FULL PRESCRIBING INFORMATION
2 DOSAGE AND ADMINISTRATION
The recommended dosage is one drop in the affected eye(s) once daily in the evening. If one dose is missed, treatment should continue with the next dose as normal.
The dosage of Latanoprost Ophthalmic Solution should not exceed once daily; the combined use of two or more prostaglandins, or prostaglandin analogs including Latanoprost Ophthalmic Solution is not recommended. It has been shown that administration of these prostaglandin drug products more than once daily may decrease the intraocular pressure (IOP) lowering effect or cause paradoxical elevations in IOP.
Reduction of the IOP starts approximately 3 to 4 hours after administration and the maximum effect is reached after 8 to 12 hours.
Latanoprost Ophthalmic Solution may be used concomitantly with other topical ophthalmic drug products to lower IOP. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart. Contact lenses should be removed prior to the administration of Latanoprost Ophthalmic Solution, and may be reinserted 15 minutes after administration
5 WARNINGS AND PRECAUTIONS
5.1 Pigmentation
Latanoprost Ophthalmic Solution has been reported to cause changes to pigmented tissues. The most frequently reported changes have been increased pigmentation of the iris, periorbital tissue (eyelid), and eyelashes. Pigmentation is expected to increase as long as latanoprost is administered.
The pigmentation change is due to increased melanin content in the melanocytes rather than to an increase in the number of melanocytes. After discontinuation of latanoprost, pigmentation of the iris is likely to be permanent, while pigmentation of the periorbital tissue and eyelash changes have been reported to be reversible in some patients. Patients who receive treatment should be informed of the possibility of increased pigmentation. Beyond 5 years the effects of increased pigmentation are not known [ see Clinical Studies (14.2)].
Iris color change may not be noticeable for several months to years. Typically, the brown pigmentation around the pupil spreads concentrically towards the periphery of the iris and the entire iris or parts of the iris become more brownish. Neither nevi nor freckles of the iris appear to be affected by treatment. While treatment with Latanoprost Ophthalmic Solution can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly [ see Patient Counseling Information (17)].
5.2 Eyelash Changes
Latanoprost Ophthalmic Solution may gradually change eyelashes and vellus hair in the treated eye; these changes include increased length, thickness, pigmentation, the number of lashes or hairs, and misdirected growth of eyelashes. Eyelash changes are usually reversible upon discontinuation of treatment [ see Patient Counseling Information (17)].
5.3 Intraocular Inflammation
Latanoprost Ophthalmic Solution should be used with caution in patients with a history of intraocular inflammation (iritis/uveitis) and should generally not be used in patients with active intraocular inflammation because inflammation may be exacerbated.
5.4 Macular Edema
Macular edema, including cystoid macular edema, has been reported during treatment with Latanoprost Ophthalmic Solution. Latanoprost Ophthalmic Solution should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema.
5.5 Herpetic Keratitis
Reactivation of Herpes Simplex keratitis has been reported during treatment with Latanoprost Ophthalmic Solution. Latanoprost Ophthalmic Solution should be used with caution in patients with a history of herpetic keratitis. Latanoprost Ophthalmic Solution should be avoided in cases of active herpes simplex keratitis because inflammation may be exacerbated.
5.6 Bacterial Keratitis
There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface [ see Patient Counseling Information (17) ].
6 ADVERSE REACTIONS
The following adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the label:
- Iris pigmentation changes [see Warnings and Precautions (5.1)]
- Eyelid skin darkening [see Warnings and Precautions (5.1)]
- Eyelash changes (increased length, thickness, pigmentation, and number of lashes) [see Warnings and Precautions (5.2)]
- Intraocular inflammation (iritis/uveitis) [see Warnings and Precautions (5.3)]
- Macular edema, including cystoid macular edema [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Latanoprost Ophthalmic Solution was studied in three multicenter, randomized, controlled clinical trials. Patients received 50 mcg/mL Latanoprost Ophthalmic Solution once daily or 5 mg/mL active-comparator (timolol) twice daily. The patient population studied had a mean age of 65±10 years. Seven percent of patients withdrew before the 6-month endpoint.
Table 1: Ocular Adverse Reactions and Ocular Signs/Symptoms Reported by 5–15% of Patients Receiving Latanoprost
|
| Adverse Reactions (incidence (%))
|
|
| Symptom/Finding
| Latanoprost
(n=460) | Timolol
(n=369) |
| Foreign body sensation
| 13
| 8
|
| Punctate keratitis
| 10
| 9
|
| Stinging
| 9
| 12
|
| Conjunctival hyperemia
| 8
| 3
|
| Blurred vision
| 8
| 8
|
| Itching
| 8
| 8
|
| Burning
| 7
| 8
|
| Increased pigmentation of the iris
| 7
| 0
|
Less than 1% of the patients treated with Latanoprost Ophthalmic Solution required discontinuation of therapy because of intolerance to conjunctival hyperemia.
Table 2: Adverse Reactions That Were Reported In 1–5% Of Patients Receiving Latanoprost
|
| Adverse Reactions (incidence (%))
|
|
|
| Latanoprost
(n=460) | Timolol
(n=369) |
| Ocular Events/Signs and Symptoms
|
|
|
| Excessive tearing
| 4
| 6
|
| Eyelid discomfort/pain
| 4
| 2
|
| Dry eye
| 3
| 3
|
| Eye pain
| 3
| 3
|
| Eyelid margin crusting
| 3
| 3
|
| Erythema of the eyelid
| 3
| 2
|
| Photophobia
| 2
| 1
|
| Eyelid edema
| 1
| 3
|
| Systemic Events
|
|
|
| Upper respiratory tract infection/nasopharyngitis/influenza
| 3
| 3
|
| Myalgia / arthralgia/back pain
| 1
| 0.5
|
| Rash/allergic skin reaction
| 1
| 0.3
|
The ocular event/signs and symptoms of blepharitis have been identified as "commonly observed" through analysis of clinical trial data.
6.2 Postmarketing Experience
The following reactions have been identified during postmarketing use of Latanoprost Ophthalmic Solution in clinical practice. Because they are reported voluntarily from a population of unknown size, it is not always possible to reliably estimates their frequency or establish a causal relationship to drug exposure. The reactions, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to Latanoprost Ophthalmic Solution, or a combination of these factors, include:
Nervous System disorders: Dizziness, headache and toxic epidermal necrolysis
Eye Disorders: Eyelash and vellus hair changes of the eyelid (increased length, thickness, pigmentation, and number of eyelashes); keratitis; corneal edema and erosions; intraocular inflammation (iritis/uveitis); macular edema, including cystoid macular edema; trichiasis; periorbital and lid changes resulting in deepening of the eyelid sulcus; iris cyst; eyelid skin darkening ; localized skin reaction on the eyelids; conjunctivitis; pseudopemphigoid of the ocular conjunctiva.
Respiratory, Thoracic and Mediastinal Disorders: Asthma and exacerbation of asthma; dyspnea
Skin and Subcutaneous Tissue Disorders: Pruritus
Infections and Infestations: Herpes keratitis
Cardiac Disorders: Angina; palpitations; angina unstable
General Disorders and Administration Site Conditions: Chest pain
7 DRUG INTERACTIONS
In vitro studies have shown that precipitation occurs when eye drops containing thimerosal are mixed with Latanoprost Ophthalmic Solution. If such drugs are used, they should be administered at least five (5) minutes apart.
The combined use of two or more prostaglandins, or prostaglandin analogs including Latanoprost Ophthalmic Solution is not recommended. It has been shown that administration of these prostaglandin drug products more than once daily may decrease the IOP lowering effect or cause paradoxical elevations in IOP.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C.
Reproduction studies have been performed in rats and rabbits. In rabbits, an incidence of 4 of 16 dams had no viable fetuses at a dose that was approximately 80 times the maximum human dose, and the highest nonembryocidal dose in rabbits was approximately 15 times the maximum human dose.
There are no adequate and well-controlled studies in pregnant women. Latanoprost Ophthalmic Solution should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
10 OVERDOSAGE
Intravenous infusion of up to 3 mcg/kg in healthy volunteers produced mean plasma concentrations 200 times higher than during clinical treatment and no adverse reactions were observed. Intravenous dosages of 5.5 to 10 mcg/kg caused abdominal pain, dizziness, fatigue, hot flushes, nausea, and sweating.
If overdosage with Latanoprost Ophthalmic Solution occurs, treatment should be symptomatic.
11 DESCRIPTION
Latanoprost is a prostaglandin F 2α analogue. Its chemical name is isopropyl-(Z)-7[(1R,2R,3R,5S)3,5-dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl]-5-heptenoate. Its molecular formula is C 26H 40O 5 and its chemical structure is:
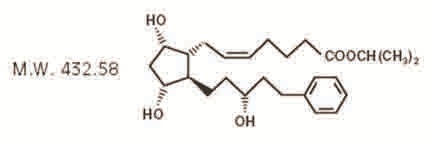
Latanoprost is a colorless to slightly yellow oil that is very soluble in acetonitrile and freely soluble in acetone, ethanol, ethyl acetate, isopropanol, methanol, and octanol. It is practically insoluble in water.
Latanoprost Ophthalmic Solution 0.005% is supplied as a sterile, isotonic, buffered aqueous solution of latanoprost with a pH of approximately 6.7 and an osmolality of approximately 267 mOsmol/kg. Each mL of Latanoprost Ophthalmic Solution contains 50 mcg of latanoprost. Benzalkonium chloride, 0.02% is added as a preservative. The inactive ingredients are: sodium chloride, sodium dihydrogen phosphate monohydrate, disodium hydrogen phosphate anhydrous, and water for injection. One drop contains approximately 1.5 mcg of latanoprost.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Latanoprost is a prostanoid selective FP receptor agonist that is believed to reduce the intraocular pressure (IOP) by increasing the outflow of aqueous humor. Studies in animals and man suggest that the main mechanism of action is increased uveoscleral outflow. Elevated IOP represents a major risk factor for glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss.
12.2 Pharmacodynamics
Reduction of the IOP in man starts about 3–4 hours after administration and maximum effect is reached after 8–12 hours. IOP reduction is present for at least 24 hours.
12.3 Pharmacokinetics
Latanoprost is absorbed through the cornea where the isopropyl ester prodrug is hydrolyzed to the acid form to become biologically active.
Distribution
The distribution volume in humans is 0.16 ± 0.02 L/kg. The acid of latanoprost can be measured in aqueous humor during the first 4 hours, and in plasma only during the first hour after local administration. Studies in man indicate that the peak concentration in the aqueous humor is reached about two hours after topical administration.
Metabolism
Latanoprost, an isopropyl ester prodrug, is hydrolyzed by esterases in the cornea to the biologically active acid. The active acid of latanoprost reaching the systemic circulation is primarily metabolized by the liver to the 1,2-dinor and 1,2,3,4-tetranor metabolites via fatty acid β-oxidation.
Excretion
The elimination of the acid of latanoprost from human plasma is rapid (t 1/2 = 17 min) after both intravenous and topical administration. Systemic clearance is approximately 7 mL/min/kg. Following hepatic β-oxidation, the metabolites are mainly eliminated via the kidneys. Approximately 88% and 98% of the administered dose are recovered in the urine after topical and intravenous dosing, respectively.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Latanoprost was not carcinogenic in either mice or rats when administered by oral gavage at doses of up to 170 mcg/kg/day (approximately 2800 times the recommended maximum human dose) for up to 20 and 24 months, respectively.
Latanoprost was not mutagenic in bacteria, in mouse lymphoma, or in mouse micronucleus tests. Chromosome aberrations were observed in vitro with human lymphocytes. Additional in vitro and in vivo studies on unscheduled DNA synthesis in rats were negative.
Latanoprost has not been found to have any effect on male or female fertility in animal studies.
14 CLINICAL STUDIES
14.1 Elevated Baseline IOP
Patients with mean baseline IOP of 24 – 25 mmHg who were treated for 6 months in multi-center, randomized, controlled trials demonstrated 6 – 8 mmHg reductions in IOP. This IOP reduction with Latanoprost Ophthalmic Solution 0.005% dosed once daily was equivalent to the effect of timolol 0.5% dosed twice daily.
14.2 Progression of Increased Iris Pigmentation
A 3-year open-label, prospective safety study with a 2-year extension phase was conducted to evaluate the progression of increased iris pigmentation with continuous use of Latanoprost Ophthalmic Solution once-daily as adjunctive therapy in 519 patients with open-angle glaucoma. The analysis was based on observed-cases population of the 380 patients who continued in the extension phase.
Results showed that the onset of noticeable increased iris pigmentation occurred within the first year of treatment for the majority of the patients who developed noticeable increased iris pigmentation. Patients continued to show signs of increasing iris pigmentation throughout the five years of the study. Observation of increased iris pigmentation did not affect the incidence, nature, or severity of adverse events (other than increased iris pigmentation) recorded in the study. IOP reduction was similar regardless of the development of increased iris pigmentation during the study.
16 HOW SUPPLIED/STORAGE AND HANDLING
Latanoprost Sterile Ophthalmic Solution is a clear, isotonic, buffered, preserved colorless solution of latanoprost 0.005% (50 mcg/mL).
It is supplied as a 2.5 mL solution filled in a 5 mL translucent low density polyethylene bottle with insert cap assembly comprising of a turquoise coloured, high density polyethylene screw cap over a low density polyethylene nozzle with tamper evident Low density polyethylene dust cover sealing the bottle cap.
2.5 mL fill, 0.005% (50 mcg/mL) : Package of 1 bottle : NDC: 55545-1010-1
Storage: Protect from light. Store unopened bottle(s) under refrigeration at 2° to 8°C (36° to 46°F). During shipment to the patient, the bottle may be maintained at temperatures up to 40°C (104°F) for a period not exceeding 8 days. Once a bottle is opened for use, it may be stored at room temperature up to 25°C (77°F) for 6 weeks.
17 PATIENT COUNSELING INFORMATION
Advise patients about the potential for increased brown pigmentation of the iris, which may be permanent. Inform patients about the possibility of eyelid skin darkening, which may be reversible after discontinuation of Latanoprost Ophthalmic Solution [see Warnings and Precautions (5.1)].
Inform patients of the possibility of eyelash and vellus hair changes in the treated eye during treatment with Latanoprost Ophthalmic Solution. These changes may result in a disparity between eyes in length, thickness, pigmentation, number of eyelashes or vellus hairs, and/or direction of eyelash growth. Eyelash changes are usually reversible upon discontinuation of treatment.
Instruct patients to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures because this could cause the tip to become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions [see Warnings and Precautions (5.6)].
Advise patients that if they develop an intercurrent ocular condition (e.g., trauma or infection) or have ocular surgery, or develop any ocular reactions, particularly conjunctivitis and eyelid reactions, they should immediately seek their physician's advice concerning the continued use of the multiple-dose container.
Advise patients that Latanoprost Ophthalmic Solution contains benzalkonium chloride, which may be absorbed by contact lenses. Contact lenses should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following administration of Latanoprost Ophthalmic Solution.
Use with Other Ophthalmic Drugs
If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart.
Made in India by:
FDC Limited
Distributed by:
Rising Pharmaceuticals, Inc.
Saddle Brook, NJ 07663
Revised: 11/2019
Latanoprost Ophthalmic Solution
Before using your latanoprost ophthalmic solution
Before using your latanoprost hydrochloride ophthalmic solution for the first time, be sure the dust cover seal is unbroken (See figure A)
Step 1. Wash hands before each use.
Step 2. Snap off the dust cover by turning it clockwise to break the seal. See figure B
Step 3. Pull off the dust cover see figure C
Step 4.Unscrew the turquoise colored cap by turning in the counterclockwise direction. See figure D
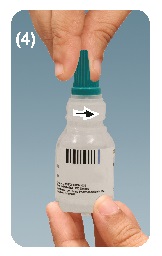
THE INSERT TIP IS DESIGNED TO DELIVER PREMEASURED DROP. THEREFORE, DO NOT ENLARGE THE HOLE OF THE INSERT TIP OR TAMPER WITH INSERT TIP.
Giving your Latanoprost Ophthalmic Solution
Step 5. Tilt your head back and pull the lower eyelid down slightly to form pocket between your eyelid and your eye. Dispense drops with gentle pressure. Do not touch your eye or eyelid with the dropper tip. See figure E.
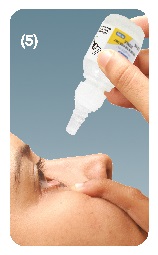
Step 6. If your doctor has told you to use Latanoprost Ophthalmic Solution in both eyes, repeat step 4 and step 5.
After using your Latanoprost Ophthalmic Solution
Step 7. Recap the bottle after every use; tighten the cap on the nozzle. See figure F
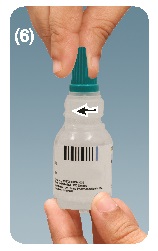
This Instruction for Use has been approved by U.S. Food and Drug Administration.
Made in India by:
FDC Limited
Distributed by:
Rising Pharmaceuticals, Inc
Saddle Brook, NJ 07663
Revised: 11/2019
