KALI ARSENICOSUM- potassium arsenite anhydrous pellet
Boiron
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
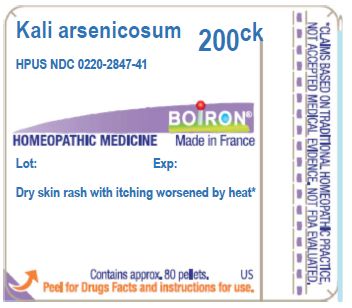
Kali arsenicosum 200CK
(**contains 0.443 mg of the active ingredient per pellet)
Dry skin rash with itching worsened by heat*
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen
If pregnant or breast-feeding ask a health professional before use
Keep out of reach of children
Do not use if pellet dispenser seal is broken.
Contains approx 80 pellets.
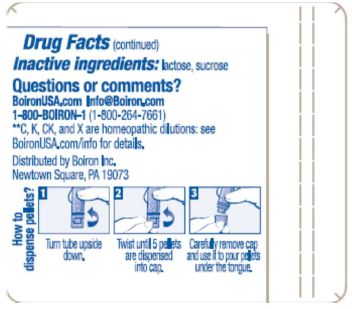
How to dispense pellets? Turn tube upside down. Twist until 5 pellets are dispensed into cap. Carefully remove the cap and use it to pour pellets under the tongue.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.
Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
1-800-BOIRON-1 (1-800-264-7661),
BoironUSA.com Info@boiron.com
Distributed by Boiron, Inc. Newtown Square, PA 19073