4246 FIRST AID KIT- 4246 first aid kit
Honeywell Safety Products USA, INC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
0498-4246: First Aid Kit (Burn Jel, alcohol wipes, HC cr, Burn Relief pump, PVP wipes, EW, ASA, amm. Inh, Foille Burn, Aypanal - 340420FP)
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only Obtain immediate medical treatment for all open wounds in or near eyes. To avoid contamination, do not touch tip of container to any surface. Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Eyewash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
Aspirin
Active ingredient (in each tablet)
Aspirin 325 mg (NSAID)* *nonsteroidal anti-inflammatory drug
Aspirin
Uses
temporarily reduces fever and relieves minor aches and pains associated with:
- a cold
- headache
- toothache
- muscular aches
- backache
- minor pain of arthritis
- premenstrual and menstrual periods
Aspirin
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:are:
- age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis or kidney disease
- you are taking a diuretic
- you have asthma
Ask a doctor or pharmacist before use if you are
- taking a prescription drug for diabetes, gout or arthritis
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- ringing in the ears or loss of hearing occurs
- any new symptoms appear
If pregnant or breast-feeding,
If pregnant or breat-feeding, ask a health professional before use. It is especially important not to use aspirin during the last three months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Aspirin
Directions
- drink a full glass of water with each dose
- adults and children 12 years of age and older: take 1 or 2 tablets every 4 hours while symptoms last, not more than 12 tablets in 24 hours
- children under 12 years of age: consult a doctor
Aspirin
Other information
- store at room temperature 15° - 30°C (59° - 86°F)
- TAMPER EVIDIENT PACKETS
- DO NOT USE IF OPEN OR TORN
Aspirin
Inactive ingredients
corn starch, croscarmellose sodium*, hypromellose*, microcrystalline cellulose*, mineral oil*, polyethylene glycol*, povidone, propylene glycol, silicon dioxide, stearic acid*, titanium dioxide*
*may contain these ingredients
Ammonia
Directions
- hold inhalant away from face and crush ampoule between thumb and forefinger at position indicated on sleeve.
- hold near nostrils for inhalation of volatile vapor
Ammonia
Inactive ingredient
alcohol USP, FD&C red #40, lavender oil, lemon oil fcc, nutmeg oil, purified water
Burn Jel
Directions
- adults and children 2 years of age and older; apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
- you may report a serious reaction to this product to 800-430-5490
Burn Jel
Inactive ingredients
carbopol 940, carbopol 1342, diazolidinyl urea, glycerin, melaleuca alternifolia (tea tree) leaf oil, methylparaben, octoxynol-9, propylene glycol, propylparaben, trolamine, water
Alcohol Wipes
Directions
- clean the affected area
- may be covered with a sterile bandage
- apply wipe to affeted are 1 to 3 times daily
- discard wipe after single use
Alcohol Wipes
Other information
- store at room temperature 15 0 to 25 0 C (59 0 to 77 0 F)
- do not use if packet is torn or opened
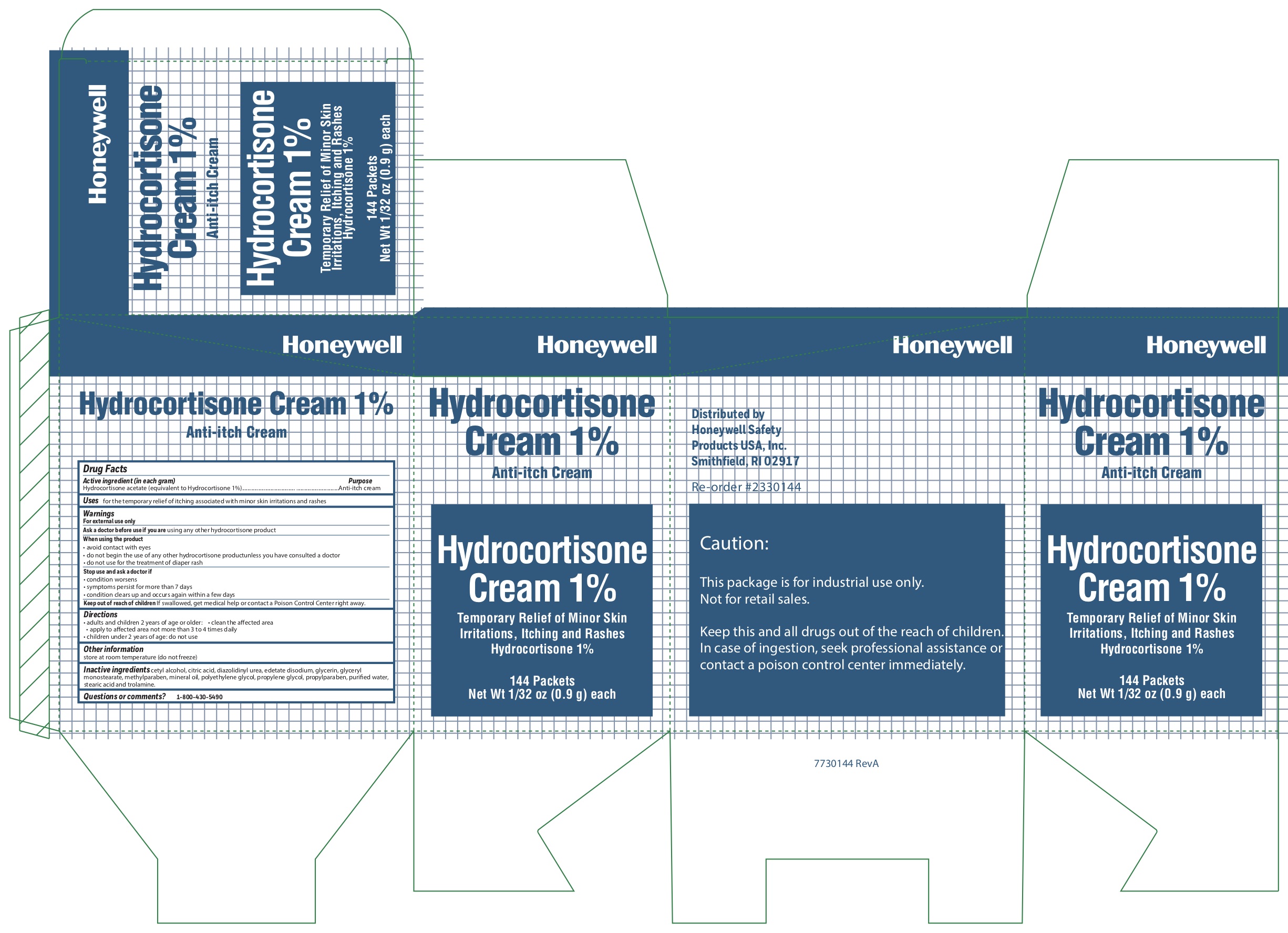
Hydrocortisone
Acitive ingredient (in each gram)
Hydrocortisone acetate (equivalent to Hydrocortisone 1%)
Hydrocortisone
Uses
for the temporary relief of itching associated with minor skin irritations and rashes
Hydrocortisone
Warnings
For external use only
When using the product
- avoid contact with eyes
- do not begin use of any other hydrocortisone product unless you have consulted a doctor
- do not use for the treatment of diaper rash
Hydrocortisone
Directions
- adults and children 2 years and older
- clean the affected area
- apply to the area not more than 3 to 4 times daily
- chil :dren under 2 years of age: consult a doctor
Hydrocortisone
Inactive ingredients
cetyl alcohol, citric acid, diazolidinyl urea, edetate disodium, glycerin, glyceryl monostearate, methylparaben, mineral oil, polyethylene glycol, propylene glycol, propylparaben, purified water, stearic acid, trolamine
PVP Wipe
Warnings
For external use only
PVP Wipe
Directions
Reverse cardboard sleeve, then crush at dot between thumb and forefinger. Allow solution to saturate tip and apply solution to injury.
- clean affected area
- apply to affected area 1 to 3 times daily
- may be covered with a sterile bandage
- discard swab after single use
PVP
Other information
- store at room temperature away from light
- keep from freezing or excessive heat
- do not use if package is torn or open
Burn Relief Spray
Warnings
For external use only
Burn Relief
Directions
adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
children under 2 years of age: ask a doctor
Burn Reelief Spray
Inactive ingredients
diazolidinyl urea, edetate disodium, glycerin, hypromellose, methylparaben, octoxynol 9, propylene glycol, propylparaben, purified water, tea tree oil, trolamine
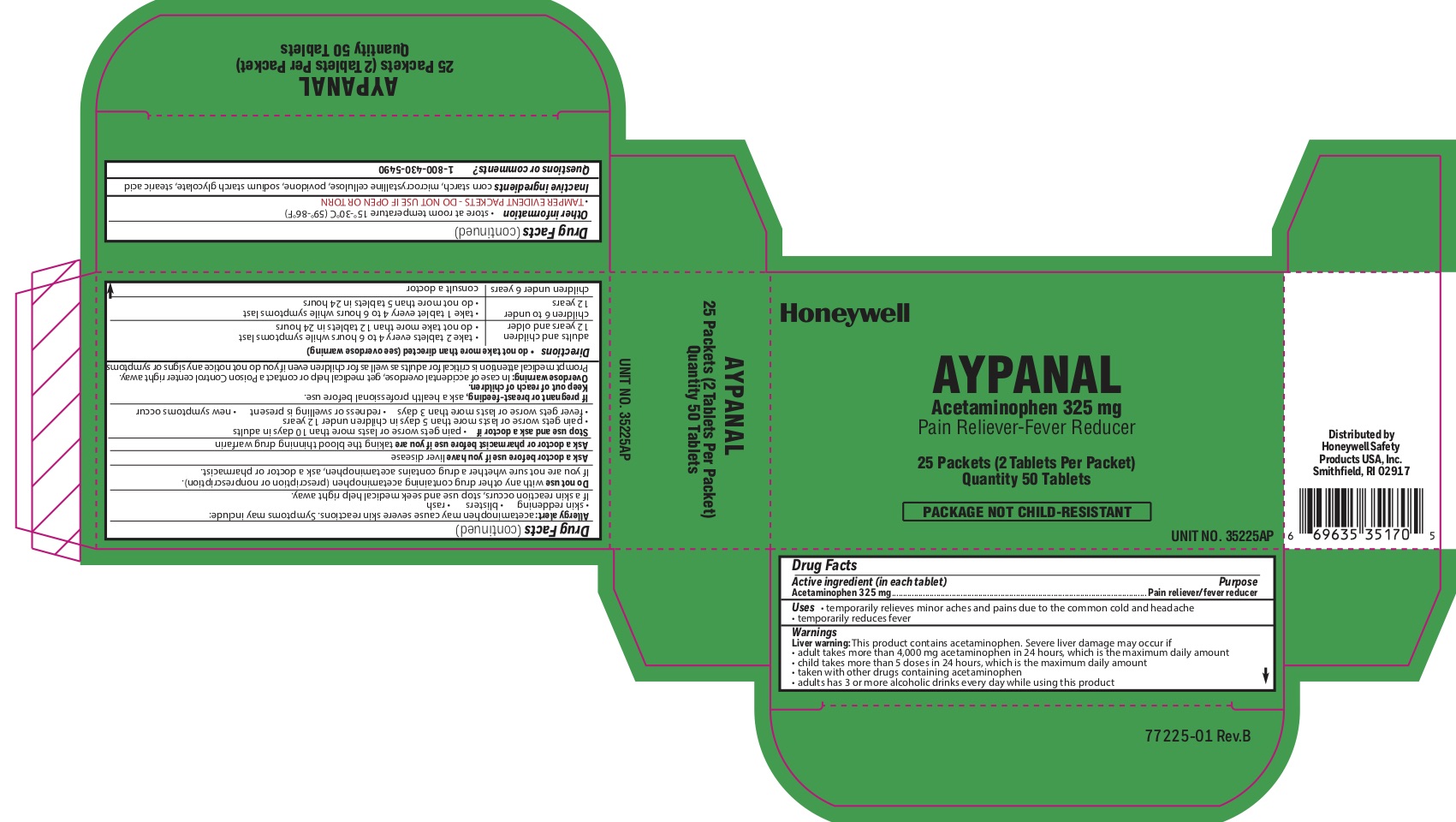
Aypanal
Uses
- temporarily relieves minor aches and pains due to the common cold and headache
- temporarily reduces fever
Aypanal
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount
- child takes more than 5 doses in 24 hours, which is the maximum daily amount
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin rash occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
Aypanal
Directions
- do not take more than directed (see overdose warning)
- adults and children 12 years of age or older
- take two tablets every 4-6 hours while symptoms last
- do not take more than 12 tablets in 24 hours
- children 6 to under 12 years of age
- take 1 tablet every 4-6 hours while symptoms last
- do not take more than 5 tablets in 24 hours
- children under 6 years
- consult a doctor
Aypanal
Other information
- store at room temperature 15 0 to 30 0 C (59 0 - 86 0 F)
- TAMPER EVIDENT PACKETS- DO NOT USE IF OPEN OR TORN
Aypanal
Inactive ingredients
corn starch, microcrystalline cellulose, povidone, sodium starch glycolate, stearic acid
Foille Ointment
Uses
- For the temporary relief of pain associated with burns, sunburn, minor cuts, scrapes, insect bites, and minor skin irritations.
- First aid to help prevent infection in minor cuts, scrapes and burns.
Foille Ointment
Warnings
For external use only.
Foille Ointment
Directions
- clean the affected area.
- adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
- children under 2 years of age: consult a physician.
Foille Oinatment
Inactive ingredients
beeswax, benzyl alcohol, calcium disodium EDTA, calcium hydroxide, ceresin, eugenol, hydrogenated vegetable oil, maleic anhydride, mono- and di-glycerides, PEG-32, purified water, sodium borate, sodium lauryl sulfate, zea mays (corn) oil.
4246
68100REC Kit Contents
1 3/4 X 3 PLAS 100/BOX
1 1X3 PLASTIC 100/BOX
1 KING SIZE 2 X 3 PLASTIC 50/BX
1 AMMONIA INHALANTS 10 PER
1 GAUZE COMPRESS, 1728 SQ IN 1
4 INSTANT COLD PACK 4" X 6"
3 BANDAGE COMP, 4" OFFSET, 1 PER
1 BURN JEL 1/8 OZ, 6 PER
1 ALCOHOL PREP PADS 10P
1 HYDROCORTISON,1.O%,1/32 OZ,10P
2 ADHESIVE TAPE W/P 1/2"X 5 YD
2 O/H PUMP BURN RELIEF 2 OZ ID G
2 GAUZE BANDAGE 1" x 2 YDS
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
6 TAPE ADHESIVE 1"X 5 YD PLSTC
2 GZE PADS STERILE 2"X 2" 10'S
2 GZE PADS STERILE 3"X 3" 10'S
2 GZE PADS STERILE 2"X 2" 25'S
5 CO-FLEX BAND 1"X5YD TAN 2/PACK
2 CO-FLEX BANDAGE 4"X5YDS TAN
1 PVP PREP PADS MEDIUM 100/BX
1 ASPIRIN IND PK 5 GR 2/ENV 100
1 212 4 OZ TUBE SKIN COND, 1 EA ID P
2 1 OZ, BUFF EYEWASH
1 SCISSOR BDGE 4" RED PLS HDL
1 KIT TWEEZER 3 1/2" SLANTED
1 180 EMPTY BLANK NO LOGO
1 TONGUE BLADES SR WRAPPED 6'S
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
2 PR LRG NITRILE GLVES ZIP BAG
2 SELF-ADH WRAP 3 X 5 YDS NORTH REV E
2 TRI BNDG NON WOVEN 40"X40"X56"
2 FOILLE BURN/F A OINT 1/2 OZ
8 EYE PADS STD OVAL STERILE
5 GAUZE PADS 4"X4" 12PLY
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
1 TRNQT W/WEB STP&BKL 1
1 AYPANAL NON-ASP 25/2
| 4246 FIRST AID KIT
4246 first aid kit kit |
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
| Labeler - Honeywell Safety Products USA, INC (118768815) |