THERATEARS EXTRA- carboxymethylcellulose sodium, unspecified form solution/ drops
MEDTECH PRODUCTS INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
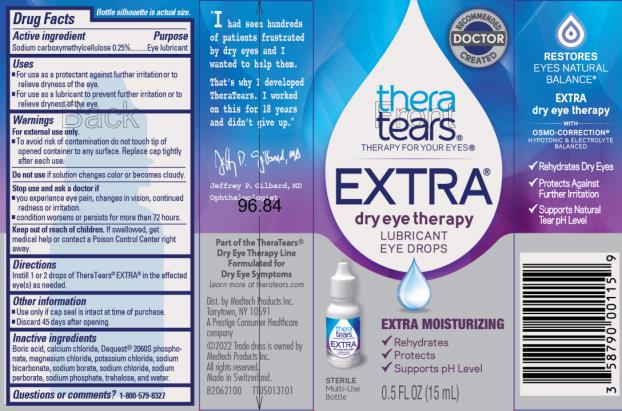
Active ingredient
Sodium carboxymethylcellulose 0.25%
Uses
- For use as a protectant against further irritation or to relieve dryness of the eye.
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Warnings
For external use only
- To avoid risk of contamination do not touch tip of opened container to any surface. Replace cap tightly after each use.
Do not use
- if solution changes color or becomes cloudy.
Stop use and ask a doctor if
- you experience eye pain, changes in vision, continued redness or irritation.
- condition worsens or persists for more than 72 hours.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Instill 1 or 2 drops of TheraTears® Extra® in the affected eye(s) as needed.
Other information
- Do not use if neck ring is broken or missing.
- Discard 45 days after opening.
Inactive ingredients
Boric acid, calcium chloride, Dequest®, 2060S phosphonate, magnesium chloride, potassium chloride, sodium bicarbonate, sodium borate, sodium chloride, sodium perborate, sodium phosphate, trehalose, and water for injection.
Questions or comments?
1-800-579-8327
Principal Display Panel Text for Container Label:
thera
tears®
THERAPY FOR YOUR EYES®
EXTRA®
dry eye therapy
LUBRICANT
EYE DROPS
EXTRA MOISTURIZING
√ Rehydrates
√ Protects
√ Supports pH Level
STERILE
Multi-Use
Bottle TWO 0.5 FL OZ (15 mL) BOTTLES

Principal Display Panel Text for Carton Label:
RECOMMENDED
DOCTOR
CREATED
thera
tears®
THERAPY FOR YOUR EYES®
EXTRA®
dry eye therapy
LUBRICANT
EYE DROPS
EXTRA MOISTURIZING
√ Rehydrates
√ Protects
√ Supports pH Level
STERILE
Multi-Use
Bottle 0.5 FL OZ (15 mL)