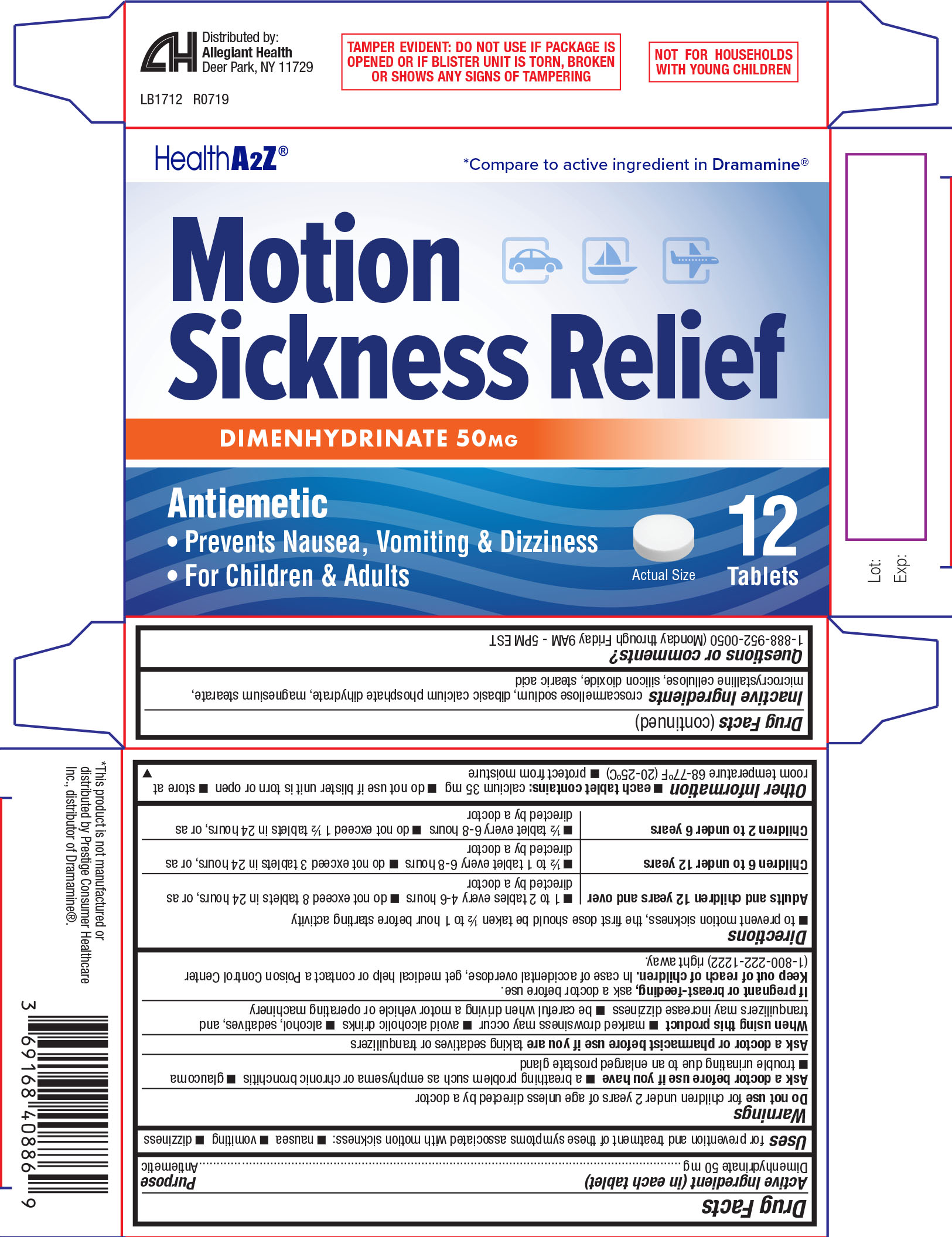
Active ingredient (in each tablet)
Dimenhydrinate 50 mg
Use
for prevention and treatment of these symptoms associated with motion sickness:
- nausea
- vomiting
- dizziness
Warnings
Do not use
for children under 2 years of age unless directed by a doctor
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are
taking sedatives or tranquilizers
When using this product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase dizziness
- be careful when driving a motor vehicle or operating machinery
If pregnant or breast-feeding,
ask a doctor before use.
Keep out of reach of children.
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
- to prevent motion sickness, the first dose should be taken ½ to 1 hour before starting activity
Adults and
children 12
years and over |
- 1 to 2 tables every 4-6 hours
- do not exceed 8 tablets in
24 hours, or as directed by a doctor
|
Children 6 to
under 12 years |
- ½ to 1 tablet every 6-8 hours
- do not exceed 3 tablets in
24 hours, or as directed by a doctor
|
Children 2 to
under 6 years |
- ½ tablet every 6-8 hours
- do not exceed 1 ½ tablets
in 24 hours, or as directed by a doctor
|
Other information
- each tablet contains: calcium 35mg
- do not use ifblister unit is torn or open
- store at room temperature 68-77ºF (20-25ºC)
- protect from moisture
Inactive ingredients
croscarmellose sodium, dibasic calcium phosphate dihydrate, magnesium stearate, microcrystalline cellulose, silicon dioxide, stearic acid
Questions or comments?
1-888-952-0050 (Monday through Friday 9AM - 5PM EST)