BACITRACIN- bacitracin ointment
Dynarex Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredient
Bacitracin (500 units in each gram)
Purpose
First Aid Antibiotic
Use
First aid to help prevent infection in minor cuts, scrapes, and burns
Warnings
For External Use Only
Do not use
- In the eyes or apply over large areas of the body
- If you are allergic to any of the ingredients
- Longer than 1 week unless directed by a doctor
Ask a doctor before use if you have
deep or puncture wounds, animal bites, or serious burns
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Clean the affected area. Apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily. May be covered with a sterile bandage.
Other information
Store at room temperature
Inactive ingredients
Hard Paraffin, Liquid Paraffin, White Soft Paraffin
Label
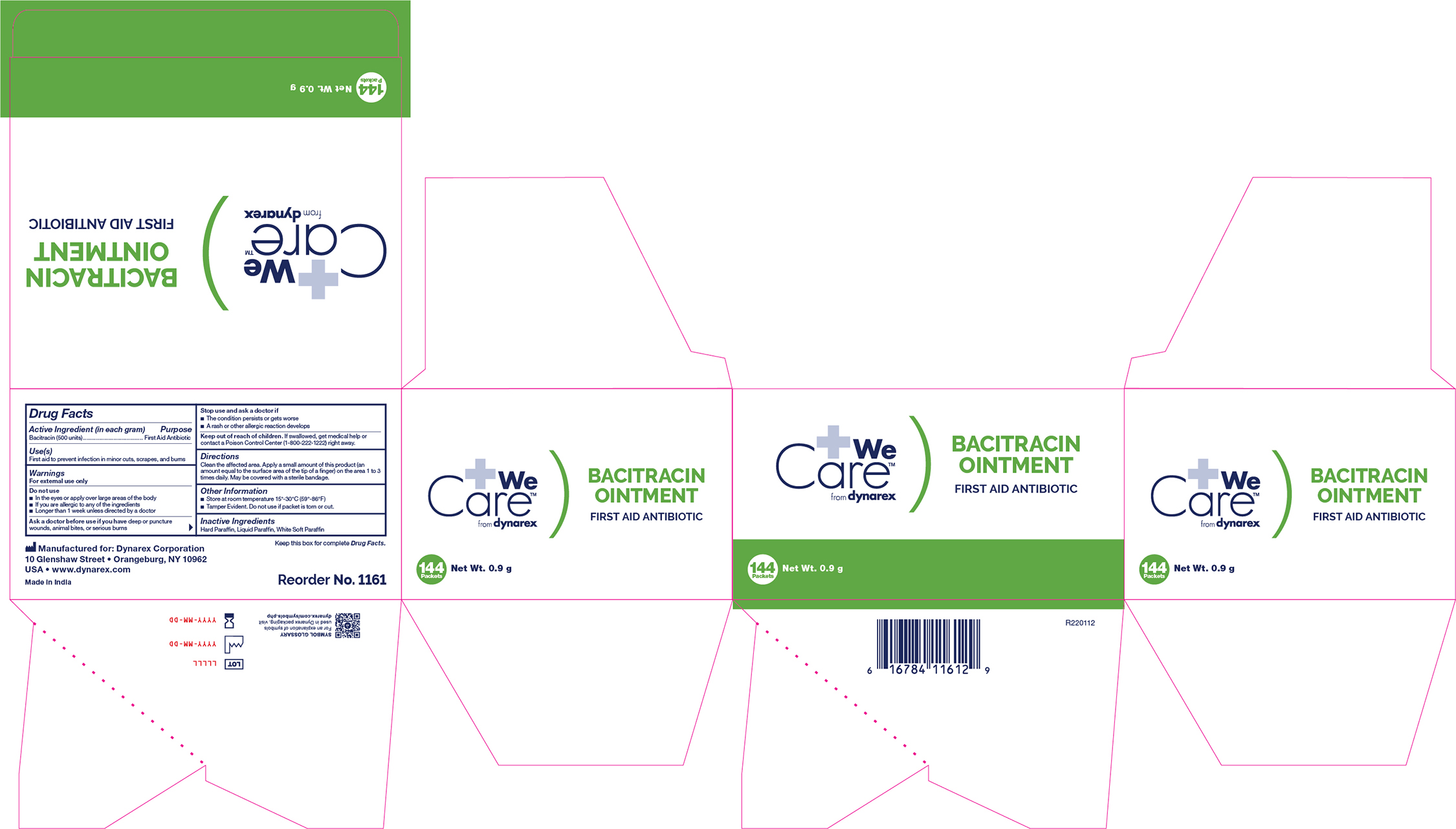 1161 Bacitracin Ointment
1161 Bacitracin Ointment
Label
 1162 Bacitracin Ointment
1162 Bacitracin Ointment
Label
 1163 Bacitracin Ointment
1163 Bacitracin Ointment