DESCRIPTION
Fluocinolone Acetonide Oil 0.01% (Ear Drops) contains fluocinolone acetonide {(6α,11β,16α)-6,9-difluoro-11,21-dihydroxy- 16,17[(1-methylethylidene)bis(oxy)]-pregna-1,4-diene-3,20-dione, cyclic 16,17 acetal with acetone}, a synthetic corticosteroid. Chemically, fluocinolone acetonide is C24H30F2O6. It has the following structural formula:
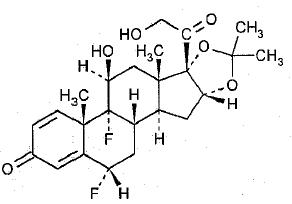
Fluocinolone acetonide in fluocinolone acetonide oil 0.01% has a molecular weight of 452.50. It is a white crystalline powder that is odorless, stable in light, and melts at 270°C with decomposition; soluble in alcohol, acetone and methanol; slightly soluble in chloroform; insoluble in water.
Each gram of fluocinolone acetonide oil 0.01% contains approximately 0.11 mg of fluocinolone acetonide in a blend of oils, which contains isopropyl alcohol, isopropyl myristate, light mineral oil, oleth-2 and refined peanut oil NF.
CLINICAL PHARMACOLOGY
Like other topical corticosteroids, fluocinolone acetonide has anti-inflammatory, antipruritic, and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Pharmacokinetics: The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle and the integrity of the epidermal barrier. Occlusion of topical corticosteroids can enhance penetration. Topical corticosteroids can be absorbed from normal intact skin. Also, inflammation and/or other disease processes in the skin can increase percutaneous absorption.
Fluocinolone acetonide oil 0.01% is in the low to medium range of potency as compared with other topical corticosteroids.
CLINICAL STUDIES
Efficacy in a placebo-controlled study for the treatment of chronic eczematous external otitis on 154 patients (adults and children 2 years of age and older) treated with five drops per ear of fluocinolone acetonide oil 0.01% twice daily, after 7 days of treatment, showed fluocinolone acetonide oil 0.01% to be superior to placebo in clearing the signs and symptoms of eczematous external otitis.
Clinical safety studies were conducted on the same formulation of fluocinolone acetonide oil 0.01%, marketed as fluocinolone acetonide topical oil, 0.01%. Open-label safety studies on 33 children (20 subjects ages 2 to 6 years, 13 subjects ages 7 to 12 years) with moderate to severe stable atopic dermatitis, and baseline body surface area involvement greater than 75% in 18 patients, and 50% to 75% in 15 patients, were treated with fluocinolone acetonide topical oil, 0.01% twice daily for 4 weeks. Morning pre-stimulation cortisol level and post-Cortrosyn stimulation cortisol level were obtained in each subject at the beginning of the trial and at the end of 4 weeks of treatment. At the end of treatment, 4 out of 18 subjects aged 2 to 5 years showed low pre-stimulation cortisol levels (3.2 to 6.6μg/dL; normal: cortisol > 7μg/dL) but all had normal responses to 0.25 mg of Cortrosyn stimulation (cortisol > 18 μg/dL).
A clinical study was conducted to assess the safety of fluocinolone acetonide topical oil, 0.01%, which contains refined peanut oil, on subjects with known peanut allergies. The study enrolled 13 patients with atopic dermatitis, 6 to 17 years of age. Of the 13 patients, 9 were Radioallergosorbent Test (RAST) positive to peanuts and 4 had no peanut sensitivity (controls). The study evaluated the responses to both prick test and patch test utilizing peanut oil NF, fluocinolone acetonide topical oil, 0.01% and histamine/saline controls on the 13 individuals. These subjects were also treated with fluocinolone acetonide topical oil, 0.01% twice daily for 7 days. Prick test and patch test results for all 13 patients were negative to fluocinolone acetonide topical oil, 0.01% and the refined peanut oil. One of the 9 peanut-sensitive patients experienced an exacerbation of atopic dermatitis after 5 days of fluocinolone acetonide topical oil, 0.01% use. Importantly, the bulk peanut oil NF, used in fluocinolone acetonide topical oil, 0.01% is heated between 232˚C – 246˚C (450˚F – 475˚F) for at least 15 minutes, which should provide for adequate decomposition of allergenic proteins.
INDICATION AND USAGE
Fluocinolone acetonide oil 0.01% is a low to medium potency corticosteroid indicated for the treatment of chronic eczematous external otitis in adults and pediatric patients 2 years and older.
CONTRAINDICATIONS
Fluocinolone acetonide oil 0.01% is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
This product contains refined peanut oil NF (see PRECAUTIONS).
PRECAUTIONS
General: Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal of treatment. Manifestations of Cushing's syndrome, hyperglycemia, and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment.
Patients applying a topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using the ACTH stimulation, A.M. plasma cortisol, and urinary free cortisol tests.
If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent corticosteroid. Infrequently, signs and symptoms of glucocorticoid insufficiency may occur requiring supplemental systemic corticosteroids. For information on systemic supplementation, see prescribing information for those products.
Children may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios. (See PRECAUTIONS-Pediatric use)
Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than noting a clinical exacerbation, which may occur with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic testing. One peanut-sensitive child experienced a flare of his atopic dermatitis after 5 days of twice daily treatment with fluocinolone acetonide topical oil, 0.01% (see CLINICAL STUDIES section).
If wheal and flare type reactions (which may be limited to pruritus) or other manifestations of hypersensitivity develop, fluocinolone acetonide topical oil, 0.01%) should be discontinued immediately and appropriate therapy instituted.
If concomitant skin infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of fluocinolone acetonide topical oil, 0.01% should be discontinued until the infection has been adequately controlled.
Fluocinolone acetonide oil 0.01% is formulated with 48% refined peanut oil NF. Physicians should use caution in prescribing fluocinolone acetonide oil 0.01% for peanut-sensitive individuals.
Information for Patients: Patients using topical corticosteroids should receive the following information and instructions:
- This medication is to be used as directed by the physician. It is for external ear use only. Do not use occlusive dressings.
- Avoid contact with the eyes. In case of contact, wash eyes liberally with water.
- This medication should not be used for any disorder other than that for which it was prescribed.
- Patients should promptly report to their physician any worsening of their skin condition.
- As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician.
Laboratory Tests: The following tests may be helpful in evaluating patients for HPA axis suppression:
ACTH stimulation test
A.M. plasma cortisol test
Urinary free cortisol test
Carcinogenesis, mutagenesis, and impairment of fertility: Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of fluocinolone acetonide oil 0.01%. Studies have not been performed to evaluate the mutagenic potential of fluocinolone acetonide, the active ingredient in fluocinolone acetonide oil 0.01%. Some corticosteroids have been found to be genotoxic in various genotoxicity tests (i.e. the in vitro human peripheral blood lymphocyte chromosome aberration assay with metabolic activation, the in vivo mouse bone marrow micronucleus assay, the Chinese hamster micronucleus test and the in vitro mouse lymphoma gene
mutation assay).
Pregnancy: Teratogenic effects: Pregnancy category C: Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals.
There are no adequate and well-controlled studies in pregnant women on teratogenic effects from fluocinolone acetonide oil 0.01%. Therefore, fluocinolone acetonide oil 0.01% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers: Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are excreted in human milk, caution should be exercised when fluocinolone acetonide oil 0.01% is administered to a nursing woman.
Pediatric Use: Fluocinolone acetonide oil 0.01% may be used twice daily for up to 2 weeks in pediatric patients 2 years of age and older with chronic eczematous external otitis.
Fluocinolone acetonide oil 0.01% is not recommended for use on the face (See ADVERSE REACTIONS section).
Because of a higher ratio of skin surface area to body mass, children are at a greater risk than adults of HPA-axis-suppression when they are treated with topical corticosteroids. They are therefore also at greater risk of glucocorticosteroid insufficiency after withdrawal of treatment and of Cushing's syndrome while on treatment. Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children. (See PRECAUTIONS).
HPA axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Children may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Fluocinolone acetonide oil 0.01% is formulated with 48% refined peanut oil NF. Physicians should use caution in prescribing fluocinolone acetonide oil 0.01% for peanut-sensitive individuals.
ADVERSE REACTIONS
The following local adverse reactions have been reported infrequently with topical corticosteroids. They may occur more frequently with the use of occlusive dressings, especially with higher potency corticosteroids. These reactions are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, and miliaria. One peanut sensitive child experienced a flare of his atopic dermatitis after 5 days of twice daily treatment with Fluocinolone Acetonide Topical Oil, 0.01%.
To report SUSPECTED ADVERSE REACTIONS, contact Rising Pharmaceuticals, Inc. at 1-866-562-4597 or FDA at 1-800-FDA-1088 or www.FDA.gov/medwatch.
OVERDOSAGE
Topically applied fluocinolone acetonide oil 0.01% can be absorbed in sufficient amounts to produce systemic effectss (See PRECAUTIONS).
DOSAGE AND ADMINISTRATION
For the treatment of chronic eczematous external otitis, using the supplied ear-dropper, apply 5 drops of fluocinolone acetonide oil 0.01% into the affected ear. To apply, tilt head to one side so that the ear is facing up. Then gently pull the ear lobe backward and upward and apply 5 drops of fluocinolone acetonide oil 0.01% into the ear. Keep head tilted for about a minute to allow fluocinolone acetonide oil 0.01% to penetrate lower into the ear canal. Gently pat excess material dripping out of the ear using a clean cotton ball. Follow these instructions twice each day for 7 to 14 days.
HOW SUPPLIED
Fluocinolone Acetonide Oil 0.01% (Ear Drops) is supplied in 1 fluid ounce bottles containing 20 mL (Dropper Included) (NDC # 64980-329-20).
Keep tightly closed. Store at 20°-25°C (68°to 77°F); excursions permitted to 15°-30°C (59°-86°F). [see USP Controlled Room Temperature].
Discard Fluocinolone Acetonide Oil 0.01%(Ear Drops) 2 months after initial use.
CAUTION: Rx only
Manufactured by:
Lyne Laboratories, Inc., Brockton, MA 02301
Manufactured for:
Rising Pharmaceuticals, Inc., Saddle Brook, NJ 07663
R1-04/21

