PVP Wipes
Warnings
For external use only.
PVP Wipes
Directions
- clean the affected area
- apply1 to 3 times daily
- may be covered with a sterile bandage
- if bandaged, let dry first
- discard wipe after single use
PVP Wipes
Other information
- do not use on individuals who are allergic or sensitive to iodine
- store at controlled temperature 59-86ºF (15-30ºC)
- do not use if pouch is open or torn
Burn Cream
Warnings
For external use only
Do not use
- in or near the eyes
- if you are allergic to any of the ingredients
- in large areas of the body particularly over raw surfaces or blistered areas
- for more than 10 days
Burn Cream
Directions
- adults and children 2 years of age and older:
- clean the affected area
- apply a small amount of this product (equal to the area of the tip of finger) onto the affected area 1 to 3 times daily
- may be covered with a sterile bandage
- children under 2 years of age: ask a doctor
Burn Cream
Inactive ingredients
aloe barbadensis juice, cetyl alcohol, diazolidinyl urea, edetate disodium, glycerin, glyceryl stearate SE, methylparaben, mineral oil, PEG-100, propylene glycol, propylparaben, stearic acid, trolamine, water
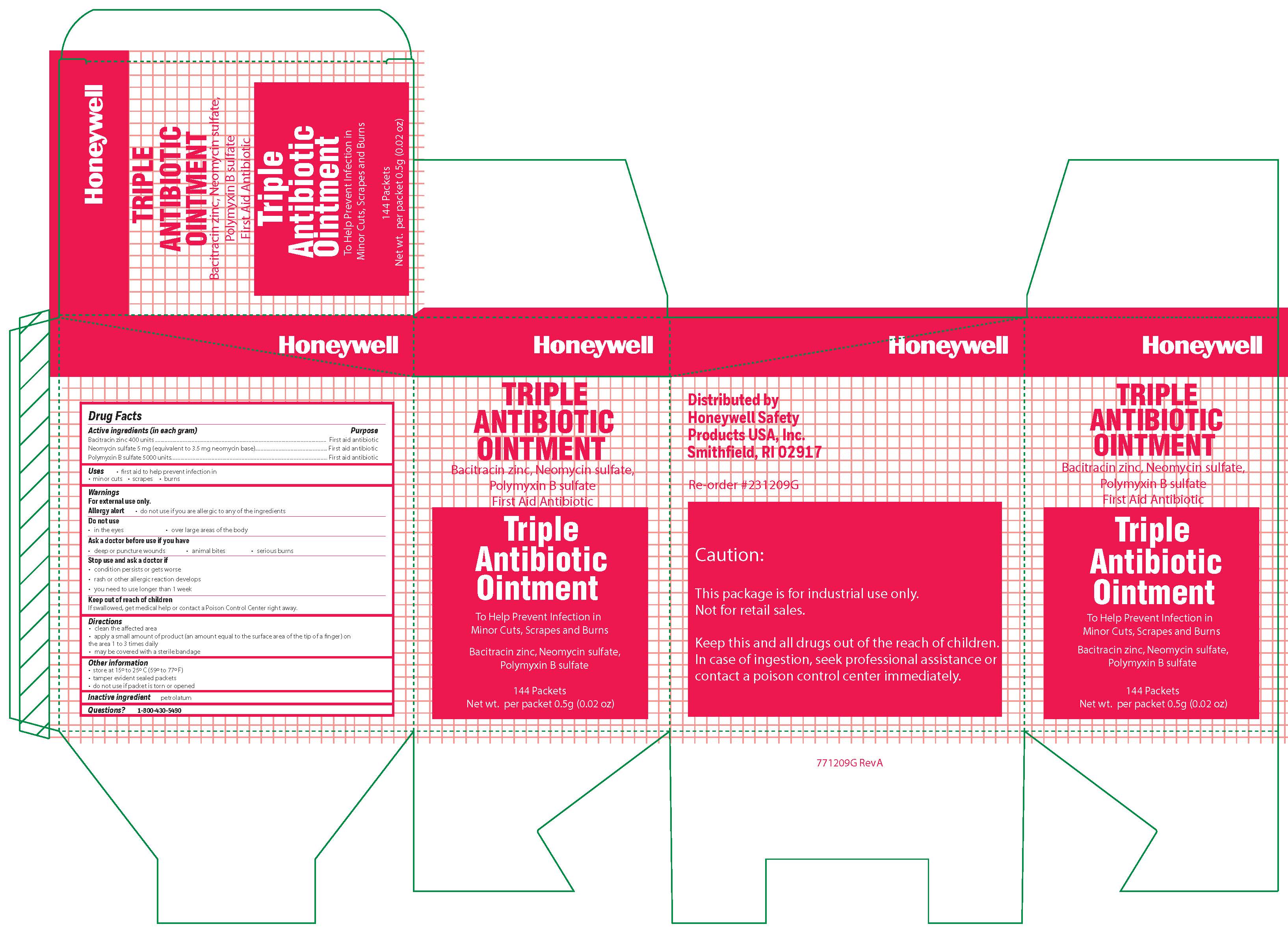
Triple
Active ingredients
Bacitracin zinc 400 units
Neonycin sulfate (5 mg equivalent to 3.5 mg Neomycin base)
Polymyxin B sulfate 5000 units
Triple
Warnings
For external use only
Allergy alert: do not use if you are allergic to any of the ingredients
Triple
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Triple
Other information
- store at 15 o to 25 o C (59 o to 77 oF)
- tamper evident sealed packets
- do not use if packet is torn or opened
4064
Z019737-0024L Kit Contents
TRIPLE ANTIBIOTIC 10 PER
FIRST AID BURN CREAM 6 PER
TRIANGULAR BDG, NON-STERILE
WIRE SPLINT 1 PER
BANDAGE COMP, 2" OFFSET, 4 PER
BANDAGE COMP, 4" OFFSET, 1 PER
ADHESIVE BDG,PLSTIC,1"X3"16PER
PVP IODINE WIPES 10 PER
NITRILE GLOVES 2PR BBP
FIRST AID GUIDE ASHI
4065
019737-0024L Kit Contents
TRIPLE ANTIBIOTIC 10 PER
FIRST AID BURN CREAM 6 PER
TRIANGULAR BDG, NON-STERILE
WIRE SPLINT 1 PER
GAUZE PADS, 3" X 3", 4 PER
BANDAGE COMP, 2" OFFSET, 4 PER
BANDAGE COMP, 4" OFFSET, 1 PER
ADHESIVE BDG,PLSTIC,1"X3"16PER
PVP IODINE WIPES 10 PER
ADHESIVE TAPE W/P 1/2"X 5 YD
1 PR LRG NITRILE GLVES ZIP BAG
4066
Z019839 Kit Contents
TRIPLE ANTIBIOTIC 10 PER
FIRST AID BURN CREAM 6 PER
TRIANGULAR BDG, NON-STERILE
GAUZE PADS, 3" X 3", 4 PER
ADH TAPE, .5" X 2.5 YD, 2 PER
GAUZE COMP, 1 SQ YARD, 1 PER
INSTANT COLD PACK 4" X 6"
ADHESIVE BDG,PLSTIC,1"X3"16PER
PVP IODINE WIPES 10 PER
NITRILE GLOVES 2PR BBP
FIRST AID GUIDE ASHI
SCISSOR BDGE 4" RED PLS HDL
4067
019708-0005L Kit Contents
TRIPLE ANTIBIOTIC 10 PER
FIRST AID BURN CREAM 6 PER
TRIANGULAR BDG, NON-STERILE
GAUZE PADS, 3" X 3", 4 PER
ADH TAPE, .5" X 2.5 YD, 2 PER
GAUZE COMP, 1 SQ YARD, 1 PER
INSTANT COLD PACK 4" X 6"
ADHESIVE BDG,PLSTIC,1"X3"16PER
PVP IODINE WIPES 10 PER
NITRILE GLOVES 2PR BBP
FIRST AID GUIDE ASHI
SCISSOR BDGE 4" RED PLS HDL