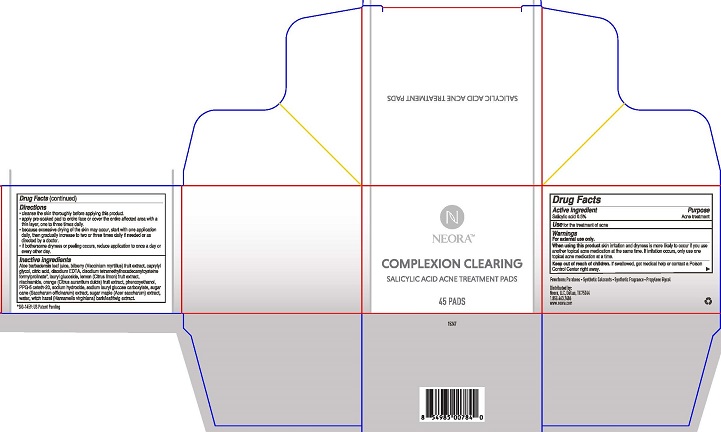
When using this product skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- cleanse the skin thoroughly before applying this product.
- apply pre-soaked pad to entire face or cover the entire affected area with a thin layer, one to three times daily.
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor.
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day.
Inactive ingredients Aloe barbadensis leaf juice, bilberry (Vaccinium myrtillus) fruit extract, caprylyl glycol, citric acid, disodium EDTA, disodium tetramethylhexadecenylcysteine formylprolinate, lauryl glucoside, lemon (Citrus limon) fruit extract, niacinamide, orange (Citrus aurantium dulcis) fruit extract, phenoxyethanol, PPG-5 ceteth-20, sodium hydroxide, sodium lauryl glucose carboxylate, sugar cane (Saccharum officinarum) extract, sugar maple (Acer saccharum) extract, water, witch hazel (Hamamelis virginiana) bark/leaf/twig extract.