4382 FIRST AID KIT- 4382 first aid
4381 FIRST AID KIT- 4381 first aid
4393 FIRST AID KIT- 4393 first aid
Honeywell Safety Products USA, Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
0498-4381, 4382, 4393: First Aid Kit (ammonia, Triple, NaCl irr, EW, HC cr, BZK wipe, antiseptic hand gel, alcohol wipe- 145225, Z145225, Z19802)
Triple
Active ingredient
Bacitracin zinc 400 units
Neomycin sulfate (5 mg equivalent to 3.5 mg Neomycin base)
Polymyxin B sulfate 5000 units
Triple
Warnings
For external use only
Allergy alert: do not use if you are allergic to any of the ingredients
Do not use
- in the eyes
- over large areas of the body
- Ask a doctor before use if you have
- a deep or puncture wounds
- animal bites
- serious burns
Triple
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Triple
Other information
- store at 15 0 to 25 0 C (59 0 to 77 0 F)
- tamper evident sealed packets
- do not use if packet is torn or opened
BZK Wipe
Warnings
For external use only
Do not use
- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
BZK Wipe
Other information
- store at room temperature 15 0 to 30 0 C (59 0 - 86 0 F)
- do not reuse towelette
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only
- Obtain immediate medical treatment for all open wounds in or near eyes.
- To avoid contamination, do not touch tip of container to any surface.
- Do not reuse.
- Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Eyeash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
Hydrocortisone
Active ingredient (in each gram)
Hydrocortisone acetate (equivalent to Hydrocortisone 1%)
Hydrocortisone
Uses
- for the temporary relief of itching associated with minor skin irritations and rashes
Hydrocortisone
Warnings
For external use only
When using the product
- avoid contact with eyes
- do not begin use of any other hydrocortisone product unless you have consulted a doctor
- do not use for the treatment of diaper rash
Hydrocortisone
Directions
- adults and children 2 years and older:
- clean the affected area
- apply to the area not more than 3 to 4 times daily
- children under 2 years of age: consult a doctor
Hydrocortisone
Inactive ingredients
cetyl alcohol, citric acid, diazolidinyl urea, edetate disodium, glycerin, glyceryl monostearate, methylparaben, mineral oil, polyethylene glycol, propylene glycol, propylparaben, purified water, stearic acid, trolamine
Hand Sanitizer
Warnings
For external use only
Flammable, keep away from fire or flame
Hand Sanitizer
Inactive ingredients
acrylates/C10-30 alkyl acrylate crosspolymer, aloe barbadensis leaf juice, dl-alpha tocopheryl acetate, fragrance, PEG-60 almond glycerides, propylene glycol, purified water, triisopropanolamine
NaCL Irrigant
Description
Each 100 mL contains:
Sodium Chloride USP 0.9 g; Water for Injection USP qs
pH adjusted with Hydrochloric Acid NF
pH: 5.0 (4.5–7.0) Calculated Osmolarity: 310 mOsmol/liter
Concentration of Electrolytes (mEq/liter): Sodium 154; Chloride 154
0.9% Sodium Chloride Irrigation USP is sterile, nonpyrogenic, isotonic and contains no bacteriostatic or antimicrobial agents.
The formula of the active ingredient is:
Ingredient Molecular Formula Molecular Weight
Sodium Chloride USP NaCl 58.44
The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs. The copolymer contains no plasticizers and exhibits virtually no leachability. The plastic container is also virtually impermeable to vapor transmission and, therefore, requires no overwrap to maintain the proper drug concentration. The safety of the plastic container has been confirmed by biological evaluation procedures. The material passes Class Vl testing as specified in the U.S. Pharmacopeia for Biological Tests — Plastic Containers. These tests have shown that the container is nontoxic and biologically inert.
The PIC™ Container is PVC-free and DEHP-free.
Ingredient Molecular Formula Molecular Weight
Sodium Chloride USP NaCl 58.44
The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs. The copolymer contains no plasticizers and exhibits virtually no leachability. The plastic container is also virtually impermeable to vapor transmission and, therefore, requires no overwrap to maintain the proper drug concentration. The safety of the plastic container has been confirmed by biological evaluation procedures. The material passes Class Vl testing as specified in the U.S. Pharmacopeia for Biological Tests — Plastic Containers. These tests have shown that the container is nontoxic and biologically inert.
The PIC™ Container is PVC-free and DEHP-free.
NaCL Irrigant
Clinical Pharmacology
0.9% Sodium Chloride Irrigation USP is utilized for a variety of clinical indications such as sterile irrigation of body cavities, tissues or wounds, indwelling urethral catheters, surgical drainage tubes, and for washing, rinsing or soaking surgical dressings, instruments and laboratory specimens. It also serves as a diluent or vehicle for drugs used for irrigation or other pharmaceutical preparations.
0.9% Sodium Chloride Irrigation USP provides an isotonic saline irrigation identical in composition with 0.9% Sodium Chloride Injection USP (normal saline).
Physiological irrigation solutions are considered generally compatible with living tissues and organs.
Sodium, the major cation of the extracellular fluid, functions primarily in the control of water distribution, fluid balance, and osmotic pressure of body fluids. Sodium is also associated with chloride and bicarbonate in the regulation of the acid-base equilibrium of body fluid.
Chloride, the major extracellular anion, closely follows the metabolism of sodium, and changes in the acid-base balance of the body are reflected by changes in the chloride concentration.
Indication and Usage
NaCl Irrigant
0.9% Sodium Chloride Irrigation USP is indicated for all general irrigation, washing, rinsing and dilution purposes which permit use of a sterile, nonpyrogenic electrolyte solution.
NaCl Irrigant
Contraindications
0.9% Sodium Chloride Irrigation USP is not for injection by usual parenteral routes.
An electrolyte solution should not be used for irrigation during electrosurgical procedures.
Warnings
NaCl Irrigant
FOR IRRIGATION ONLY. NOT FOR INJECTION.
Irrigating fluids have been demonstrated to enter the systemic circulation in relatively large volumes; thus, irrigation solutions must be regarded as systemic drugs. Absorption of large amounts can cause fluid and/or solute overload resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
The risk of dilutional states is inversely proportional to the electrolyte concentrations of the administered parenteral solutions. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentration.
Do not warm above 150°F (66°C).
After opening container, its contents should be used promptly to minimize the possibility of bacterial growth or pyrogen formation.
Discard unused portion of irrigating solution since it contains no preservatives.
Precautions
NaCl Irrigant
General
Use aseptic technique when preparing and administering sterile irrigation solutions.
Use only if solution is clear and container and seal are intact.
Do not use for irrigation that may result in absorption of large amounts of fluid into the blood.
Caution should be observed when the solution is used for continuous irrigation or allowed to "dwell" inside body cavities because of possible absorption into the blood stream and the production of circulatory overload.
When used for irrigation via appropriate irrigation equipment, the administration set should be attached promptly. Unused portions should be discarded and a fresh container of appropriate size used for the start up of each cycle or repeat procedure. For repeated irrigations of urethral catheters, a separate container should be used for each patient.
Laboratory Tests
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance after prolonged irrigation, when fluid absorption is suspected, or whenever the condition of the patient warrants such evaluation.
Drug Interactions
Some additives may be incompatible. Consult with pharmacist.When introducing additives, use aseptic technique.Mix thoroughly.
Do not store.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies with 0.9% Sodium Chloride Irrigation USP have not been performed to evaluate carcinogenic potential, mutagenic potential, or effects on fertility.
Pregnancy
Teratogenic Effects
Animal reproduction studies have not been conducted with 0.9% Sodium Chloride Irrigation USP. It is also not known whether 0.9% Sodium Chloride Irrigation USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. 0.9% Sodium Chloride Irrigation USP should be given to a pregnant woman only if clearly needed.
Labor and Delivery
Safety and effectiveness of 0.9% Sodium Chloride Irrigation USP during labor and delivery have not been established. Caution should be exercised, and the fluid balance, glucose and electrolyte concentrations, and acid-base balance, of both mother and fetus should be evaluated periodically or whenever warranted by the condition of the patient or fetus.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when 0.9% Sodium Chloride Irrigation USP is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of 0.9% Sodium Chloride Irrigation USP in pediatric patients have not been established. Its limited use in pediatric patients has been inadequate to fully define proper dosage and limitations for use.
Geriatric Use
Clinical studies of 0.9% Sodium Chloride Irrigation USP did not include a sufficient number of patients age 65 years and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function.Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.Frequent laboratory determinations and clinical evaluations are recommended to monitor changes in blood glucose, electrolyte concentrations, and renal function.
Adverse Reactions
Possible adverse effects arising from the irrigation of body cavities, tissues, or indwelling catheters and tubes can be minimized when proper procedures are followed. Displaced catheters or drainage tubes can lead to irrigation or infiltration of unintended structures or cavities. Excessive volume or pressure during irrigation of closed cavities may cause undue distension or disruption of tissues. Accidental contamination from careless technique may transmit infection.
If an adverse reaction does occur, discontinue administration of the irrigant, evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
Overdosage
In the event of overhydration or solute overload, reevaluate the patient's condition, and institute appropriate corrective treatment. Intravasular volume overload may respond to hemodialysis. See WARNINGS, PRECAUTIONS, and ADVERSE REACTIONS.
Dosage and Administration
As required for irrigation.
When used as a diluent, or vehicle for other drugs, the drug manufacturer's recommendations should be followed.
Some additives may be incompatible. Consult with pharmacist. When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.
Solutions should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permits.
How Supplied
0.9% Sodium Chloride Irrigation USP is supplied sterile and nonpyrogenic in PIC™ (Plastic Irrigation Container). The 1000 mL and 500 mL containers are packaged 16 per case, the 2000 mL containers are packaged 8 per case, and the 4000 mL containers are packaged 4 per case.
0.9% Sodium Chloride Irrigation USP
NDC Cat. No. REF SIZE
0264-2201-00 R5200-01 1000 mL
0264-2201-10 R5201-01 500 mL
0264-2201-50 R5205-01 2000 mL
0264-2201-70 R5207 ,,,,4000 mL
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.
Do not warm above 150°F (66°C).
DIRECTIONS FOR USE OF PIC™ (PLASTIC IRRIGATION CONTAINER)
Not for injection.
Aseptic technique is required.
Caution – Before use, perform the following checks:
(a) Read the label. Ensure solution is the one ordered and is within the expiration date.
(b) Invert container and inspect the solution in good light for cloudiness, haze, or particulate matter; check the
container for leakage or damage. Any container which is suspect should not be used.
Use only if solution is clear and container and seal are intact
Single unit container. Discard unused portion.
Outer Closure Removal – Grasp the container with one hand and turn the breakaway ring counterclockwise with the other hand until slight resistance is felt. Then, twisting the container in the opposite direction, turn the breakaway ring sharply until the entire outer cap is loose and can be lifted off.
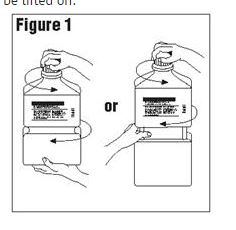
Figure 1
Connect the administration set through the sterile set port according to set instructions or remove screw cap and pour.
[Fig 2]
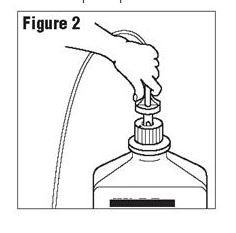
Figure 2
Do not warm above 150°F (66°C) to assure minimal bottle distortion. Keep bottles upright.
Alcohol Wipe
Directions
- clean the affected area
- may be covered with a sterile bandage
- apply wipe to affeted are 1 to 3 times daily
- discard wipe after single use
Alcohol Wipe
Other information
- store at room temperature 15 0 to 25 0 C (59 0 to 77 0F)
- do not use if packet is torn or opened
Ammonia Inhalent
Directions
- hold inhalant away from face and crush ampoule between thumb and forefinger at position indicated on sleeve.
- hold near nostrils for inhalation of volatile vapor
Ammonia Inhalent
Inactive ingredients
alcohol USP, FD&C red #40, lavender oil, lemon oil fcc, nutmeg oil, purified water at room temperature away from light
4381
145225 Kit Contents
1 AMMONIA INHALANTS 10 PER
1 EYE DRESS PKT W/4 ADH STRIPS
1 ADHESIVE TAPE W/P 1" X 10YDS
1 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
2 ELASTIC ROLLED GZ 3" ST
2 ELASTIC ROLLED GZ 4" ST
2 BLOODSTOPPER
8 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 3"X 3" 10'S
1 GZE PADS STERILE 4"X 4" 10'S
6 ABD PADS 8"X10" STERILE
4 MULTI-TRAUMA DRESSING 12"X30"
2 MEDI-RIP BANDAGE 6"X5YDS EA
6 ELASTIC BANDAGE 3" X 4.5YD
4 ELASTIC BANDAGE 6" X 4.5 YD
1 CPR FILTERSHIELD 77-100
1 FLASHLIGHT STD
1 RADIO AM FREQ BATTERY POWERED
1 WATER JEL FACIAL DRS 12X16 EA
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 SOD. CHLORIDE 0.9% 500ML EA
2 INSTA-GLUCOSE 31 GRAMS
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPHYG ANEROID(NO PIN) ADULT
1 STETHESCOPE NURSES VARIOUS COLORS
1 PENLIGHT DISPOSABLE EACH
1 FORCEPS WITH MAGNIFIER
1 SCISSOR UTILITY SHEARS 7-1/4"
1 SCISSOR LISTER BDG S/S 5 1/2"
2 BATTERY - RAYOVAC SIZE AA
2 BATTERY - SIZE D
1 TONGUE BLADES SR. WRAPPED 12'S
1 SAFETY PINS LARGE #3 DOZ
6 SPLINT-CARDBOARD HAND & WRIST
4 SPLINT BOARD W/PAD SML 12"X6"
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 10 PR NITRILE GLVES ZIP BAG
4 EMER.YELLOW BLKT 54"X80" POLYP
1 ANTISEPTIC HAND GEL 4OZ
1 ANTISEPTIC WIPES 20'S ZIP LOCK
1 HYDROCORTISONE 20'S ZIP LOCK
1 ALCOHOL WIPES 50'S ZIP LOCK
1 BAG FOR DELUXE TRAUMA KIT
4 COLD PACK 5"X 9" BULK
2 TRI BNDG NON WOVEN 40"X40"X56"
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
4382
Z145225 Kit Contents
1 AMMONIA INHALANTS 10 PER
1 EYE DRESS PKT W/4 ADH STRIPS
1 ADHESIVE TAPE W/P 1" X 10YDS
1 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
2 ELASTIC ROLLED GZ 3" ST
2 ELASTIC ROLLED GZ 4" ST
2 BLOODSTOPPER
8 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 3"X 3" 10'S
1 GZE PADS STERILE 4"X 4" 10'S
6 ABD PADS 8"X10" STERILE
4 MULTI-TRAUMA DRESSING 12"X30"
2 MEDI-RIP BANDAGE 6"X5YDS EA
6 ELASTIC BANDAGE 3" X 4.5YD
4 ELASTIC BANDAGE 6" X 4.5 YD
1 CPR FILTERSHIELD 77-100
1 FLASHLIGHT STD
1 RADIO AM FREQ BATTERY POWERED
1 WATER JEL FACIAL DRS 12X16 EA
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 SOD. CHLORIDE 0.9% 500ML EA
2 INSTA-GLUCOSE 31 GRAMS
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPHYG ANEROID(NO PIN) ADULT
1 STETHESCOPE NURSES VARIOUS COLORS
1 PENLIGHT DISPOSABLE EACH
1 FORCEPS WITH MAGNIFIER
1 SCISSOR UTILITY SHEARS 7-1/4"
1 SCISSOR LISTER BDG S/S 5 1/2"
2 BATTERY - RAYOVAC SIZE AA
2 BATTERY - SIZE D
1 TONGUE BLADES SR. WRAPPED 12'S
1 SAFETY PINS LARGE #3 DOZ
6 SPLINT-CARDBOARD HAND & WRIST
4 SPLINT BOARD W/PAD SML 12"X6"
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 10 PR NITRILE GLVES ZIP BAG
4 EMER.YELLOW BLKT 54"X80" POLYP
1 ANTISEPTIC HAND GEL 4OZ
1 ANTISEPTIC WIPES 20'S ZIP LOCK
1 HYDROCORTISONE 20'S ZIP LOCK
1 ALCOHOL WIPES 50'S ZIP LOCK
1 BAG FOR DELUXE TRAUMA KIT
4 COLD PACK 5"X 9" BULK
2 TRI BNDG NON WOVEN 40"X40"X56"
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
4393
Z19802 kit contents
1 AMMONIA INHALANTS 10 PER
1 EYE DRESS PKT W/4 ADH STRIPS
1 ADHESIVE TAPE W/P 1" X 10YDS
1 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
2 ELASTIC ROLLED GZ 3" ST
2 ELASTIC ROLLED GZ 4" ST
2 BLOODSTOPPER
8 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 3"X 3" 10'S
1 GZE PADS STERILE 4"X 4" 10'S
6 ABD PADS 8"X10" STERILE
4 MULTI-TRAUMA DRESSING 12"X30"
2 MEDI-RIP BANDAGE 6"X5YDS EA
6 ELASTIC BANDAGE 3" X 4.5YD
4 ELASTIC BANDAGE 6" X 4.5 YD
1 CPR FILTERSHIELD 77-100
1 FLASHLIGHT STD
1 RADIO AM FREQ BATTERY POWERED
1 WATER JEL FACIAL DRS 12X16 EA
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 SOD. CHLORIDE 0.9% 500ML EA
2 INSTA-GLUCOSE 31 GRAMS
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPHYG ANEROID(NO PIN) ADULT
1 STETHESCOPE NURSES VARIOUS COLORS
1 PENLIGHT DISPOSABLE EACH
1 FORCEPS WITH MAGNIFIER
1 SCISSOR UTILITY SHEARS 7-1/4"
1 SCISSOR LISTER BDG S/S 5 1/2"
2 BATTERY - RAYOVAC SIZE AA
2 BATTERY - SIZE D
1 BG POLY 32" x 34"
1 TONGUE BLADES SR. WRAPPED 12'S
1 SAFETY PINS LARGE #3 DOZ
6 SPLINT-CARDBOARD HAND & WRIST
4 SPLINT BOARD W/PAD SML 12"X6"
1 LBL STOCK 4"X2-7/8"
1 LBL CONTS 8"X8",CUSTOM ID B
1 LBL STOCK PLAIN 3.2"x7/8"
1 GRAINGER , DELUXE TRAUMA BAG
2 x5 PR LRG NITRILE GLVES ZIP BAG
4 EMER.YELLOW BLKT 54"X80" POLYP
1 ANTISEPTIC HAND GEL 4OZ
1 ANTISEPTIC WIPES 20'S ZIP LOCK
1 HYDROCORTISONE 20'S ZIP LOCK
1 ALCOHOL WIPES 50'S ZIP LOCK
4 COLD PACK 5"X 9" BULK
2 TRI BNDG NON WOVEN 40"X40"X56"
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
| 4382 FIRST AID KIT
4382 first aid kit |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| 4381 FIRST AID KIT
4381 first aid kit |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| 4393 FIRST AID KIT
4393 first aid kit |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Honeywell Safety Products USA, Inc. (118768815) |