Burn Jel
Warnings
For external use only
Burn JEl
Directions
- adults and children 2 years of age and older; apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
- you may report a serious reaction to this product to 800-430-5490
Burn Jel
Inactive ingredients
carbopol 940, carbopol 1342, diazolidinyl urea, glycerin, melaleuca alternifolia (tea tree) leaf oil, methylparaben, octoxynol-9, propylene glycol, propylparaben, trolamine, water
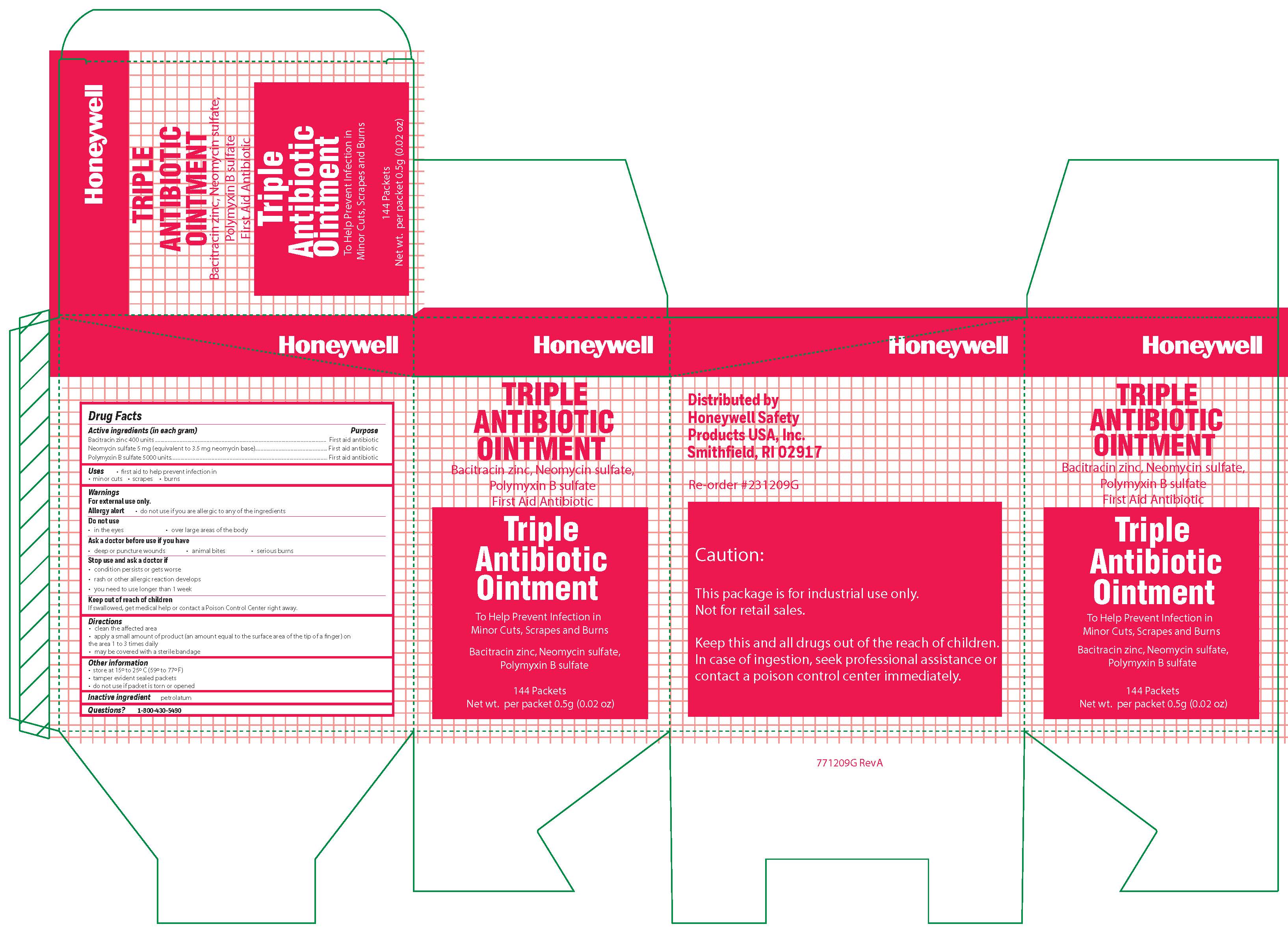
Triple
Active ingredient
Bacitracin zinc 400 units
Neomycin sulfate (5 mg equivalent to 3.5 mg Neomycin base)
Polymyxin B sulfate 5000 units
Triple
Warnings
For external use only
Allergy alert: do not use if you are allergic to any of the ingredients
Do not use
- in the eyes
- over large areas of the body
- Ask a doctor before use if you have
- a deep or puncture wounds
- animal bites
- serious burns
Triple
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Triple
Other information
- store at 15 0 to 25 0 C (59 0 to 77 0 F)
- tamper evident sealed packets
- do not use if packet is torn or opened
Sting Relief
Uses
- prevent infection in minor scrapes, and temporary relief of itching of insect bites
Sting Relief
Warnings
For external use only
Flammable, keep away from open fire or flame
Sting Relief
Directions
- adults and children 2 years and older: Apply to cleaned affected area not more than 3 times daily.
- children under 2 years of age: consult a doctor.
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only
- Obtain immediate medical treatment for all open wounds in or near eyes.
- To avoid contamination, do not touch tip of container to any surface.
- Do not reuse.
- Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Eyeash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
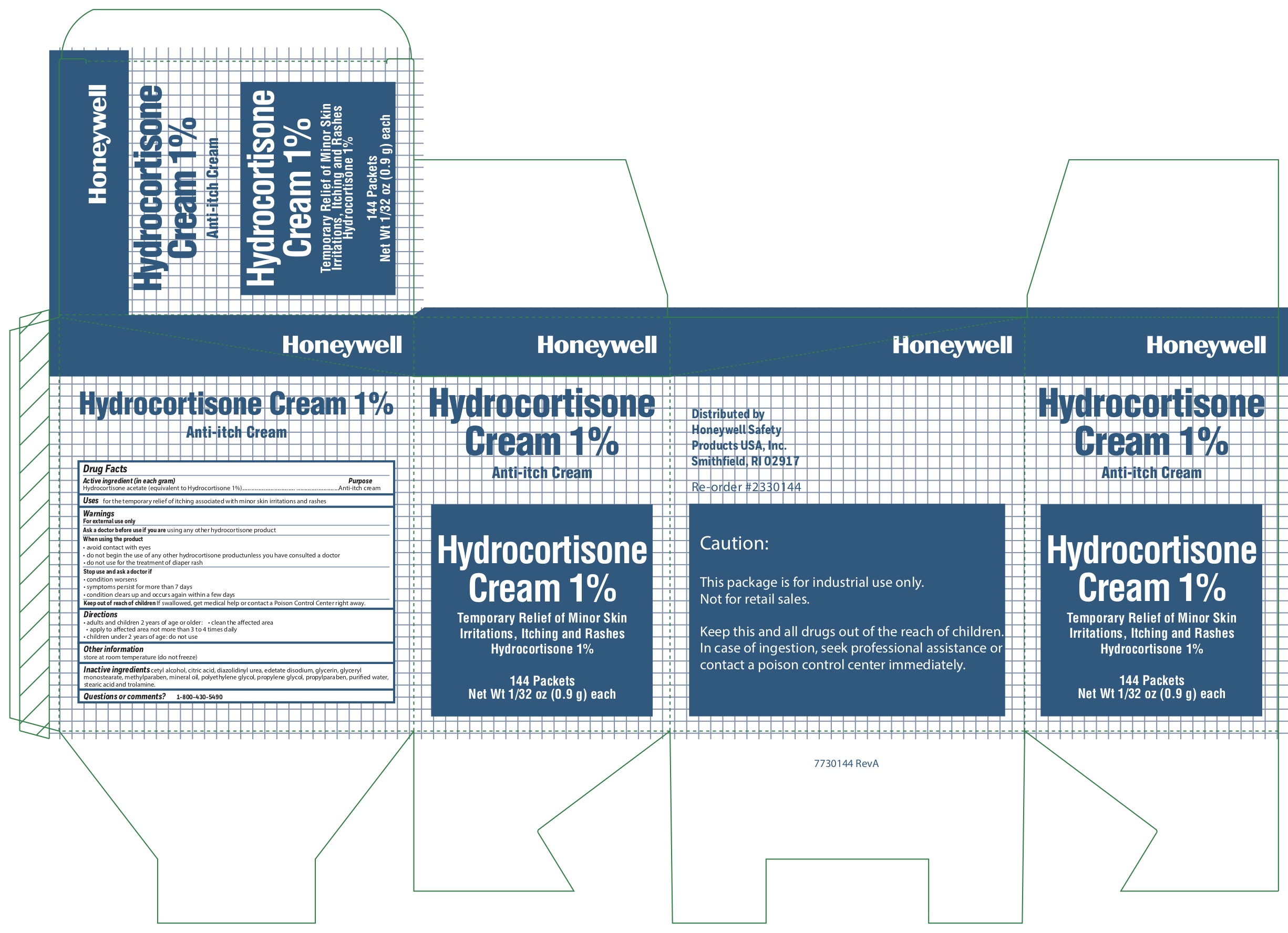
Hydrocortisone
Active ingredient (in each gram)
Hydrocortisone acetate (equivalent to Hydrocortisone 1%)
Hydrocortisone
Uses
- for the temporary relief of itching associated with minor skin irritations and rashes
Hydrocortisone
Warnings
For external use only
When using the product
- avoid contact with eyes
- do not begin use of any other hydrocortisone product unless you have consulted a doctor
- do not use for the treatment of diaper rash
Hydrocortisone
Directions
- adults and children 2 years and older:
- clean the affected area
- apply to the area not more than 3 to 4 times daily
- children under 2 years of age: consult a doctor
Hydrocortisone
Inactive ingredients
cetyl alcohol, citric acid, diazolidinyl urea, edetate disodium, glycerin, glyceryl monostearate, methylparaben, mineral oil, polyethylene glycol, propylene glycol, propylparaben, purified water, stearic acid, trolamine
PVP
Warnings
For external use only.
PVP
Directions
- clean the affected area
- apply1 to 3 times daily
- may be covered with a sterile bandage
- if bandaged, let dry first
- discard wipe after single use
PVP
Other information
- do not use on individuals who are allergic or sensitive to iodine
- store at controlled temperature 59-86ºF (15-30ºC)
- do not use if pouch is open or torn
BZK
Warnings
For external use only
BZK
Other information
- store at room temperature 15 0 to30 0 C (59 0 - 86 0 F)
- do not reuse towelette
4318
SF00004559 Kit Contents
1 TRIPLE ANTIBIOTIC 10 PER
1 TRIANGULAR BDG, NON-STERILE
1 GAUZE PADS, 3" X 3", 4 PER
1 ADH TAPE, .5" X 2.5 YD, 2 PER
1 GAUZE COMP, 1 SQ YARD, 1 PER
1 ADHESIVE BDG,PLSTIC,1"X3"16PER
1 1 OZ EYE WASH W/PADS & STRIPS
1 BURN JEL 1/8 OZ, 6 PER
1 HYDROCORTISON,1.O%,1/32 OZ,10P
1 PVP IODINE WIPES 10 PER
1 BIOHAZARD BAG/SCRAPER BBP
1 ANTIMCRBL ANTSPTC TWLETTS 6PER
1 FIRST AID GUIDE ASHI
1 MICROSHIELD BAGGED 72-151
LBL STOCK 6-3/8"X4"
1 LBL STOCK 3"x1-7/8"
2 PR LRG NITRILE GLVES ZIP BAG
1 KIT STL 16 UN (HORIZONTAL)
1 STING WIPES 10
4319
SF00004560 kit contents
1 TRIPLE ANTIBIOTIC 10 PER
1 TRIANGULAR BDG, NON-STERILE
1 GAUZE PADS, 3" X 3", 4 PER
1 ADH TAPE, .5" X 2.5 YD, 2 PER
1 GAUZE COMP, 1 SQ YARD, 1 PER
1 ADHESIVE BDG,PLSTIC,1"X3"16PER
1 ADH BAND, EXTRA LARGE, 6 PER
1 1 OZ EYE WASH W/PADS & STRIPS
1 BURN JEL 1/8 OZ, 6 PER
1 WATER JEL DRESSING 4" X 4"
1 WATER JEL DRESSING,2" X 6"
1 HYDROCORTISON,1.O%,1/32 OZ,10P
1 PVP IODINE WIPES 10 PER
1 STING RELIEF WIPES 10 PER BOX
1 BIOHAZARD BAG/SCRAPER BBP
1 ANTIMCRBL ANTSPTC TWLETTS 6PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
1 MICROSHIELD BAGGED 72-151
1 BANDAGE COMP 4" W/TELFA PAD 1
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
2 PR LRG NITRILE GLVES ZIP BAG
1 WATER JEL BURN DRESSING 4 X 16
1 KIT STL 36 UN WHT 01 HOR SHELF
1 COLD PACK UNIT 4"X6" BULK
1 TELFA PADS 2"X 3" 4