CIPROFLOXACIN AND FLUOCINOLONE ACETONIDE- ciprofloxacin and fluocinolone acetonide solution
Wilshire Pharmaceuticals
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Ciprofloxacin and Fluocinolone Acetonide Otic Solution safely and effectively. See full prescribing information for Ciprofloxacin and Fluocinolone Acetonide Otic Solution.
Ciprofloxacin and Fluocinolone Acetonide otic solution Initial U.S. Approval: 2016 INDICATIONS AND USAGECiprofloxacin and Fluocinolone Acetonide Otic Solution is a combination of ciprofloxacin, a fluoroquinolone antibacterial, and fluocinolone acetonide, a corticosteroid, indicated for the treatment of acute otitis media with tympanostomy tubes (AOMT) in pediatric patients (aged 6 months and older) due to Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa ( 1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSOtic Solution: Each single-dose vial of Ciprofloxacin and Fluocinolone Acetonide Otic Solution (ciprofloxacin 0.3 % and fluocinolone acetonide 0.025 %) delivers 0.25 mL of solution equivalent to ciprofloxacin 0.75 mg and fluocinolone acetonide 0.0625 mg. (3) CONTRAINDICATIONSCiprofloxacin and Fluocinolone Acetonide Otic Solution is contraindicated in:
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions that occurred in ≥1 patient were otorrhea, excessive granulation tissue, ear infection, ear pruritus, tympanic membrane disorder, auricular swelling and balance disorder ( 6.1) To report SUSPECTED ADVERSE REACTIONS, contact Wilshire Pharmaceuticals at 1-877-495-6856 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION. Revised: 5/2019 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Ciprofloxacin and Fluocinolone Acetonide Otic Solution is indicated for the treatment of acute otitis media with tympanostomy tubes (AOMT) in pediatric patients (aged 6 months and older) due to Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa.
2 DOSAGE AND ADMINISTRATION
- Ciprofloxacin and Fluocinolone Acetonide Otic Solution is for otic use only. It is not for ophthalmic use, or for injection.
The recommended dosage regimen is as follows:
- Instill the contents of one single-dose vial 0.25 mL into the affected ear canal twice daily (approximately every 12 hours) for 7 days. Use this dosing for patients aged 6 months of age and older.
- Warm the solution by holding the vial in the hand for 1 to 2 minutes. This is to avoid dizziness, which may result from the instillation of a cold solution into the ear canal.
- The patient should lie with the affected ear upward, and then instill the medication.
- Pump the tragus 4 times by pushing inward to facilitate penetration of the medication into the middle ear.
- Maintain this position for 1 minute. Repeat, if necessary, for the opposite ear [see Instructions for Use] .
3 DOSAGE FORMS AND STRENGTHS
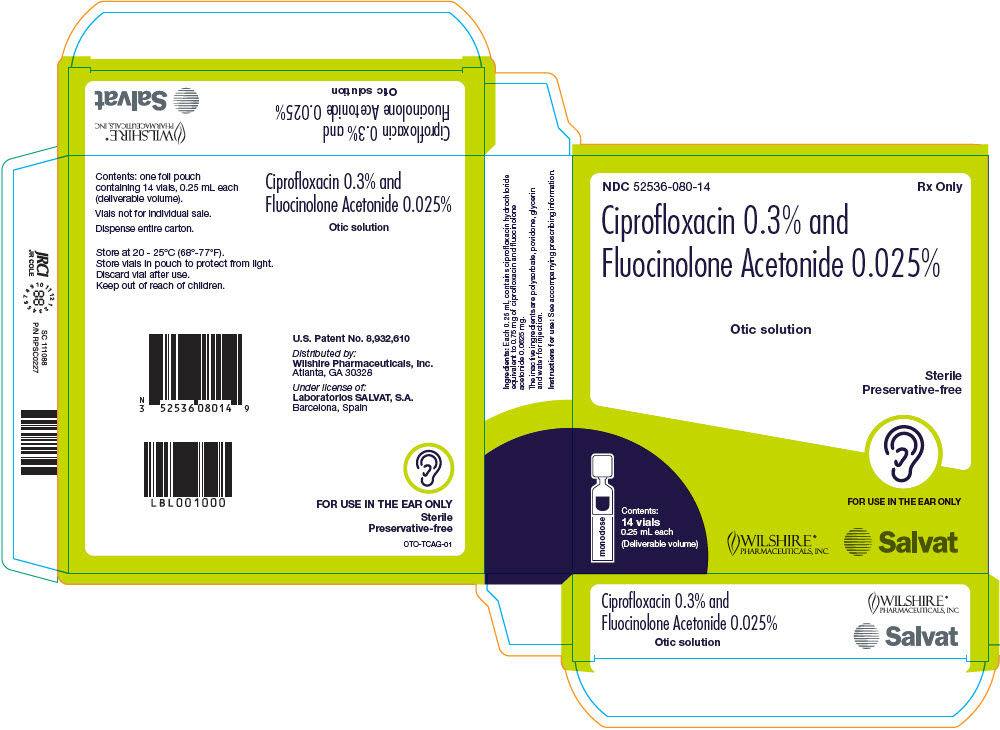
Otic Solution: Each single-dose vial of Ciprofloxacin and Fluocinolone Acetonide Otic Solution (ciprofloxacin 0.3 % and fluocinolone acetonide 0.025 %) delivers 0.25 mL of solution equivalent to ciprofloxacin 0.75 mg and fluocinolone acetonide 0.0625 mg.
4 CONTRAINDICATIONS
Ciprofloxacin and Fluocinolone Acetonide Otic Solution is contraindicated in:
- Patients with known hypersensitivity to fluocinolone acetonide or other corticosteroids, ciprofloxacin or other quinolones, or to any other components of Ciprofloxacin and Fluocinolone Acetonide Otic Solution.
- Viral infections of the external ear canal, including varicella and herpes simplex infections and fungal otic infections.
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Ciprofloxacin and Fluocinolone Acetonide Otic Solution should be discontinued at the first appearance of a skin rash or any other sign of hypersensitivity. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions, some following the first dose, have been reported in patients receiving systemic quinolones. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria and itching. Serious acute hypersensitivity reactions may require immediate emergency treatment.
5.2 Potential for Microbial Overgrowth with Prolonged Use
Prolonged use of Ciprofloxacin and Fluocinolone Acetonide Otic Solution may result in overgrowth of non-susceptible bacteria and fungi. If the infection is not improved after one week of treatment, cultures should be obtained to guide further treatment. If such infections occur, discontinue use and institute alternative therapy.
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
Hypersensitivity Reactions [ see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, 224 patients with AOMT were treated with Ciprofloxacin and Fluocinolone Acetonide Otic Solution for a median duration of 7 days. All the patients received at least one dose of Ciprofloxacin and Fluocinolone Acetonide Otic Solution. There were 220 patients who received at least one dose of ciprofloxacin (CIPRO) and 213 patients received at least one dose of fluocinolone acetonide (FLUO).
The most common adverse reactions that occurred in 1 or more patients are as follows:
| Adverse Reactions * | Number (%) of Patients | ||
|---|---|---|---|
| Ciprofloxacin and Fluocinolone Acetonide Otic Solution
N=224 | CIPRO
N=220 | FLUO
N=213 |
|
|
|||
| Otorrhea | 12 (5.4%) | 9 (4.1%) | 12 (5.6%) |
| Excessive granulation tissue | 3 (1.3%) | 0 (0.0%) | 2 (0.9%) |
| Ear infection | 2 (0.9%) | 3 (1.4%) | 1 (0.5%) |
| Ear pruritus | 2 (0.9%) | 1 (0.5%) | 1 (0.5%) |
| Tympanic membrane disorder | 2 (0.9%) | 0 (0.0%) | 0 (0.0%) |
| Auricular swelling | 1 (0.4%) | 1 (0.5%) | 0 (0.0%) |
| Balance disorder | 1 (0.4%) | 0 (0.0%) | 0 (0.0%) |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ciprofloxacin and fluocinolone acetonide otic solution, 0.3% / 0.025% outside the US. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Immune system disorders:allergic reaction.
- Infections and infestations:candidiasis.
- Nervous system disorders:dysgeusia, paresthesia (tingling in ears), dizziness, headache.
- Ear and labyrinth disorders:ear discomfort, hypoacusis, tinnitus, ear congestion.
- Vascular disorders:flushing.
- Skin and subcutaneous tissue disorders:skin exfoliation.
- Injury, poisoning and procedural complications:device occlusion (tympanostomy tube obstruction).
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Ciprofloxacin and Fluocinolone Acetonide Otic Solution is negligibly absorbed following otic administration and maternal use is not expected to result in fetal exposure to ciprofloxacin and fluocinolone acetonide [see Clinical Pharmacology (12.3)].
8.2 Lactation
Risk Summary
Ciprofloxacin and Fluocinolone Acetonide Otic Solution is negligibly absorbed by the mother following otic administration and breastfeeding is not expected to result in exposure of the infant to ciprofloxacin and fluocinolone acetonide [ see Clinical Pharmacology (12.3)].
8.4 Pediatric Use
Ciprofloxacin and Fluocinolone Acetonide Otic Solution has been studied in patients as young as 6 months in adequate and well-controlled clinical trials. No major differences in safety and effectiveness have been observed between adult and pediatric patients [ see Indications and Usage (1)and Dosage and Administration (2)].
8.5 Geriatric Use
Clinical studies of Ciprofloxacin and Fluocinolone Acetonide Otic Solution did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
10 OVERDOSAGE
Due to the characteristics of this preparation, no toxic effects are to be expected with an otic overdose of Ciprofloxacin and Fluocinolone Acetonide Otic Solution.
11 DESCRIPTION
Ciprofloxacin and Fluocinolone Acetonide Otic Solution, 0.3% / 0.025% is a sterile, preservative-free, clear otic solution containing the fluoroquinolone antibacterial, ciprofloxacin hydrochloride, combined with the corticosteroid, fluocinolone acetonide. Each single-dose vial contains a deliverable volume of 0.25 mL solution of ciprofloxacin hydrochloride equivalent to 0.75 mg ciprofloxacin and 0.0625 mg fluocinolone acetonide. The pH of the solution ranges from 3.5 to 5.0. The inactive ingredients are polysorbate 80, glycerin, povidone K90F and water for injection.
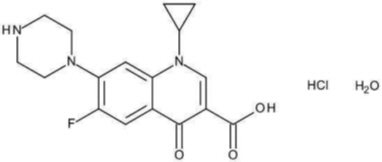
Ciprofloxacin is available as the monohydrochloride, monohydrate salt of 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinolinecarboxylic acid. Its molecular formula is C 17H 18FN 3O 3∙HCl∙H 2O.
The chemical structure of ciprofloxacin hydrochloride is:
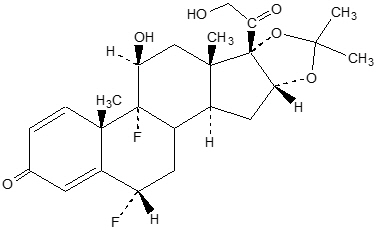
The chemical name of fluocinolone acetonide is (6α,11β,16α)-6,9-difluoro-11,21-dihydroxy- 16,17[(1-methylethylidene)bis(oxy)]-pregna-1,4-diene-3,20-dione, cyclic 16,17 acetal with acetone[67-73-2]. Its molecular formula is C 24H 30F 2O 6.
The chemical structure of fluocinolone acetonide is:
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ciprofloxacin is a fluoroquinolone antibacterial [ see Microbiology (12.4)].
Fluocinolone acetonide, a corticosteroid, inhibits the local biosynthesis of prostaglandins, which explains part of its anti-inflammatory efficacy. At the cellular level, corticosteroids induce peptides called lipocortins. Lipocortins antagonize phospholipase A2, an enzyme which causes the breakdown of leukocyte lysosomal membranes to release arachidonic acid. This action decreases the subsequent formation and release of endogenous inflammatory mediators including prostaglandins, kinins, histamine, liposomal enzymes and the complement system.
12.3 Pharmacokinetics
In two studies in children with AOMT aged ≥ 6 months to 12 years, blood samples were taken in subgroups of 16 and 14 patients, at Visit 1 (prior to the first dose) and Visit 3 (within 1 and 2 hours after the last dose) respectively, to determine the plasma concentrations of ciprofloxacin and/or fluocinolone acetonide following administration of Ciprofloxacin and Fluocinolone Acetonide Otic Solution at the recommended dosage regimen of 0.25 mL twice daily. Pharmacokinetic (PK) analysis resulted in only 1 sample showing a detectable concentration of ciprofloxacin in plasma of 3.0 mcg/L after 7 days of treatment, and no detectable concentrations in plasma of fluocinolone acetonide were observed. However, the sample with detectable ciprofloxacin concentrations was from a patient who had bilateral AOMT (protocol deviation because all patients participating in the PK study were to have unilateral otorrhea) and who received treatment in both ears with ciprofloxacin 0.3% otic solution, the active comparator.
12.4 Microbiology
Mechanism of Action
The bactericidal action of ciprofloxacin results from interference with the enzyme DNA gyrase, which is needed for the synthesis of bacterial DNA.
Resistance
Bacterial resistance to quinolones can develop through chromosomal or plasmid-mediated mechanisms.
In vitrostudies demonstrated cross-resistance between ciprofloxacin and some fluoroquinolones. There is generally no cross-resistance between ciprofloxacin and other classes of antibacterial agents such as beta-lactams or aminoglycosides.
Antimicrobial Activity
Ciprofloxacin has been shown to be active against most isolates of the following bacteria, both in vitroand clinically in otic infections [ see Indications and Usage (1)]:
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No long term studies of Ciprofloxacin and Fluocinolone Acetonide Otic Solution have been performed to evaluate carcinogenic potential.
Long-term carcinogenicity studies in mice and rats have been completed for ciprofloxacin. After daily oral doses of 750 mg/kg (mice) and 250 mg/kg (rats) were administered for up to 2 years, there was no evidence that ciprofloxacin had any carcinogenic or tumorigenic effects in these species.
Long-term animal studies have not been performed to evaluate the carcinogenic potential of fluocinolone acetonide.
Mutagenesis
Eight in vitromutagenicity tests have been conducted with ciprofloxacin, and the test results are listed below:
- Salmonella/Microsome Test (Negative)
- E. coliDNA Repair Assay (Negative)
- Mouse Lymphoma Cell Forward Mutation Assay (Positive)
- Chinese Hamster V79 Cell HGPRT Test (Negative)
- Syrian Hamster Embryo Cell Transformation Assay (Negative)
- Saccharomyces cerevisiaePoint Mutation Assay (Negative)
- Saccharomyces cerevisiaeMitotic Crossover and Gene Conversion Assay (Negative)
- Rat Hepatocyte DNA Repair Assay (Positive)
Thus, 2 of the 8 tests were positive, but results of the following 3 in vivotest systems gave negative results:
- Rat Hepatocyte DNA Repair Assay
- Micronucleus Test (Mice)
- Dominant Lethal Test (Mice)
Studies have not been performed to evaluate the mutagenic potential of fluocinolone acetonide. Some corticosteroids have been found to be genotoxic.
Impairment of Fertility
No reproduction toxicity studies were conducted with Ciprofloxacin and Fluocinolone Acetonide Otic Solution. Absorption of ciprofloxacin and fluocinolone acetonide following otic administration of Ciprofloxacin and Fluocinolone Acetonide Otic Solution at the recommended dosage is negligible [ see Clinical Pharmacology (12.3)].
14 CLINICAL STUDIES
Two phase 3 multicenter, randomized, double-blind, active-controlled, parallel group trials were conducted in 662 pediatric patients in total (aged 6 months to 12 years old) with AOMT, to assess the efficacy and safety of Ciprofloxacin and Fluocinolone Acetonide Otic Solution compared to ciprofloxacin otic solution and to fluocinolone acetonide otic solution (Trial 1 and Trial 2).
In both trials, the Ciprofloxacin and Fluocinolone Acetonide Otic Solution treatment arms showed significantly shorter times to cessation of otorrhea in comparison to both the ciprofloxacin and fluocinolone acetonide alone arms demonstrating the contribution of both components of Ciprofloxacin and Fluocinolone Acetonide Otic Solution. The results are presented in the table below:
| Treatment arm | |||
|---|---|---|---|
| n.e.: not estimable because the number of censored patients was greater than the number of patients with cessation of otorrhea | |||
| Trial 1 | Ciprofloxacin and Fluocinolone Acetonide Otic Solution
(N=112) | CIPRO
(N=109) | FLUO
(N=110) |
| Number (%) with cessation of otorrhea by Day 22 | 88 (78.6%) | 73 (67.0%) | 53 (48.2%) |
| Median time to cessation *(days) | 3.75 | 7.69 | n.e. |
| p-value vs Ciprofloxacin and Fluocinolone Acetonide Otic Solution † | <0.001 | <0.001 | |
| Trial 2 | Ciprofloxacin and Fluocinolone Acetonide Otic Solution
(N=111) | CIPRO
(N=112) | FLUO
(N=108) |
| Number (%) with cessation of otorrhea by Day 22 | 87 (78.4%) | 77 (68.8%) | 47 (43.5%) |
| Median time to cessation *(days) | 4.94 | 6.83 | n.e. |
| p-value vs Ciprofloxacin and Fluocinolone Acetonide Otic Solution † | 0.028 | <0.001 | |
16 HOW SUPPLIED/STORAGE AND HANDLING
How supplied
Ciprofloxacin and Fluocinolone Acetonide Otic Solution, 0.3 %/0.025 %, is a sterile, preservative-free, clear otic solution supplied in blue translucent single-dose 0.25 mL vials. Fourteen single-dose vials are packaged in a protective foil pouch contained in a carton (NDC 52536-080-14).
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling ( Patient Informationand Instructions for Use).
Administration Instructions
- Advise patients that Ciprofloxacin and Fluocinolone Acetonide Otic Solution is for otic use only. It is not to be used in the eyes.
- Advise patients to warm the otic solution by holding the vial in the hand for 1 to 2 minutes before instilling it in the ear, to avoid dizziness.
Hypersensitivity Reactions
- Advise patients to immediately discontinue Ciprofloxacin and Fluocinolone Acetonide Otic Solution at the first appearance of a skin rash or any other sign of hypersensitivity [see Warnings and Precautions (5.1)]
Ciprofloxacin and Fluocinolone Acetonide Otic Solution is:
Distributed by:
Wilshire Pharmaceuticals, Inc.
Atlanta, GA 30328
Under license of Laboratorios SALVAT, S.A.
U.S. Patent No: 8,932,610
OTO-PIAG-00
INSTRUCTIONS FOR USE
CIPROFLOXACIN AND FLUOCINOLONE ACETONIDE
(sip-roh-flok-suh-sin and floo-oh-SIN-oh-lone uh-SEET-oh-nide)
Otic Solution
Read this Instructions for Use that comes with Ciprofloxacin and Fluocinolone Acetonide Otic Solution before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
Important information about Ciprofloxacin and Fluocinolone Acetonide Otic Solution:
- Ciprofloxacin and Fluocinolone Acetonide Otic Solution is for use in the ear only (otic use). Do not inject Ciprofloxacin and Fluocinolone Acetonide Otic Solution or use Ciprofloxacin and Fluocinolone Acetonide Otic Solution in the eye.
- Use Ciprofloxacin and Fluocinolone Acetonide Otic Solution exactly as your healthcare provider tells you to use it.
How should I use Ciprofloxacin and Fluocinolone Acetonide Otic Solution?
| Step 1. | You or your caregiver should wash their hands with soap and water. |  |
| Step 2. | Gently clean any fluid (discharge) from the outer ear using a clean cloth or tissue. Do notput a cotton swab or any other object in the ear canal. | 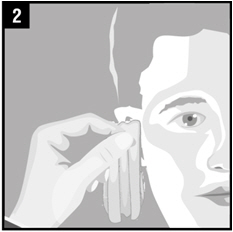 |
| Step 3. | Remove Ciprofloxacin and Fluocinolone Acetonide Otic Solution from the protective foil pouch. Pull apart 1 single-dose vial of Ciprofloxacin and Fluocinolone Acetonide Otic Solution as shown, by tearing along the dotted lines (perforations) until it is fully separated. | 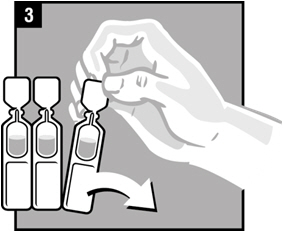 |
| Step 4. | Warm the dose of Ciprofloxacin and Fluocinolone Acetonide Otic Solution by holding the vial in your hand for 1 to 2 minutes. | 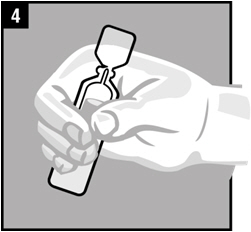 |
| Step 5. | Twist off the vial cap in the direction of the arrow as shown. | 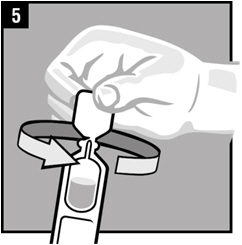 |
| Step 6. | The person receiving Ciprofloxacin and Fluocinolone Acetonide Otic Solution should be on his/her side with the infected ear up as shown. | 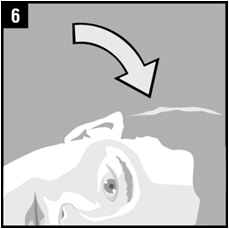 |
| Step 7. | Hold the vial of Ciprofloxacin and Fluocinolone Acetonide Otic Solution in your hand and place the vial close to the ear. Let the entire dose of Ciprofloxacin and Fluocinolone Acetonide Otic Solution fall into the affected ear. | 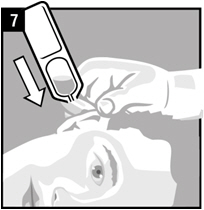 |
| Step 8. | Gently press the part of the ear known as the tragus 4 timesusing a pumping motion as shown. This will allow the drops of Ciprofloxacin and Fluocinolone Acetonide Otic Solution to enter the middle ear. | 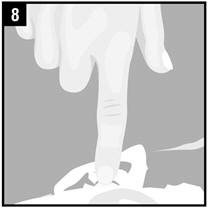 |
| Step 9. | Remain on your side with the affected ear facing upward for 1 minute. | 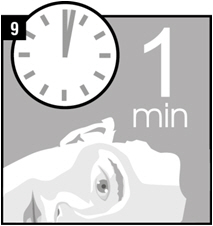 |
| Step 10. | If your healthcare provider has told you to use Ciprofloxacin and Fluocinolone Acetonide Otic Solution in both ears, repeat Steps 2-9 for the other ear. | |
| Step 11. | Safely throw away Ciprofloxacin and Fluocinolone Acetonide Otic Solution vials after use. |
This Instructions for Use has been approved by the Food and Drug Administration.
Rev. 05/2019
| CIPROFLOXACIN AND FLUOCINOLONE ACETONIDE
ciprofloxacin and fluocinolone acetonide solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Wilshire Pharmaceuticals (078657245) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| The Ritedose Corporation | 837769546 | manufacture(52536-080) , pack(52536-080) | |