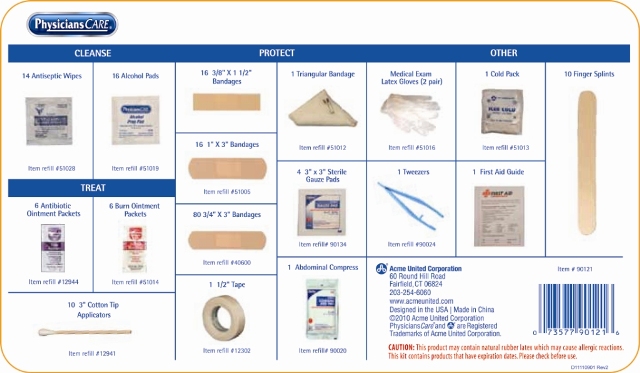
Back Label
Physicians Care
CLEANSE
Antiseptics
14 Antiseptic Wipes
16 Alcohol Pads
TREAT
6 Antibiotic Ointment Packets
6 Burn Ointment Packets
10 3" Cotton Tip Applicator
16 3/8” x 1 1/2” Bandages
16 1” x 3” Bandages
80 3/4” x 3” Bandages
1 1/2" Tape
1 Triangular Bandage
4 3" x 3" Sterile Gauze Pads
1 Abdominal Compress
Other
Medical Exam Latex Gloves (2 Pair)
1 Tweezers
1 Cold Pack
1 First Aid Guide
10 Finger Splints
Acme United Corporation
60 Round Hill Road
Fairfield CT 06824
www.acmeunited.com
Designed in the USA | Made in China
CAUTION: This product may contain natural rubber latex which may cause allergic reactions.
This kit contains products that have expiration dates. Please check before use.
BZK Towelette Labeling
Reorder 855
NDC 65517-0004-1
Dukal
BZK TOWELETTE
Contains Benzalkonium Chloride
For External Use Only
Helps Prevent Infection
1 / Pouch
DUKAL CORPORATION
(631) 656-3800
Ronkonkoma, NY 11779 www.dukal.com
Made in China
Drug Facts
Active Ingredients..................... Benzalkonium Chloride 0.133% w/v
Purpose................................. First Aid Antiseptic
USE: Antiseptic Cleansing of face, hands and
body without soap and water. Airs dries in seconds
DO NOT USE: in the eyes or apply over large
areas of the body.
STOP USE: If irritation, redness or other symptoms
develop. Consult a doctor if the conditions persists
or gets worse.
CAUTION: Keep out of reach of children. If
swallowed get medical help or contact a Poison
Control Center right away.
DIRECTIONS: Tear open packet, unfold and use
as a washcloth.
INACTIVE INGREDIENTS: Distilled Water
Prep Pad Labeling
Reorder 852
Dukal Corporation
NDC 65517-0001-1
NPN 80003156
ALCOHOL PREP PAD
Saturated with 70% Isopropyl Alcohol
For External Use Only
1 / Pouch
Dukal Corporation
Ronkonkoma, NY 11779
631-656-3800
www.dukal.com
Made in China
Drug Facts
Active Ingredients
Isopropyl Alcohol 70%
Purpose
Antiseptic CleanserUse
For Preparation of Skin prior to an injectionWarnings
- For External Use Only
- Flammable, Keep away from fire or flame
Do Not Use
- with electrocautery procedures
- In the Eyes. If contact occurs, flush eyes with water
Stop Use
If irritation and redness develop. If condition persists, consult your health care practitioner.Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.Directions
Wipe injection site vigorously and discard.Other Information
Store at Room Temperature 15 - 30 C (59 - 86 F)Inactive Ingredient
purified waterTriple Antibiotic Labeling
WaterJel
Triple Antibiotic
First Aid Ointment
To Help Prevent Infection
Each Gram contains
Bacitracin Zinc 400 units
Neomycin Sulphate 5 mg
(equivalent to 3.5 mg Neomycin base)
Polymyxin B Sulfate 5000 units
Water-Jel Technologies
Carlstadt, NJ 07072
Drug Facts
Uses to help prevent infection in
minor cuts, scrapes, burns
Warnings
For external use only
Do not use
in the eyes or apply over large areas of the body
If you are allergic to any of the ingredients
longer than 1 week unless directed by a doctor
Stop use and ask a doctor if
the condition persists or gets worse
a rash or other allergic reaction develops
Keep out of reach of children
if ingested contact Poison Control Center right away
Directions
clean affected area apply a small amount of product
(an amount equal to the surface area of the tip of a finger)
on the area 1 to 3 times daily may be covered with a sterile bandage
First Aid Burn Cream Labeling
WaterJel
First Aid Burn Cream
Antiseptic Pain Relief with Aloe
Active Ingredients:
Benzalkonium Chloride 0.13%
Lidocaine HCL 0.5%
Water-Jel Technologies
Carlstadt, NJ 07072
Drug Facts
Purpose
First Aid Antiseptic, External analgesic
Uses
first aid to help prevent infection and for temporary
relief of pain an itching associated with minor cuts,
scrapes, burns
Warnings
For external use only
Do not use
in the eyes
in large quantities over raw or blistered areas or on
deep puncture wounds, animal bites, or serious burns
Keep out of reach of children
if ingested contact Poison Control Center right away
Directions
clean affected area apply a small amount not more
than 3 times daily may be covered with a sterile bandage
Other Information
Store at room temperature