FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Pulmonary Arterial Hypertension
TADLIQ® is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise ability. Studies establishing effectiveness included predominately patients with NYHA Functional Class II – III symptoms and etiologies of idiopathic or heritable PAH (61%) or PAH associated with connective tissue diseases (23%).
2 DOSAGE AND ADMINISTRATION
2.1 Pulmonary Arterial Hypertension
The recommended dose of TADLIQ is 40 mg (10 mL) taken once daily with or without food.
2.2 Dose Adjustment in Renal Impairment
Mild (creatinine clearance 51 to 80 mL/min) or moderate (creatinine clearance 31 to 50 mL/min): Start dosing at 20 mg (5mL) once daily. Increase to 40 mg (10 mL) once daily based on individual tolerability.
Severe (creatinine clearance <30 mL/min and on hemodialysis): Avoid use of TADLIQ because of increased tadalafil exposure (AUC), limited clinical experience, and the lack of ability to influence clearance by dialysis [see Use in Specific Populations (8.6)].
2.3 Dose Adjustment in Hepatic Impairment
Mild or moderate (Child Pugh Class A or B): Because of limited clinical experience in patients with mild to moderate hepatic cirrhosis, consider a starting dose of 20 mg (5 mL) once per day.
Severe (Child Pugh Class C): Patients with severe hepatic cirrhosis have not been studied. Avoid use of TADLIQ [see Use in Specific Populations (8.7)].
2.4 Dose Adjustment for Use with Ritonavir
Co-administration of TADLIQ in Patients on Ritonavir
In patients receiving ritonavir for at least one week, start TADLIQ at 20 mg (5 mL) once daily. Increase to 40 mg (10 mL) once daily based upon individual tolerability [see Drug Interactions (7.5) and Clinical Pharmacology (12.3)].
Co-administration of Ritonavir in Patients on TADLIQ
Avoid use of TADLIQ during the initiation of ritonavir. Stop TADLIQ at least 24 hours prior to starting ritonavir. After at least one week following the initiation of ritonavir, resume TADLIQ at 20 mg (5 mL) once daily. Increase to 40 mg (10 mL) once daily based upon individual tolerability [see Drug Interactions (7.5) and Clinical Pharmacology (12.3)].
3 DOSAGE FORMS AND STRENGTHS
Oral Suspension: 20 mg/5 mL; white to off-white opaque suspension with a peppermint flavor.
4 CONTRAINDICATIONS
4.1 Concomitant Organic Nitrates
TADLIQ is contraindicated in patients who are using any form of organic nitrate, either regularly or intermittently. Do not use nitrates within 48 hours of the last dose of TADLIQ. TADLIQ potentiates the hypotensive effect of nitrates. This potentiation is thought to result from the combined effects of nitrates and TADLIQ on the nitric oxide/cGMP pathway [see Clinical Pharmacology (12.2)].
4.2 Concomitant Guanylate Cyclase (GC) Stimulators
Coadministration of GC stimulators such as riociguat with TADLIQ is contraindicated. TADLIQ may potentiate the hypotensive effects of GC stimulators.
4.3 Hypersensitivity Reactions
TADLIQ is contraindicated in patients with a known serious hypersensitivity to tadalafil (TADLIQ, ADCIRCA® or CIALIS®). Hypersensitivity reactions have been reported, including Stevens-Johnson syndrome and exfoliative dermatitis [see Adverse Reactions (6.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
TADLIQ has vasodilatory properties that may result in transient decreases in blood pressure. Prior to prescribing TADLIQ, carefully consider whether patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects. Patients with preexisting hypotension, with autonomic dysfunction, with left ventricular outflow obstruction, may be particularly sensitive to the actions of vasodilators.
5.2 Worsening Pulmonary Vascular Occlusive Disease
Pulmonary vasodilators may significantly worsen the cardiovascular status of patients with pulmonary veno-occlusive disease (PVOD). Since there are no clinical data on administration of TADLIQ to patients with veno-occlusive disease, administration of TADLIQ to such patients is not recommended. Should signs of pulmonary edema occur when TADLIQ is administered, the possibility of associated PVOD should be considered.
5.3 Visual Loss
When used to treat erectile dysfunction, non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported postmarketing in temporal association with the use of phosphodiesterase type 5 (PDE-5) inhibitors, including tadalafil. Most, but not all, of these patients had underlying anatomic or vascular risk factors for development of NAION, including but not necessarily limited to: low cup to disc ratio (“crowded disc”), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia, and smoking. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged ≥50 in the general population. An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, typical of erectile dysfunction treatment, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate doubling in the risk of NAION. Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies.
Patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, were not included in the clinical trials, and use in these patients is not recommended.
5.4 Hearing Impairment
Cases of sudden decrease or loss of hearing, which may be accompanied by tinnitus and dizziness, have been reported in patients taking tadalafil. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.2)].
5.5 Combination with Other PDE5 Inhibitors
Tadalafil is also marketed for erectile dysfunction. The safety and efficacy of taking TADLIQ together with another PDE5 inhibitor has not been studied. Inform patients taking TADLIQ not to take other PDE5 inhibitors.
5.6 Prolonged Erection
There have been reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds. Patients with conditions that might predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia), or in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis, or Peyronie's disease) are at an increased risk. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Patients who have an erection lasting greater than 4 hours, whether painful or not, should seek emergency medical attention.
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed elsewhere in the labeling:
- Hypotension [see Warnings and Precautions (5.1)]
- Visual Loss [see Warnings and Precautions (5.3)]
- Hearing loss [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Tadalafil was administered to 398 patients with PAH during clinical trials worldwide. In trials of tadalafil, a total of 311 and 251 subjects have been treated for at least 182 days and 360 days, respectively. The overall rates of discontinuation because of an adverse event (AE) in the placebo-controlled trial were 9% for tadalafil 40 mg and 15% for placebo. The rates of discontinuation because of AEs, other than those related to worsening of PAH, in patients treated with tadalafil 40 mg was 4% compared to 5% in placebo-treated patients.
In the placebo-controlled study, the most common AEs were generally transient and mild to moderate in intensity. Table 1 presents treatment-emergent adverse events reported by ≥9% of patients in the tadalafil 40 mg group and occurring more frequently than with placebo.
| EVENT | Placebo (%)
(N=82) | Tadalafil 20 mg (%)
(N=82) | Tadalafil 40 mg (%)
(N=79) |
| Headache | 15 | 32 | 42 |
| Myalgia | 4 | 9 | 14 |
| Nasopharyngitis | 7 | 2 | 13 |
| Flushing | 2 | 6 | 13 |
| Respiratory Tract Infection (Upper and Lower) | 6 | 7 | 13 |
| Pain in Extremity | 2 | 5 | 11 |
| Nausea | 6 | 10 | 11 |
| Back Pain | 6 | 12 | 10 |
| Dyspepsia | 2 | 13 | 10 |
| Nasal Congestion (Including sinus congestion) | 1 | 0 | 9 |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tadalafil. These events have been chosen for inclusion either because of their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure. The list does not include adverse events that are reported from clinical trials and that are listed elsewhere in this section.
Cardiovascular and cerebrovascular — Serious cardiovascular events, including myocardial infarction, sudden cardiac death, stroke, chest pain, palpitations, and tachycardia, have been reported postmarketing in temporal association with the use of tadalafil [see Contraindications (4.1)]. Most, but not all, of these patients had preexisting cardiovascular risk factors. Many of these events were reported to occur during or shortly after sexual activity, and a few were reported to occur shortly after the use of tadalafil without sexual activity. Others were reported to have occurred hours to days after the use of tadalafil and sexual activity. It is not possible to determine whether these events are related directly to tadalafil, to sexual activity, to the patient's underlying cardiovascular disease, to a combination of these factors, or to other factors.
Body as a whole — Hypersensitivity reactions including urticaria, Stevens–Johnson syndrome, and exfoliative dermatitis
Nervous — Migraine, seizure and seizure recurrence, and transient global amnesia
Ophthalmologic — Visual field defect, retinal vein occlusion, retinal artery occlusion, and NAION [see Warnings and Precautions (5.3)].
Otologic — Cases of sudden decrease or loss of hearing have been reported postmarketing in temporal association with the use of PDE5 inhibitors, including tadalafil. In some of the cases, medical conditions and other factors were reported that may have also played a role in the otologic adverse events. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of tadalafil, to the patient's underlying risk factors for hearing loss, a combination of these factors, or to other factors [see Warnings and Precautions (5.4)].
Urogenital — Priapism [see Warnings and Precautions (5.6)].
7 DRUG INTERACTIONS
7.1 Nitrates
Administration of nitrates within 48 hours after the last dose of TADLIQ is contraindicated [see Contraindications (4.1)].
7.2 Alpha-Blockers
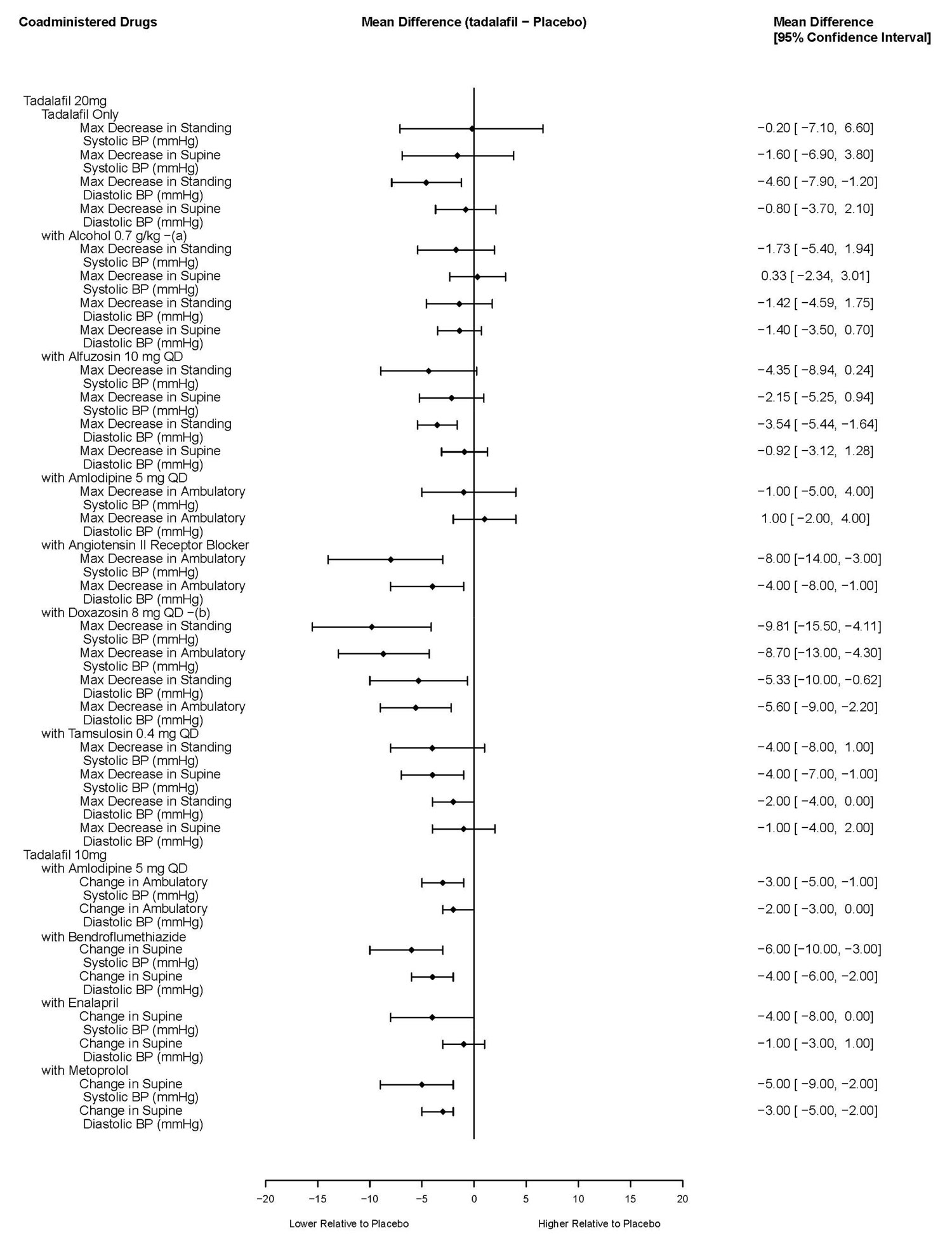
PDE5 inhibitors, including TADLIQ, and alpha–adrenergic blocking agents are both vasodilators with blood-pressure-lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may be anticipated. Clinical pharmacology studies have been conducted with coadministration of tadalafil with doxazosin, alfuzosin or tamsulosin [see Clinical Pharmacology (12.2)].
7.3 Antihypertensives
PDE5 inhibitors, including TADLIQ, are mild systemic vasodilators. Clinical pharmacology studies were conducted to assess the effect of tadalafil on the potentiation of the blood–pressure–lowering effects of selected antihypertensive medications (amlodipine, angiotensin II receptor blockers, bendroflumethiazide, enalapril, and metoprolol). Small reductions in blood pressure occurred following coadministration of tadalafil with these agents compared with placebo [see Clinical Pharmacology (12.2)].
7.4 Alcohol
Both alcohol and tadalafil, a PDE5 inhibitor, act as mild vasodilators. When mild vasodilators are taken in combination, blood pressure–lowering effects of each individual compound may be increased. Substantial consumption of alcohol (e.g., 5 units or greater) in combination with TADLIQ can increase the potential for orthostatic signs and symptoms, including increase in heart rate, decrease in standing blood pressure, dizziness, and headache. Tadalafil (10 mg or 20 mg) did not affect alcohol plasma concentrations and alcohol did not affect tadalafil plasma concentrations. [See Clinical Pharmacology (12.2)].
7.5 CYP3A Inhibitors/Inducers
Ritonavir
Ritonavir initially inhibits and later induces CYP3A, the enzyme involved in the metabolism of tadalafil. At steady state of ritonavir (about 1 week), the exposure to tadalafil is similar as in the absence of ritonavir [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
Potent Inhibitors of CYP3A
Tadalafil is metabolized predominantly by CYP3A in the liver. In patients taking potent inhibitors of CYP3A such as ketoconazole, and itraconazole, avoid use of TADLIQ [see Clinical Pharmacology (12.3)].
Potent Inducers of CYP3A
For patients chronically taking potent inducers of CYP3A, such as rifampin, avoid use of TADLIQ [see Clinical Pharmacology (12.3)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Pregnant women with untreated pulmonary arterial hypertension are at risk for adverse maternal and fetal outcomes (see Clinical Considerations). Available data from a randomized controlled trial, observational studies, and case series with tadalafil use in pregnant women have not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental effects were observed with oral administration of tadalafil to pregnant rats or mice during organogenesis at exposures 7 times the exposure at the maximum recommended human dose (MRHD) of 40 mg/day based on AUC (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15- 20%, respectively.
Data
Animal Data
Tadalafil and/or its metabolites cross the placenta, resulting in fetal exposure in rats.
Animal reproduction studies showed no evidence of teratogenicity, embryotoxicity, or fetotoxicity when tadalafil was given to pregnant rats or mice at unbound tadalafil exposures up to 7 times the exposure at the maximum recommended human dose (MRHD) of 40 mg/day during organogenesis based on AUC. In one of two perinatal/postnatal developmental studies in rats, a reduction of postnatal pup survival was observed at dose levels of 60, 200 and 1000 mg/kg. The no-observed-effect-level (NOEL) for developmental toxicity was 30 mg/kg, which provided maternal exposure to unbound tadalafil concentrations approximately 5 times the exposure at the MRHD based on AUC. Signs of maternal toxicity occurred at doses greater than 200 mg/kg/day, which produced AUCs greater than 8 times the exposure at the MRHD. Surviving offspring had normal development and reproductive performance.
8.2 Lactation
Risk Summary
There are no data on the presence of tadalafil and/or its metabolites in human milk, the effects on the breastfed child, or the effects on milk production. Tadalafil and/or its metabolites are present in the milk of lactating rats at concentrations approximately 2.4-times that found in the plasma. When a drug is present in animal milk, it is likely that the drug will be present in human milk.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for TADLIQ and any potential adverse effects on the breastfed child from TADLIQ or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Infertility
Males
Based on the data from 3 studies in adult males, tadalafil decreased sperm concentrations in the study of 10 mg tadalafil for 6 months and the study of 20 mg tadalafil for 9 months. This effect was not seen in the study of 20 mg tadalafil taken for 6 months. There was no adverse effect of tadalafil 10 mg or 20 mg on mean concentrations of testosterone, luteinizing hormone or follicle stimulating hormone. The clinical significance of the decreased sperm concentrations in the two studies is unknown. There have been no studies evaluating the effect of tadalafil on fertility in men or women [see Clinical Pharmacology (12.2)].
8.4 Pediatric Use
Safety and effectiveness of TADLIQ in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of subjects in the clinical study of tadalafil for pulmonary arterial hypertension, 28 percent were 65 and over, while 8 percent were 75 and over. No overall differences in safety were observed between subjects over 65 years of age compared to younger subjects or those over 75 years of age. No dose adjustment is warranted based on age alone; however, a greater sensitivity to medications in some older individuals should be considered. [See Clinical Pharmacology (12.3)].
8.6 Renal Impairment
For patients with mild or moderate renal impairment, start TADLIQ at 20 mg (5 mL) once daily. Increase the dose to 40 mg (10 mL) once daily based upon individual tolerability [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
In patients with severe renal impairment, avoid use of TADLIQ because of increased tadalafil exposure (AUC), limited clinical experience, and the lack of ability to influence clearance by dialysis [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Because of limited clinical experience in patients with mild to moderate hepatic cirrhosis (Child-Pugh Class A or B), consider a starting dose of TADLIQ 20 mg (5 mL) once daily. Patients with severe hepatic cirrhosis (Child-Pugh Class C) have not been studied, thus avoid use of TADLIQ in such patients [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
10 OVERDOSAGE
Single doses up to 500 mg have been given to healthy male subjects, and multiple daily doses up to 100 mg have been given to male patients with erectile dysfunction. Adverse reactions were similar to those seen at lower doses. Doses greater than 40 mg have not been studied in patients with pulmonary arterial hypertension. In cases of overdose, standard supportive measures should be adopted as needed. Hemodialysis contributes negligibly to tadalafil elimination.
11 DESCRIPTION
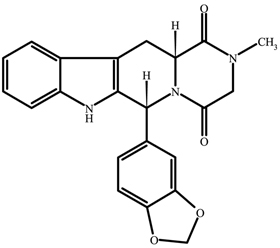
TADLIQ contains tadalafil, a selective inhibitor of cyclic guanosine monophosphate (cGMP)–specific phosphodiesterase type 5 (PDE5). Tadalafil has the empirical formula C22H19N3O4 representing a molecular weight of 389.41. The structural formula is:
The chemical designation is pyrazino[1´,2´:1,6]pyrido[3,4–b]indole-1,4-dione, 6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-, (6R,12aR)-. It is a crystalline solid that is practically insoluble in water and very slightly soluble in ethanol.
TADLIQ oral suspension contains 20 mg/5 mL of tadalafil and the following inactive ingredients: 30% simethicone emulsion, citric acid monohydrate, glycerin, peppermint flavor, polysorbate 80, purified water, sodium benzoate, sucralose, trisodium citrate dihydrate, xanthan gum.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Tadalafil is an inhibitor of phosphodiesterase type 5 (PDE5), the enzyme responsible for the degradation of cyclic guanosine monophosphate (cGMP). Pulmonary arterial hypertension is associated with impaired release of nitric oxide by the vascular endothelium and consequent reduction of cGMP concentrations in the pulmonary vascular smooth muscle. PDE5 is the predominant phosphodiesterase in the pulmonary vasculature. Inhibition of PDE5 by tadalafil increases the concentrations of cGMP resulting in relaxation of pulmonary vascular smooth muscle cells and vasodilation of the pulmonary vascular bed.
Studies in vitro have demonstrated that tadalafil is a selective inhibitor of PDE5. PDE5 is found in pulmonary vascular smooth muscle, visceral smooth muscle, corpus cavernosum, skeletal muscle, platelets, kidney, lung, cerebellum, and pancreas.
In vitro studies have shown that the effect of tadalafil is more potent on PDE5 than on other phosphodiesterases. These studies have shown that tadalafil is >10,000 times as potent for PDE5 than for PDE1, PDE2, PDE4, and PDE7 enzymes, which are found in the heart, brain, blood vessels, liver, leukocytes, skeletal muscle, and other organs. Tadalafil is >10,000 times as potent for PDE5 than for PDE3, an enzyme found in the heart and blood vessels. Additionally, tadalafil is 700–fold as potent for PDE5 than for PDE6, which is found in the retina and is responsible for phototransduction. Tadalafil is >9,000-fold as potent for PDE5 than for PDE8, PDE9, and PDE10. Tadalafil is 14 times as potent for PDE5 than for PDE11A1 and 40 times as potent for PDE5 than for PDE11A4, two of the four known forms of PDE11. PDE11 is an enzyme found in human prostate, testes, skeletal muscle and in other tissues. In vitro, tadalafil inhibits human recombinant PDE11A1 and, to a lesser degree, PDE11A4 activities at concentrations within the therapeutic range. The physiological role and clinical consequence of PDE11 inhibition in humans have not been defined.
12.2 Pharmacodynamics
Effects on Blood Pressure When Administered with Nitrates
In clinical pharmacology studies, tadalafil (5 to 20 mg) was shown to potentiate the hypotensive effect of nitrates. Do not use TADLIQ in patients taking any form of nitrates [see Contraindications (4.1)].
A double–blind, placebo–controlled, crossover study in 150 male subjects at least 40 years of age (including subjects with diabetes mellitus and/or controlled hypertension) assessed the interaction between nitroglycerin and tadalafil. Subjects received daily doses of tadalafil 20 mg or matching placebo for 7 days and then were given a single dose of 0.4 mg sublingual nitroglycerin (NTG) at pre–specified timepoints following their last dose of tadalafil (2, 4, 8, 24, 48, 72, and 96 hours after tadalafil). A significant interaction between tadalafil and NTG was observed at each timepoint up to and including 24 hours. At 48 hours, by most hemodynamic measures, the interaction between tadalafil and NTG was not observed, although a few more tadalafil subjects compared to placebo experienced greater blood–pressure lowering effects at this timepoint. After 48 hours, the interaction was not detectable. [See Contraindications (4.1)].
Effects on Blood Pressure
The effects of tadalafil on blood pressure alone and administered with antihypertensives, alcohol, and alpha-blockers is shown in Figure 1.
Effects on Cardiac Electrophysiology
The effect of a single 100 mg dose of tadalafil (2.5 times the recommended dose) on the QT interval was evaluated at the time of peak tadalafil concentration in a randomized, double–blinded, placebo, and active–controlled (intravenous ibutilide) crossover study in 90 healthy males aged 18 to 53 years. The mean change in QTc (Fridericia QT correction) for tadalafil, relative to placebo, was 3.5 milliseconds (two–sided 90% CI=1.9, 5.1). The mean change in QTc (Individual QT correction) for tadalafil, relative to placebo, was 2.8 milliseconds (two–sided 90% CI=1.2, 4.4). In this study, the mean increase in heart rate associated with a 100 mg dose of tadalafil compared to placebo was 3.1 beats per minute.
Effects on Exercise Stress Testing
The effects of tadalafil on cardiac function, hemodynamics, and exercise tolerance were investigated in a single clinical pharmacology study. In this blinded crossover trial, 23 subjects with stable coronary artery disease and evidence of exercise–induced cardiac ischemia were enrolled. The primary endpoint was time to cardiac ischemia. The mean difference in total exercise time was 3 seconds (tadalafil 10 mg minus placebo), which represented no clinically meaningful difference. Further statistical analysis demonstrated that tadalafil was similar to placebo with respect to time to ischemia. Of note, in this study, in some subjects who received tadalafil followed by sublingual nitroglycerin in the post–exercise period, clinically significant reductions in blood pressure were observed, consistent with the augmentation by tadalafil of the blood–pressure–lowering effects of nitrates.
Effects on Vision
Single oral doses of PDE inhibitors have demonstrated transient dose-related impairment of color discrimination (blue/green), using the Farnsworth–Munsell 100–hue test, with peak effects near the time of peak plasma levels. This finding is consistent with the inhibition of PDE6, which is involved in phototransduction in the retina. In a study to assess the effects of a single dose of tadalafil 40 mg on vision (N=59), no effects were observed on visual acuity, intraocular pressure, or pupillometry. Across all clinical studies with tadalafil, reports of changes in color vision were rare (<0.1% of patients).
Effects on Sperm Characteristics
Three studies were conducted in men to assess the potential effect on sperm characteristics of tadalafil 10 mg (one 6-month study) and 20 mg (one 6-month and one 9-month study) administered daily. There were no adverse effects on sperm morphology or sperm motility in any of the three studies. In the study of 10 mg tadalafil for 6 months and the study of 20 mg tadalafil for 9 months, results showed a decrease in mean sperm concentrations relative to placebo, although these differences were not clinically meaningful. This effect was not seen in the study of 20 mg tadalafil taken for 6 months. In addition there was no adverse effect on mean concentrations of reproductive hormones, testosterone, luteinizing hormone or follicle stimulating hormone with either 10 or 20 mg of tadalafil compared to placebo.
Dose-Response Relationship
Dose-response relationships, between 20 mg and 40 mg, were not observed for 6-minute walk distance or pulmonary vascular resistance (PVR) in subjects with PAH in the placebo-controlled study. Median change from baseline in 6-minute walk distance was 32 meters and 35 meters at 16 weeks in subjects receiving 20 mg and 40 mg daily, respectively. Mean change from baseline PVR was -254 dynes*sec*cm-5 and -209 dynes*sec*cm-5 at 16 weeks in patients receiving 20 mg and 40 mg daily, respectively.
12.3 Pharmacokinetics
Over a dose range of 2.5 to 20 mg, tadalafil exposure (AUC) increases proportionally with dose in healthy subjects. In PAH patients administered between 20 and 40 mg of tadalafil, an approximately 50% greater AUC was observed indicating a less than proportional increase in exposure over the entire dose range of 2.5 to 40 mg. During tadalafil 20 and 40 mg once daily dosing, steady-state plasma concentrations were attained within 5 days, and exposure was approximately 30% higher than after a single dose.
Absorption — After single oral-dose administration, the maximum observed plasma concentration (Cmax) of tadalafil is achieved between 2 and 8 hours (median time of 4 hours). Absolute bioavailability of tadalafil following oral dosing has not been determined.
The rate and extent of absorption of tadalafil are not influenced by food; thus, TADLIQ may be taken with or without food.
Distribution — The mean apparent volume of distribution following oral administration is approximately 77 L, indicating that tadalafil is distributed into tissues. At therapeutic concentrations, 94% of tadalafil in plasma is bound to proteins.
Metabolism — Tadalafil is predominantly metabolized by CYP3A to a catechol metabolite. The catechol metabolite undergoes extensive methylation and glucuronidation to form the methylcatechol and methylcatechol glucuronide conjugate, respectively. The major circulating metabolite is the methylcatechol glucuronide. Methylcatechol concentrations are less than 10% of glucuronide concentrations. In vitro data suggests that metabolites are not expected to be pharmacologically active at observed metabolite concentrations.
Elimination — Following 40 mg, the mean oral clearance for tadalafil is 3.4 L/hr and the mean terminal half-life is 15 hours in healthy subjects. In patients with pulmonary hypertension not receiving concomitant bosentan, the mean oral clearance for tadalafil is 1.6 L/hr, and the mean terminal half-life is 35 hours. Tadalafil is excreted predominantly as metabolites, mainly in the feces (approximately 61% of the dose) and to a lesser extent in the urine (approximately 36% of the dose).
Population pharmacokinetics — In patients with pulmonary hypertension not receiving concomitant bosentan, the average tadalafil exposure at steady-state following 40 mg was 26% higher when compared to those of healthy volunteers. The results suggest a lower clearance of tadalafil in patients with pulmonary hypertension compared to healthy volunteers.
Geriatric patients
In healthy male elderly subjects (65 years or over) after a 10 mg dose, a lower oral clearance of tadalafil, resulting in 25% higher exposure (AUC) with no effect on Cmax was observed relative to that in healthy subjects 19 to 45 years of age.
Renal impairment
In clinical pharmacology studies using single-dose tadalafil (5 to 10 mg), tadalafil exposure (AUC) doubled in subjects with mild (creatinine clearance 51 to 80 mL/min) or moderate (creatinine clearance 31 to 50 mL/min) renal impairment. In subjects with end-stage renal disease on hemodialysis, there was a doubling in Cmax and AUC was 2.7- to 4.1 times as high following single-dose administration of 10 or 20 mg tadalafil, respectively. Exposure to total methylcatechol (unconjugated plus glucuronide) was 2- to 4 times as high in subjects with renal impairment, compared to those with normal renal function. Hemodialysis (performed between 24 and 30 hours post-dose) contributed negligibly to tadalafil or metabolite elimination [see Dosage and Administration (2.2)].
Hepatic impairment
In clinical pharmacology studies, tadalafil exposure (AUC) in subjects with mild or moderate hepatic impairment (Child-Pugh Class A or B) was comparable to exposure in healthy subjects when a dose of 10 mg was administered. There are no available data for doses higher than 10 mg of tadalafil in patients with hepatic impairment. Insufficient data are available for subjects with severe hepatic impairment (Child-Pugh Class C) [see Dosage and Administration (2.3)].
Patients with diabetes mellitus
In male patients with diabetes mellitus after a 10 mg tadalafil dose, exposure (AUC) was reduced approximately 19% and Cmax was 5% lower than that observed in healthy subjects. No dose adjustment is warranted.
Race
Pharmacokinetic studies have included subjects from different ethnic groups, and no differences in the typical exposure to tadalafil have been identified. No dose adjustment is warranted.
Gender
In healthy female and male subjects following single and multiple-doses of tadalafil, no clinically relevant differences in exposure (AUC and Cmax) were observed. No dose adjustment is warranted.
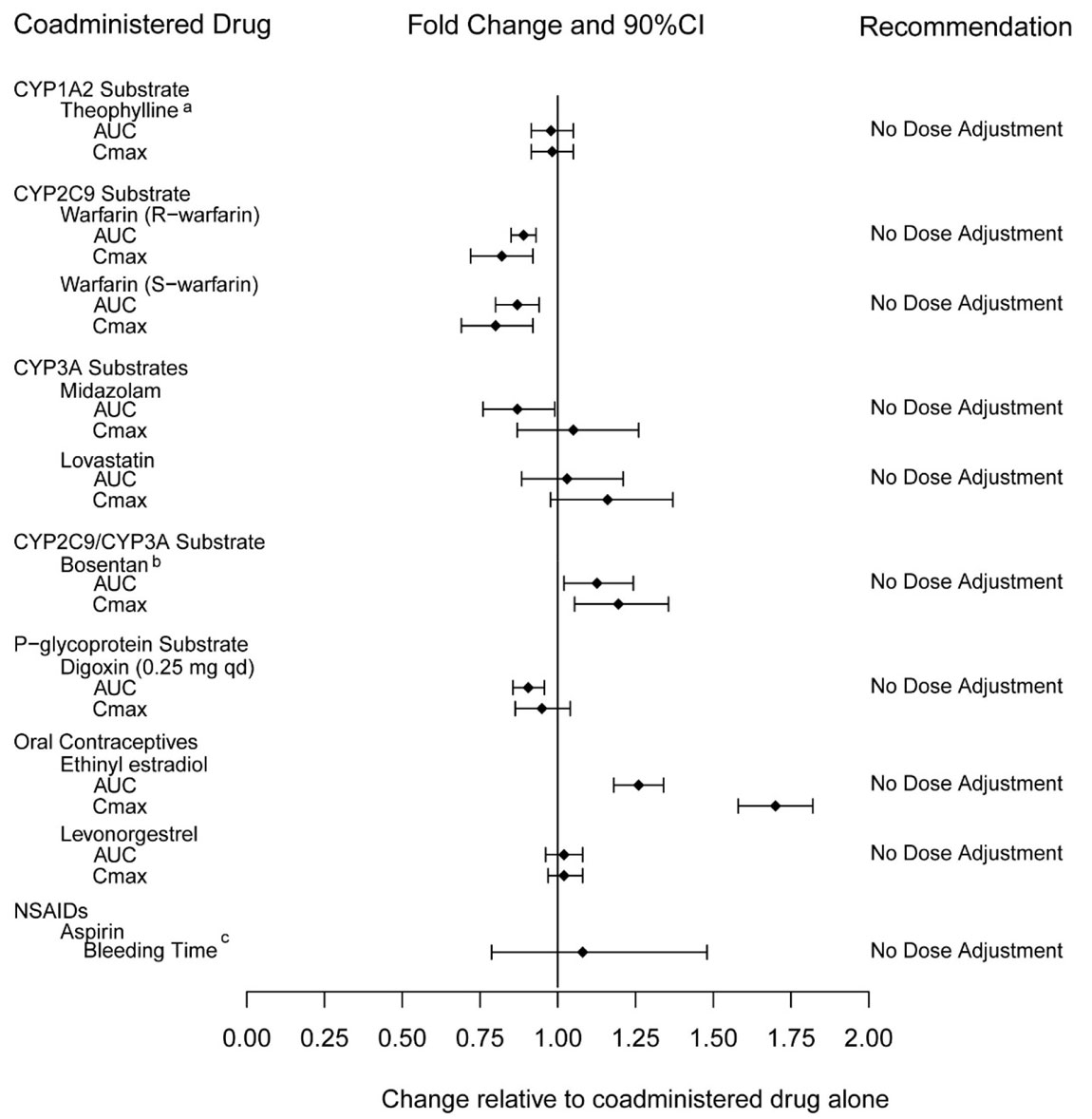
Cytochrome P450 3A4 inhibitors
Ketoconazole increased tadalafil exposure relative to the values for tadalafil alone (Figure 2). Although specific interactions have not been studied, other CYP3A inhibitors, such as erythromycin, itraconazole, and grapefruit juice, would likely increase tadalafil exposure.
Ritonavir
Ritonavir increased tadalafil 20–mg single-dose exposure relative to the values for tadalafil alone. Ritonavir inhibits and induces CYP3A, the enzyme involved in the metabolism of tadalafil, in a time-dependent manner. The initial inhibitory effect of ritonavir on CYP3A may be mitigated by a more slowly evolving induction effect so that after about 1 week of ritonavir twice daily, the exposure of tadalafil is similar in the presence of and absence of ritonavir [see Dosage and Administration (2.4) and Drug Interactions (7.5)]. Although specific interactions have not been studied, other HIV protease inhibitors would likely increase tadalafil exposure.
Cytochrome P450 3A4 inducers
Rifampin (600 mg daily), a CYP3A inducer, reduced tadalafil 10 mg single–dose exposure (AUC) by 88% and Cmax by 46%, relative to the values for tadalafil 10 mg alone [see Drug Interactions (7.5)].
Bosentan, a substrate of CYP2C9 and CYP3A and a moderate inducer of CYP3A, CYP2C9 and possibly CYP2C19, reduced tadalafil systemic exposure following multiple-dose co-administration (Figure 2). Although specific interactions have not been studied, other CYP3A inducers, such as carbamazepine, phenytoin, and phenobarbital, would likely decrease tadalafil exposure.
Exposure changes of tadalafil following co-administration with other drugs are shown in Figure 2.
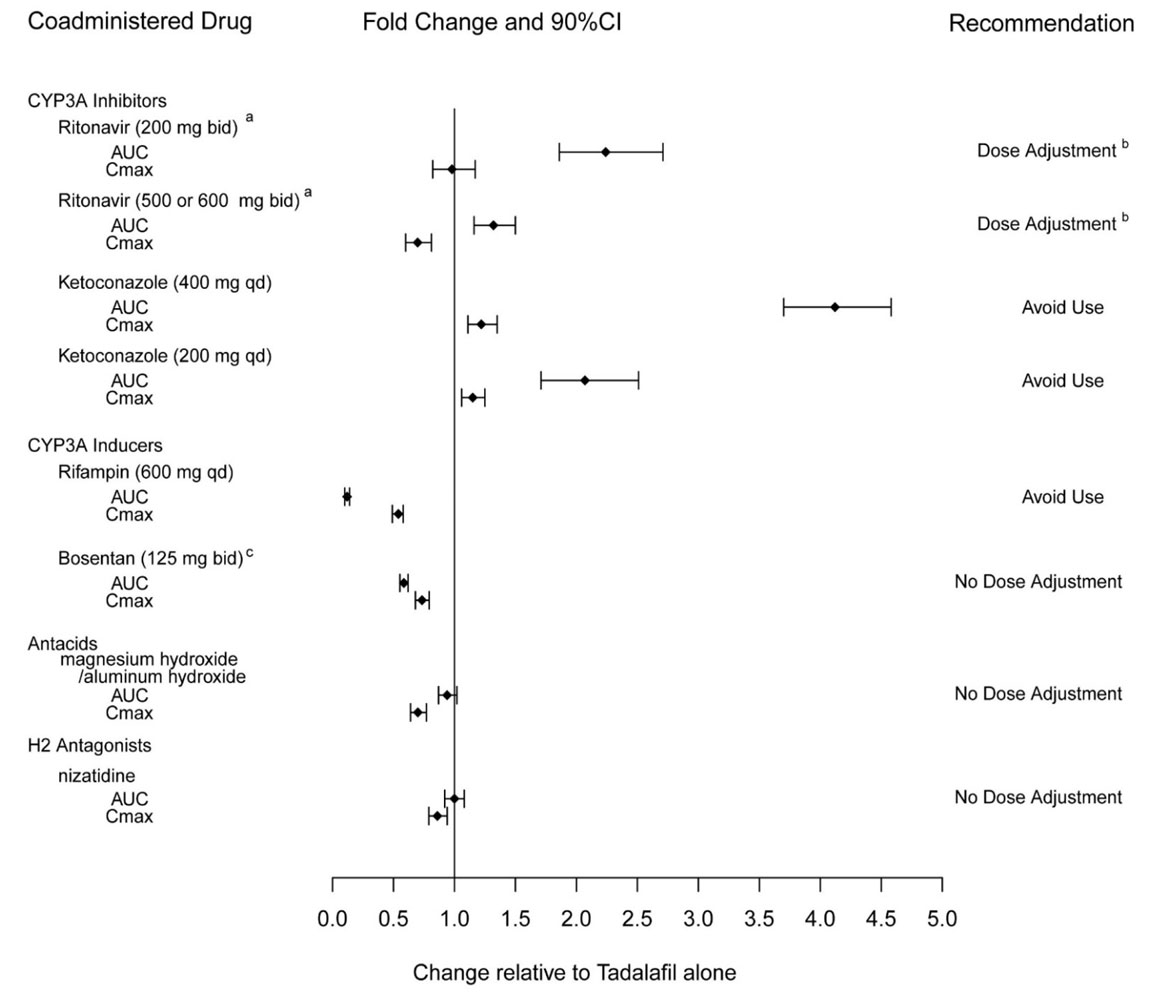 |
| a Ritonavir is also a CYP2C9/CYP2C19/CYP2D6 Inhibitor and CYP3A inducer. |
| b [see Dosage and Administration (2.4)]. |
| c Bosentan is also a CYP2C9/CYP2C19 inducer. |
| Figure 2: Impact of Other Drugs on the Pharmacokinetics of Tadalafil |
Cytochrome P450 substrates
Tadalafil is not expected to cause clinically significant inhibition or induction of the clearance of drugs metabolized by cytochrome P450 (CYP) isoforms.
Exposure changes of drugs following co-administration with tadalafil are shown in Figure 3.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis — Tadalafil was not carcinogenic to rats or mice when administered daily for 2 years at doses up to 400 mg/kg/day. Systemic drug exposures, as measured by AUC of unbound tadalafil, were approximately 5–fold for mice, and 7– and 14–fold for male and female rats, respectively, the exposures at the maximum recommended human dose (MRHD) of 40 mg.
Mutagenesis — Tadalafil was not mutagenic in the in vitro bacterial Ames assays or the forward mutation test in mouse lymphoma cells. Tadalafil was not clastogenic in the in vitro chromosomal aberration test in human lymphocytes or the in vivo rat micronucleus assays.
Impairment of Fertility — There were no effects on fertility, reproductive performance or reproductive organ morphology in male or female rats given oral doses of tadalafil up to 400 mg/kg/day, a dose producing AUCs for unbound tadalafil of 6–fold for males or 17–fold for females the exposures at the MRHD of 40 mg. In beagle dogs given tadalafil daily for 3 to 12 months, there was treatment–related non–reversible degeneration and atrophy of the seminiferous tubular epithelium in the testes in 20–100% of the dogs that resulted in a decrease in spermatogenesis in 40–75% of the dogs at doses of ≥10 mg/kg/day. Systemic exposure (based on AUC) at no–observed–adverse-effect–level (NOAEL) (10 mg/kg/day) for unbound tadalafil was similar to that expected in humans at the MRHD of 40 mg.
There were no treatment–related testicular findings in rats or mice treated with doses up to 400 mg/kg/day for 2 years.
13.2 Animal Toxicology and/or Pharmacology
Animal studies showed vascular inflammation in tadalafil–treated mice, rats, and dogs. In mice and rats, lymphoid necrosis and hemorrhage were seen in the spleen, thymus, and mesenteric lymph nodes at unbound tadalafil exposure of 1– to 17–fold the human exposure (AUCs) at the MRHD of 40 mg. In dogs, an increased incidence of disseminated arteritis was observed in 1– and 6-month studies at unbound tadalafil exposure of 0.5– to 38–fold the human exposure (AUC) at the MRHD of 40 mg. In a 12–month dog study, no disseminated arteritis was observed, but 2 dogs exhibited marked decreases in white blood cells (neutrophils) and moderate decreases in platelets with inflammatory signs at unbound tadalafil exposures of approximately 4– to 10–fold the human exposure at the MRHD of 40 mg. The abnormal blood–cell findings were reversible within 2 weeks upon removal of the drug.
14 CLINICAL STUDIES
14.1 Tadalafil for Pulmonary Arterial Hypertension
A randomized, double-blind, 16 week placebo-controlled study was conducted in 405 patients with pulmonary arterial hypertension, defined as a resting mean pulmonary artery pressure (mPAP) ≥25 mm Hg, pulmonary capillary wedge pressure (PCWP) ≤15 mm Hg, and pulmonary vascular resistance (PVR) ≥3 Wood units via right heart catheterization. Allowed background therapy included bosentan (maintenance dosing up to 125 mg twice daily) and chronic anticoagulation. The use of prostacyclin or analogue, L–arginine, phosphodiesterase inhibitor, or other chronic PAH medications were not permitted.
Subjects were randomly assigned to 1 of 5 treatment groups (tadalafil 2.5, 10, 20, 40 mg, or placebo) in a 1:1:1:1:1 ratio. Subjects had to be at least 12 years of age and had a diagnosis of PAH that was idiopathic, heritable, related to connective tissue disease, anorexigen use, human immunodeficiency virus (HIV) infection, associated with an atrial-septal defect, or associated with surgical repair of a congenital systemic-to-pulmonary shunt of least 1 year in duration (for example, ventricular septal defect, patent ductus arteriosus). Patients with a history of left-sided heart disease, severe renal insufficiency, or pulmonary hypertension related to conditions other than specified in the inclusion criteria were not eligible for enrollment.
The mean age of all subjects was 54 years (range 14 - 90 years) with the majority of subjects being Caucasian (81%) and female (78%). PAH etiologies were predominantly idiopathic or heritable PAH (61%) and related to connective tissue disease (23%). More than half (53%) of the subjects in the study were receiving concomitant bosentan therapy. The majority of subjects had a World Health Organization (WHO) Functional Class III (65%) or II (32%). The mean baseline 6-minute walk distance (6-MWD) was 343 meters. Of the 405 subjects, 341 completed the study.
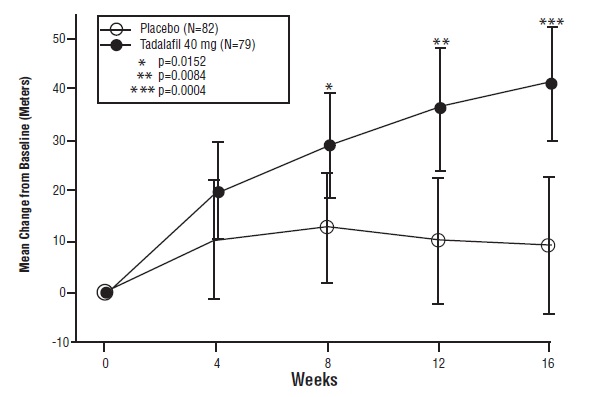
The primary efficacy endpoint was the change from baseline at week 16 in 6-MWD (see Figure 4). In the tadalafil 40 mg treatment group, the placebo-adjusted mean change increase in 6-MWD was 33 meters (95% C.I. 15-50 meters; p=0.0004). The improvement in 6-MWD was apparent at 8 weeks of treatment and then maintained at week 12 and week 16.
Placebo-adjusted changes in 6-MWD at 16 weeks were evaluated in subgroups (see Figure 5). In patients taking only 40 mg tadalafil (i.e., without concomitant bosentan), the placebo-adjusted mean change in 6-MWD was 44 meters. In patients taking 40 mg of tadalafil and concomitant bosentan therapy, the placebo adjusted mean change in 6-MWD was 23 meters.
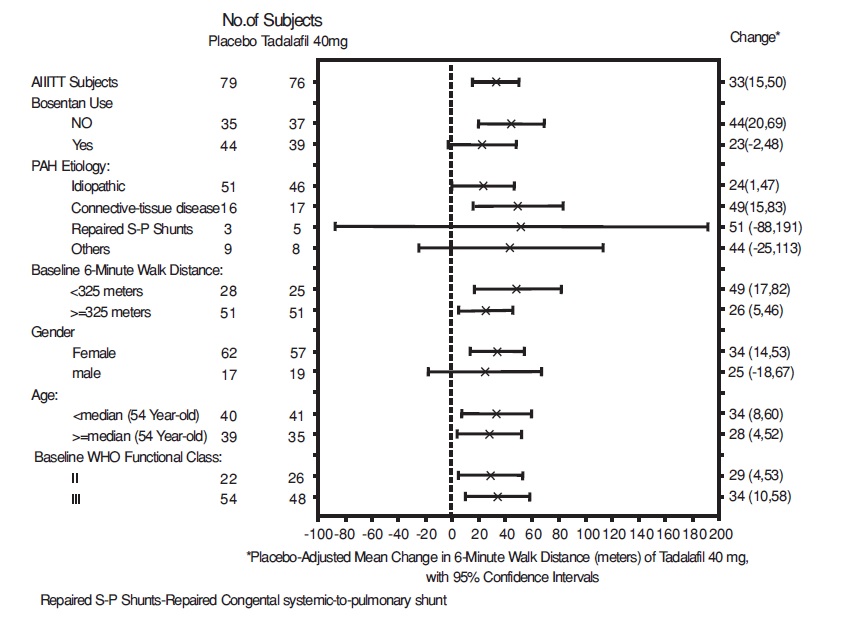
Figure 5: Placebo-adjusted Mean Change in 6-Minute Walk Distance (meters) of Tadalafil 40 mg, with 95% Confidence Intervals
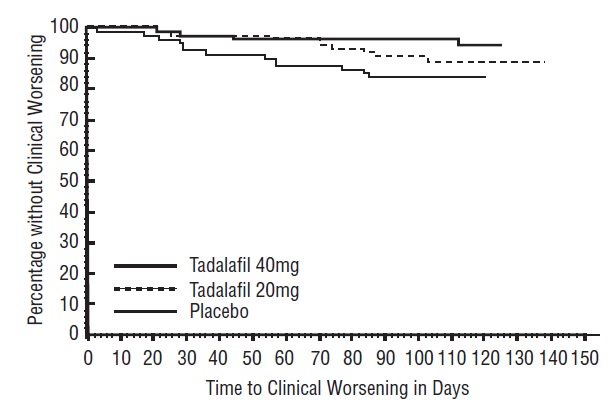
There was less clinical worsening (defined as death, lung transplantation, atrial septostomy, hospitalization because of worsening PAH, initiation of new PAH therapy [prostacyclin or analog, endothelin receptor antagonist, PDE5 inhibitor], or worsening WHO functional class) in the Tadalafil 40 mg group compared to the placebo group and the groups that used lower doses of tadalafil.
|
a Subjects may be counted in more than one category |
|||||
| Tadalafil | |||||
| Placebo N=82 | 2.5 mg N=82 | 10 mg N=80 | 20 mg N=82 | 40 mg N=79 |
|
| Total with clinical worsening | 13 (16) | 10 (12) | 7 (9) | 8 (10) | 4 (5) |
| Death | 1 | 0 | 1 | 0 | 0 |
| Hospitalization for worsening PAH | 2 | 2 | 3 | 0 | 1 |
| New PAH therapy | 0 | 1 | 0 | 2 | 1 |
| Worsening WHO class | 11 | 10 | 6 | 6 | 3 |
The Kaplan-Meier plot of times to clinical worsening is shown below in Figure 6.
14.2 Long-Term Treatment of Pulmonary Arterial Hypertension
Patients (N=357) from the placebo-controlled study entered a long-term extension study. Of these, 311 patients have been treated with tadalafil for at least 6 months and 182 for 1 year (median exposure 356 days; range 2 days to 415 days). The survival rate in the extension study was 96.5 per 100 patient years. Without a control group, these data must be interpreted cautiously.
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
Advise patient to read the FDA-Approved patient labeling (Patient Information)
- Inform patients that tadalafil is also marketed as CIALIS for erectile dysfunction (ED) and for the signs and symptoms of benign prostatic hyperplasia (BPH). Advise patients taking TADLIQ not to take CIALIS or other PDE5 inhibitors.
- Advise patients to seek immediate medical attention in the event of a sudden loss of vision in one or both eyes while taking TADLIQ. Such an event may be a sign of NAION. Also discuss with patients that there is an increased risk of NAION in individuals who have already experienced NAION in one eye, including whether such individuals could be adversely affected by use of vasodilators such as PDE5 inhibitors.
- Advise patients to seek prompt medical attention in the event of sudden decrease or loss of hearing while taking TADLIQ. These events may be accompanied by tinnitus and dizziness.
PATIENT INFORMATION
TADLIQ® (Tad-leek)
(tadalafil) oral suspension
What is the most important information I should know about TADLIQ?
Never take TADLIQ with any nitrate or guanylate cyclase stimulator medicines:
- Your blood pressure could drop quickly to an unsafe level.
- You could get dizzy, faint and even have a heart attack or stroke.
Nitrates include:
- Medicines that treat chest pain (angina)
- Nitroglycerin in any form including tablets, patches, sprays, and ointments
- Other nitrate medicines (isosorbide mononitrate or dinitrate)
- Street drugs that are inhaled, called “poppers” (amyl nitrate, butyl nitrate or nitrite)
Guanylate cyclase stimulators include:
- Riociguat, a medicine that treats pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension
Ask your healthcare provider or pharmacist if you are not sure if you take a nitrate or guanylate cyclase stimulator medicine.
See “What are the possible side effects of TADLIQ?” for more information about side effects.
What is TADLIQ?
TADLIQ is a prescription medicine used to treat pulmonary arterial hypertension (PAH) to improve your ability to exercise. PAH is a type of high blood pressure in the arteries of your lungs.
It is not known if TADLIQ is safe or effective in children.
Do not take TADLIQ if you:
- take any medicines called nitrates
- use recreational drugs called “poppers” like amyl nitrate, butyl nitrate or nitrite
- take any medicines called guanylate cyclase stimulators
- are allergic to tadalafil (TADLIQ or the medicines ADCIRCA or CIALIS) or any other ingredient in TADLIQ. See the end of this leaflet for a complete list of ingredients in TADLIQ.
Before you take TADLIQ, tell your healthcare provider about all of your medical conditions, including if you:
- have low blood pressure
- have pulmonary veno-occlusive disease (PVOD)
- have heart problems
- have liver problems
- have kidney problems or get dialysis
- have retinitis pigmentosa, a rare genetic eye disease
- have ever had any sudden vision loss, including an eye problem called non-arteritic anterior ischemic optic neuropathy (NAION).
- have ever had hearing problems such as ringing in the ears, dizziness, or loss of hearing
- have a deformed penis shape or Peyronie's disease
- have blood cell problems such as sickle cell anemia, multiple myeloma, or leukemia
- are pregnant or plan to become pregnant. It is not known if TADLIQ will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if TADLIQ passes into your breast milk. Talk with your healthcare provider about the best way to feed your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the counter medicines, vitamins, and herbal supplements. TADLIQ and other medicines may affect each other.
Especially tell your healthcare provider if you take:
- nitrates or guanylate cyclase stimulators (see “What is the most important information I should know about TADLIQ?”)
- medicines for erectile dysfunction (impotence). TADLIQ contains tadalafil, which is the same medicine found in another medicine called CIALIS. CIALIS is used for the treatment of male erectile dysfunction and for the signs and symptoms of benign prostatic hyperplasia. Do not take CIALIS or other medicines for erectile dysfunction, or other PDE5 inhibitors during treatment with TADLIQ.
Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take TADLIQ?
- Take TADLIQ exactly as your healthcare provider tells you.
- Take TADLIQ 1 time each day.
- Shake the TADLIQ bottle well for 30 seconds before measuring your dose.
- TADLIQ can be taken with or without food.
- Your healthcare provider may change your dose.
- Do not change your dose or stop taking TADLIQ without speaking to your healthcare provider.
- If you take too much TADLIQ, call your healthcare provider or go to the nearest hospital emergency room right away.
What should I avoid while taking TADLIQ?
Do not have more than 5 alcohol-containing drinks in a short period of time during treatment with TADLIQ. Drinking too much alcohol can increase your chances of low blood pressure, increased heart rate, dizziness and headache.
What are the possible side effects of TADLIQ?
TADLIQ may cause serious side effects, including:
- See “What is the most important information I should know about TADLIQ?”
- Decreased blood pressure. TADLIQ may cause low blood pressure that last for a short time.
- Decreased eyesight or permanent loss of vision in one or both eyes could be a sign of non-arteritic anterior ischemic optic neuropathy (NAION). There is an increased risk of NAION in people who have already experienced NAION in one eye. If you notice a sudden decrease or loss of vision in one or both eyes, contact your healthcare provider right away.
- Sudden decrease or loss of hearing, sometimes with ringing in the ears and dizziness. If you notice a sudden decrease or loss of hearing, contact your healthcare provider right away.
- In men, an erection that lasts more than 4 hours (with or without pain). Talk to your healthcare provider or get emergency medical help right away. An erection that lasts more than 4 hours must be treated as soon as possible or you can have lasting damage to your penis, including the inability to have erections.
The most common side effects of TADLIQ is headache.
These are not all of the possible side effects of TADLIQ.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store TADLIQ?
- Store TADLIQ at room temperature between 68°F to 77°F (20°C to 25°C).
Keep TADLIQ and all medicines out of the reach of children.
General information about the safe and effective use of TADLIQ.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use TADLIQ for a condition for which it was not prescribed. Do not give TADLIQ to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about TADLIQ that is written for healthcare professionals.
What are the ingredients in TADLIQ?
Active Ingredient: tadalafil
Inactive Ingredients: 30% simethicone emulsion, citric acid monohydrate, glycerin, peppermint flavor, polysorbate 80, purified water, sodium benzoate, sucralose, tri-sodium citrate dihydrate, xanthan gum.
Distributed by: CMP Pharma, Inc., 8026 US Highway 264A, Farmville, NC 27828
For more information, go to www.TADLIQ.com or call 1-844-321-1443
This Patient Information has been approved by the U.S. Food and Drug Administration.
Issued: 10/2022