- Tekturna HCT has not been studied in children under 18 years of age
What should I tell my doctor before taking Tekturna HCT?
Before taking Tekturna HCT, tell your doctor if you:
- have kidney problems
- are pregnant or planning to become pregnant. See “What is the most important information I should know about Tekturna HCT?”
- have any allergies or asthma
- have liver problems
- have systemic lupus erythematosus (SLE). Tekturna HCT can make your SLE active or worse.
- have ever had an allergic reaction called angioedema, to another blood pressure medicine. Symptoms may include swelling of the face, lips, tongue, throat,arms, and legs, and may cause difficulty breathing (angioedema).
- are breastfeeding. It is not known if Tekturna HCT passes into your breast milk and if it can harm your baby.
- have any other medical problems
Tell your doctor about all the medicines you take, including prescription and nonprescription medicines, vitamins and herbal supplements. Tekturna HCT and certain other medicines may affect each other and cause side effects.
Especially tell your doctor if you are taking:
- a kind of medicine to control blood pressure called angiotensin receptor blocker (ARB) or angiotensin-converting enzyme inhibitor (ACEI)
- cholesterol-lowering medicines
- simvastatin (Zocor ®) or atorvastatin (Lipitor ®)
- cholestyramine (Questran, Questran Light, Cholestyramine Light, Locholest Light, Locholest, Prevalite)
- colestipol (Colestipol hydrochloride, Colestid, Flavored Colestid)
- medicines used to lower blood pressure, water pills (also called “diuretics”), especially potassium-sparing diuretics
- medicines for treating fungus or fungal infections (like itraconazole or ketoconazole)
- cyclosporine (Gengraf ®, Neoral, Sandimmune), a medicine used to suppress the immune system
- potassium-containing medicines, potassium supplements, or salt substitutes containing potassium
- medicines used to treat diabetes, including insulin
- lithium, a medicine used in some types of depression
- medicines used to relieve pain or inflammation, especially nonsteroidal anti- inflammatory drugs (NSAIDs) (like ibuprofen or naproxen), including selective Cyclooxygenase-2-inhibitors (COX-2 inhibitors). Ask your doctor if you are not sure if you are taking one of these medicines.
Ask your doctor if you are not sure whether you are taking one of the medicines listed above. Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine. Your doctor or pharmacist will know what medicines are safe to take together.
How should I take Tekturna HCT?
- Take Tekturna HCT exactly as prescribed by your doctor. It is important to take Tekturna HCT every day to control your blood pressure.
- Take Tekturna HCT once each day, about the same time each day.
- Take Tekturna HCT the same way every day, either with or without a meal.
- Your doctor may change your dose of Tekturna HCT if needed.
- If you miss a dose of Tekturna HCT, take it as soon as you remember. If it is close to your next dose, do not take the missed dose. Just take the next dose at your regular time.
- If you take too much Tekturna HCT, call your doctor or a Poison Control Center, or go to the nearest hospital emergency room.
What should I avoid while taking Tekturna HCT?
Drinking alcohol. Drinking alcohol during treatment with Tekturna HCT can cause you to have low blood pressure. See “ What are the possible side effects of Tekturna HCT?”
What are the possible side effects of Tekturna HCT?
Tekturna HCT may cause serious side effects:
- Harm to an unborn baby, causing injury or death. See “ What is the most important information I should know about Tekturna HCT?”
- Severe Allergic Reactions and Angioedema (hypersensitivity). Aliskiren, one of the medicines in Tekturna HCT, can cause difficulty breathing or swallowing, tightness of the chest, hives, general rash, swelling, itching, dizziness, vomiting, or abdominal pain (signs of a severe allergic reaction called anaphylactic reaction). Aliskiren can also cause swelling of the face, lips, tongue, throat, arms and legs, or the whole body (signs of angioedema). Stop taking Tekturna HCT and get medical help right away. Tell your doctor if you get any one or more of these symptoms. Angioedema can happen at any time while you are taking Tekturna HCT.
- Low blood pressure (hypotension). Your blood pressure may get too low if you also take water pills, are on a low-salt diet, get dialysis treatments, have heart problems, or get sick with vomiting or diarrhea. Drinking alcohol and taking certain medicines (barbiturates or narcotics) can cause low blood pressure to get worse. Lie down if you feel faint or dizzy, and call your doctor right away.
- Renal Impairment or Failure. Aliskiren, one of the medicines in Tekturna HCT, may cause renal disorder with symptoms such as severely decreased urine output or decreased urine output (signs of renal impairment or failure).
- Tekturna HCT may affect your potassium levels. Your doctor will do blood tests to check your potassium levels.
- Allergic reactions: Hydrochlorothiazide, one of the medicines in Tekturna HCT, can cause allergic reactions.
- Active or Worsened Systemic Lupus Erythematosus (SLE). If you have SLE, tell your doctor right away if you get any new or worse symptoms. Possible signs of SLE are facial rash, joint pain, muscle disorder, fever.
-
Eye problems. One of the medicines in Tekturna HCT can cause eye problems that may lead to vision loss. Symptoms of eye problems can happen within hours to weeks of starting Tekturna HCT. Tell your doctor right away if you have:
- Decrease in vision
- Eye pain
Common side effects of Tekturna HCT include:
- dizziness
- flu-like symptoms
- diarrhea
- cough
- tiredness
- high levels of potassium in the blood (hyperkalemia)
- vertigo
- arthralgia
Less common side effects include skin rash, severe skin reactions (signs may include severe blistering of the lips, eyes or mouth, rash with fever and skin peeling), liver disorder (signs may include nausea, loss of appetite, dark colored urine or yellowing of skin and eyes) and low level of sodium in blood.
Protect your skin from the sun and undergo regular skin cancer screening, as one of the medicines in Tekturna HCT may cause non-melanoma skin cancer.
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Tekturna HCT. For a complete list of side effects, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store Tekturna HCT?
- Store Tekturna HCT tablets at room temperature between 59°F to 86°F (15°C to 30°C).
- Keep Tekturna HCT in the original prescription bottle in a dry place. Do not remove the desiccant (drying agent) from the bottle.
Keep Tekturna HCT and all medicines out of the reach of children.
General information about Tekturna HCT
Medicines are sometimes prescribed for conditions not listed in the patient information leaflet. Do not take Tekturna HCT for a condition for which it was not prescribed. Do not give Tekturna HCT to other people, even if they have the same condition or symptoms you have. It may harm them.
This leaflet summarizes the most important information about Tekturna HCT. If you have questions about Tekturna HCT talk with your doctor or pharmacist. You can ask your doctor or pharmacist for information that is written for healthcare professionals.
For more information about Tekturna HCT, visit www.Tekturna.com, or call 1-844-399-5701.
What are the ingredients in Tekturna HCT?
Active ingredients: aliskiren hemifumarate and hydrochlorothiazide Inactive ingredients: colloidal silicon dioxide, crospovidone, hydroxypropyl methylcellulose, iron oxide colorants, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, talc, titanium dioxide, and wheat starch.
Distributed by:
NODEN PHARMA USA INC
2800 Discovery Drive, Suite 100,
Orlando, FL 32826
© 2020 Noden Pharma USA Inc. All rights reserved.
September 2020
Principal Display Panel - Tekturna HCT 150/12.5 mg Label
NDC 70839-112-30 30 tablets
Tekturna HCT®
(aliskiren and hydrochlorothiazide)
Tablets
150 mg/12.5 mg
Rx only
NODEN PHARMA™
Principal Display Panel - Tekturna HCT 150/25 mg Label
NDC 70839-125-30 30 tablets
Tekturna HCT®
(aliskiren and hydrochlorothiazide)
Tablets
150 mg/25 mg
Rx only
NODEN PHARMA™