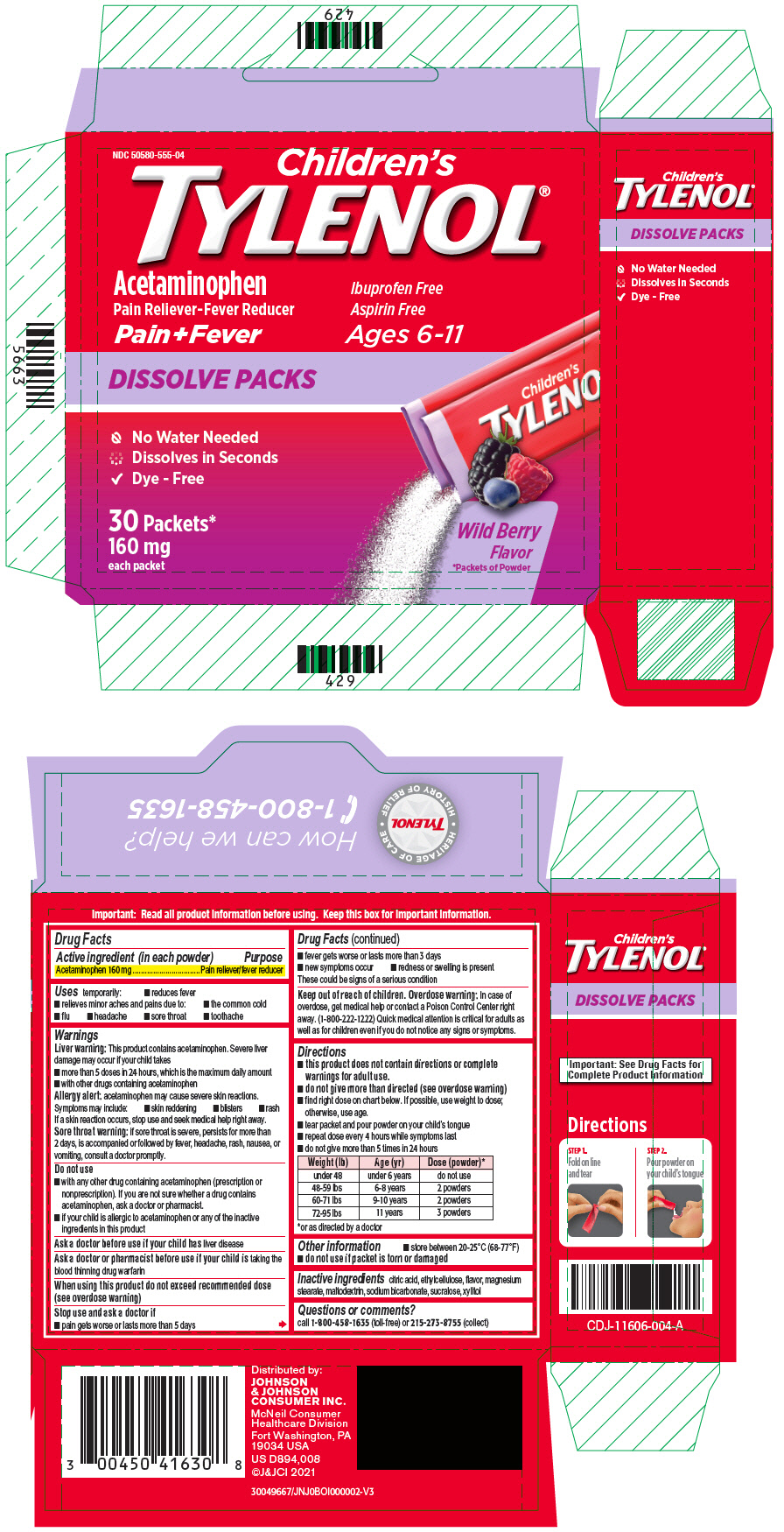
Uses
temporarily:
- reduces fever
- relieves minor aches and pains due to:
- the common cold
- flu
- headache
- sore throat
- toothache
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if your child takes
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if your child is allergic to acetaminophen or any of the inactive ingredients in this product
When using this product do not exceed recommended dose (see overdose warning)
Directions
- this product does not contain directions or complete warnings for adult use.
- do not give more than directed (see overdose warning)
- find right dose on chart below. If possible, use weight to dose; otherwise, use age.
- tear packet and pour powder on your child's tongue
- repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
| Weight (lb) | Age (yr) | Dose (powder) * |
|---|---|---|
|
||
| under 48 | under 6 years | do not use |
| 48-59 lbs | 6-8 years | 2 powders |
| 60-71 lbs | 9-10 years | 2 powders |
| 72-95 lbs | 11 years | 3 powders |