Alcohol Wipe
Directions
- clean the affected area
- apply wipe to affected area 1 to 3 times daily
- may be covered with a sterile bandage
- discard wipe after single use
Aspirin
Active ingredient (in each tablet)
Aspirin 325 mg (NSAID)* *nonsteroidal anti-inflammatory drug
Aspirin
Uses
temporarily reduces fever and relieves minor aches and pains associated with:
- a cold
- headache
- toothache
- muscular aches
- backache
- minor pain of arthritis
- premenstrual and menstrual periods
Aspirin
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:are:
- age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis or kidney disease
- you are taking a diuretic
- you have asthma
Ask a doctor or pharmacist before use if you are
- taking a prescription drug for diabetes, gout or arthritis
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- ringing in the ears or loss of hearing occurs
- any new symptoms appear
If pregnant or breast-feeding,
If pregnant or breat-feeding, ask a health professional before use. It is especially important not to use aspirin during the last three months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Aspirin
Directions
- drink a full glass of water with each dose
- adults and children 12 years of age and older: take 1 or 2 tablets every 4 hours while symptoms last, not more than 12 tablets in 24 hours
- children under 12 years of age: consult a doctor
Aspirin
Other information
- store at room temperature 15° - 30°C (59° - 86°F)
- TAMPER EVIDIENT PACKETS
- DO NOT USE IF OPEN OR TORN
Aspirin
Inactive ingredients
corn starch, croscarmellose sodium*, hypromellose*, microcrystalline cellulose*, mineral oil*, polyethylene glycol*, povidone, propylene glycol, silicon dioxide, stearic acid*, titanium dioxide*
*may contain these ingredients
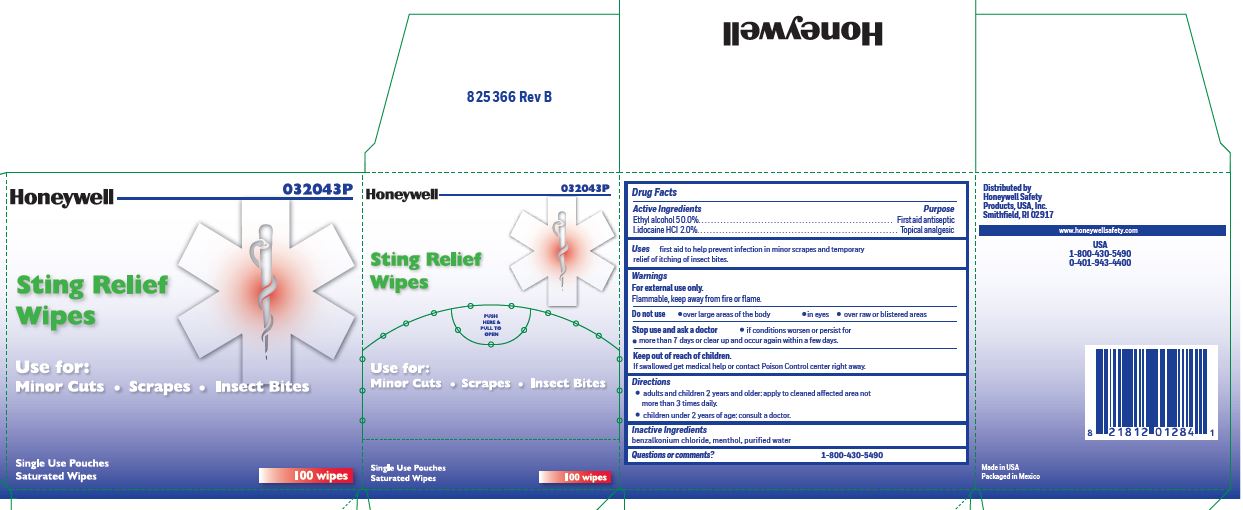
Sting Relief
Uses
- prevent infection in minor scrapes, and temporary relief of itching of insect bites
Sting Relief
Warnings
For external use only
Flammable, keep away from open fire or flame
sting Relief
Directions
- adults and children 2 years and older: Apply to cleaned affected area not more than 3 times daily.
- children under 2 years of age: consult a doctor.
BZK
Warnings
For external use only
Do not use
- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
BZK
Other information
- store at room temperature 15 0 to 30 0 C (59 0 - 86 0 F)
- do not reuse towelette
Neomycin
Active ingredient (each gram contains)
Neomycin sulfate (5 mg equivalent to 3.5 mg Neomycin base)
Neomycin
Warnings
For external use only
Neomycin
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Aypanal EX
Uses
- temporarily relieves minor aches and pains due to the common cold and headache - temporarily reduces fever
Aypanal EX
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount.
- with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If skin reaction occurs, stop use and seek medical help right away
Do Not Use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
Aypanal EX
Directions
- do not take more than directed (see overdose warning)
- adults and children 12 years of age and over: Take 2 tablets with water every 6 hours while symptoms last.
- do not take any more than 8 tablets in 24 hours.
- children under 12: consult a doctor
Aypanal EX
Other information
- store at room temperature 15 0 -30 0 C (59 0 -86 0 F)
- TAMPER EVIDENT- DO NOT USE IF OPEN OR TORN
Aypanal EX
Inactive ingredients
microcrystalline cellulose, povidone, sodium starch glycolate, starch, stearic acid
4342
Z019845 KIT CONTENTS
1 XTRA LRG 2" X 4" AWC
1 FIRST AID GUIDE ASHI
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
3 BZK ANTISEPTIC WIPE, BULK
1 PR LRG NITRILE GLVES ZIP BAG
1 LIGHT STICKS 6" 12 HR
1 TAPE ADHESIVE 1 X 2.5 125135
2 POUCH NEOMYCIN ANTIBIOTIC .9 G
6 WIPE ALCOHOL PREP IPA 70% (DUKAL)
1 KIT, PP 10 UNIT FA
1 SAFETEC STING RELIEF WIPES BULK
10 PLASTIC BANDAGE 3/4" X 3"
2 WOVEN FINGERTIP BANDAGE 2"
1 WOVEN KNUCKLE BANDAGE
1 ASPIRIN BULK 2/PK
1 AYPANAL EXTRA BULK 2/PK
1 BANDAGE, PATCH, ADHESIVE, 2X3