HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use NITROGLYCERIN LINGUAL SPRAY safely and effectively. See full prescribing information for NITROGLYCERIN LINGUAL SPRAY .
NITROGLYCERIN LINGUAL SPRAY (nitroglycerin) lingual spray
Initial U.S. Approval: 1997
INDICATIONS AND USAGE
Nitroglycerin Lingual Spray is a nitrate vasodilator indicated for acute relief of an attack or prophylaxis of angina pectoris due to coronary artery disease ( 1).
DOSAGE AND ADMINISTRATION
At the onset of an attack, administer onto or under the tongue. Repeat every 5 minutes as needed (
2-2.1).
Do up to three metered sprays within a 15-minute period. If chest pain persists, advise prompt medical attention (
2-2.1).
May be used prophylactically 5 to 10 minutes prior to engaging in activities that might precipitate an acute attack (
2-2.1).
DOSAGE FORMS AND STRENGTHS
Lingual spray, 400 mcg per spray, available in 60 or 200 metered sprays per container ( 3).
CONTRAINDICATIONS
Use of phosphodiesterase type 5 (PDE-5) Inhibitors, such as avanafil, sildenafil, tadalafil, or vardenafil, or soluble guanylate cyclase (sGC) stimulator (riociguat). (
4-4.1,
7-7.1)
Severe anemia. (
4-4.2)
Increased intracranial pressure. (
4-4.3)
Hypersensitivity to Nitroglycerin Lingual Spray or to other nitrates or nitrites or any excipient. (
4-4.4)
WARNINGS AND PRECAUTIONS
Tolerance: Excessive use may lead to tolerance. ( 5-5.1)
Hypotension: Severe hypotension may occur. ( 5-5.2)
ADVERSE REACTIONS
Most common adverse reactions occurring at a frequency greater than 2 % are headache, dizzinesss and paresthesia (6).
To report SUSPECTED ADVERSE REACTIONS, contact Jacksonville Pharmaceuticals, Inc. at 1-800-235-6203 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Antihypertensives: Possible additive hypotensive effects. (
7-7.2)
Ergotamine: increased bioavailability of ergotamine. Avoid concomitant use (
7-7.3)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 8/2018
FULL PRESCRIBING INFORMATION:CONTENTS*
1. Indications & Usage
2. Dosage & Administration
2.1 Recommended Dosage
2.2 Priming
2.3 Administration
3. DOSAGE FORMS AND STRENGTHS
4. Contraindications4.1 PDE-5-Inhibitors and sGC-Stimulators
4.2 Severe Anemia
4.3 Increased Intracranial Pressure
4.4 Hypersensitivity
4.5 Circulatory Failure and Shock
5. Warnings and Precautions
5.1 Tolerance
5.2 Hypotension
5.3 Hypertrophic Obstructive Cardiomyopathy
5.4 Headache
6. ADVERSE REACTIONS6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7. DRUG INTERACTIONS7.1 PDE-5-Inhibitors and sGC-Stimulators
7.2 Antihypertensives
7.3 Ergotamine
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric use
8.5 Geriatric use
10. OVERDOSAGE10.1 Signs and Symptoms, Methemoglobinemia
10.2 Treatment of Overdosage
11. DESCRIPTION
12. CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14. CLINICAL STUDIES16 HOW SUPPLIED/STORAGE AND HANDLING
16. HOW SUPPLIED/STORAGE AND HANDELING17. PATIENT COUNSELING INFORMATION
*Sections or subsections omitted from the full prescribing information are not listed.
1. Indications & Usage
Nitroglycerin Lingual Spray is indicated for acute relief of an attack or prophylaxis of angina pectoris due to coronary artery disease.
2. Dosage & Administration
2.1 Recommended Dosage
Instruct the patient to administer one or two metered sprays (400 mcg of nitroglycerin per spray) at the onset of an attack onto or under the tongue. A spray may be repeated approximately every five minutes as needed. No more than three metered sprays are recommended within a 15-minute period. If the chest pain persists after a total of three sprays, advise prompt medical attention. Nitroglycerin Lingual Spray may be used prophylactically 5 to 10 minutes prior to engaging in activities that might precipitate an acute attack.
2.2 Priming
The pump must be primed prior to the first use. Each metered spray of Nitroglycerin Lingual Spray delivers 48 mg of solution containing 400 mcg of nitroglycerin after an initial priming of five sprays. It will remain adequately primed for 6 weeks. If the product is not used within 6 weeks it can be adequately re-primed with one spray. If the product is not used within 3 months it can be adequately re-primed with up to five sprays. There are 60 or 200 metered sprays per bottle. The total number of available doses is dependent, however, on the number of sprays per use (1 or 2 sprays), and the frequency of priming.
2.3 Administration
Instruct patients that during administration, the patient should rest, ideally in the sitting position. Hold the container vertically with the valve head uppermost and the spray orifice as close to the mouth as possible. Spray the dose preferably onto or under the tongue by pressing the grooved-button firmly and the mouth closed immediately after each dose. THE SPRAY SHOULD NOT BE INHALED. The medication should not be expectorated or the mouth rinsed for 5 to 10 minutes following administration. Instruct patients to familiarize themselves with the position of the spray orifice, which can be identified by the finger rest on top of the valve, in order to facilitate orientation for administration at night [see Patient Information (17)].
The amount of liquid remaining in the container should be checked periodically. The transparent container can be used for continuous monitoring of the consumption. With the container upright and level, check to be sure the end of the center tube extends below the level of the liquid. Once fluid falls below the level of the center tube, remaining sprays will not deliver intended dose.
3. Dosage Forms & Strengths
Lingual spray, 400 mcg per spray available in either 60 or 200 metered sprays per container.
4. Contraindications
4.1 PDE-5-Inhibitors and sGC-Stimulators
Do not use Nitroglycerin Lingual Spray in patients who are taking PDE-5-Inhibitors, such as avanafil, sildenafil, tadalafil, or vardenafil. Concomitant use can cause severe hypotension, syncope, or myocardial ischemia [see Drug Interactions (7.1)].
Do not use Nitroglycerin Lingual Spray in patients who are taking soluble guanylate cyclase (sGC) stimulators, such as riociguat. Concomitant use can cause hypotension.
4.2 Severe Anemia
Nitroglycerin Lingual Spray is contraindicated in patients with severe anemia (large doses of nitroglycerin may cause oxidation of hemoglobin to methemoglobin and could exacerbate anemia).
4.3 Increased Intracranial Pressure
Nitroglycerin Lingual Spray may precipitate or aggravate increased intracranial pressure and thus should not be used in patients with possible increased intracranial pressure (e. g. cerebral hemorrhage or traumatic brain injury).
4.4 Hypersensitivity
Nitroglycerin Lingual Spray is contraindicated in patients who are allergic to nitroglycerin, other nitrates or nitrites or any excipient.
4.5 Circulatory Failure and Shock
Nitroglycerin Lingual Spray is contraindicated in patients with acute circulatory failure or shock.
5. Warnings and Precautions
5.1 Tolerance
Excessive use may lead to the development of tolerance. Only the smallest number of doses required for effective relief of the acute angina attack should be used [see Dosage and Administration (2.1)].
5.2 Hypotension
Severe hypotension, particularly with upright posture, may occur even with small doses of nitroglycerin particularly in patients with constrictive pericarditis, aortic or mitral stenosis, patients who may be volume-depleted, or are already hypotensive. Hypotension induced by nitroglycerin may be accompanied by paradoxical bradycardia and increased angina pectoris. Symptoms of severe hypotension (nausea, vomiting, weakness, pallor, perspiration and collapse/syncope) may occur even with therapeutic doses.
5.3 Hypertrophic Obstructive Cardiomyopathy
Nitrate therapy may aggravate the angina caused by hypertrophic obstructive cardiomyopathy.
5.4 Headache
Nitroglycerin produces dose-related headaches, especially at the start of nitroglycerin therapy, which may be severe and persistent but usually subside with continued use.
6. Adverse Reactions
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Adverse reactions occurring at a frequency greater than 2 % and greater than placebo included: headache, dizziness, and paresthesia.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Nitroglycerin Lingual Spray and other nitroglycerin drugs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or establish a causal relationship to drug exposure.
Neurologic: weakness, drowsiness
Dermatologic: cutaneous vasodilation, flushing, drug rash, exfoliative dermatitits
Gastrointestinal: nausea, vomiting
Respiratory: transient hypoxemia
Cardiovascular: tachycardia
To report SUSPECTED ADVERSE REACTIONS, contact Jacksonville Pharmaceuticals, Inc. at 1-800-235-6203 or FDA at 1-800-332-1088 or www.fda.gov/medwatch.
7. Drug Interactions
7.1 PDE-5-Inhibitors and sGC-Stimulators
Nitroglycerin Lingual Spray is contraindicated in patients who are using a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE-5). PDE-5-Inhibitors such as avanafil, sildenafil, vardenafil, and tadalafil have been shown to potentiate the hypotensive effects of organic nitrates.
Nitroglycerin Lingual Spray is contraindicated in patients who are taking soluble guanylate cyclase (sGC) stimulators. Concomitant use can cause hypotension.
The time course and dose dependence of these interactions have not been studied, and use within a few days of one another is not recommended. Appropriate supportive care for the severe hypotension has not been studied, but it seems reasonable to treat this as a nitrate overdose, with elevation of the extremities and with central volume expansion.
7.2 Antihypertensives
Patients receiving antihypertensive drugs, beta-adrenergic blockers, and nitrates should be observed for possible additive hypotensive effects. Marked orthostatic hypotension has been reported when calcium channel blockers and organic nitrates were used concomitantly.
Beta-adrenergic blockers blunt the reflex tachycardia produced by nitroglycerin without preventing its hypotensive effects. If beta-blockers are used with nitroglycerin in patients with angina pectoris, additional hypotensive effects may occur.
7.3 Ergotamine
Oral administration of nitroglycerin markedly decreases the first-pass metabolism of dihydroergotamine and subsequently increases its oral bioavailability. Ergotamine is known to precipitate angina pectoris. Therefore, patients receiving sublingual nitroglycerin should avoid ergotamine and related drugs or be monitored for symptoms of ergotism if this is not possible.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 8/2018
8. Use in Specific Populations
8.1 Pregnancy
Risk summary
Limited published data on the use of nitroglycerin are insufficient to determine a drug associated risk of major birth defects or miscarriage. In animal reproduction studies, there were no adverse developmental effects when nitroglycerin was administered intravenously to rabbits or intraperitoneally to rats during organogenesis at doses greater than 64-times the human dose [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 – 4% and 15 – 20%, respectively.
Data
Animal Data
No embryotoxic or postnatal development effects were observed with transdermal application in pregnant rabbits and rats at doses up to 240 mg/kg/day for 13 days, at intraperitoneal doses in pregnant rats up to 20 mg/kg/day for 11 days, and at intravenous doses in pregnant rabbits up to 4 mg/kg/day for 13 days.
8.2 Lactation
Risk summary
Sublingual nitroglycerin has not been studied in lactating women. It is not known if nitroglycerin is present in human milk or if nitroglycerin has effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for nitroglycerin and any potential adverse effects on the breastfed child from nitroglycerin or from the underlying maternal condition.
8.4 Pediatric use
Safety and effectiveness of nitroglycerin in pediatric patients have not been established.
8.5 Geriatric use
Clinical studies did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly (greater than or equal to 65 years) and younger (less than 65 years) patients. In general, dose selection for an elderly patient should start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
10. Overdosage
10.1 Signs and Symptoms, Methemoglobinemia
Nitrate overdosage may result in: severe hypotension, persistent throbbing headache, vertigo, palpitation, visual disturbance, flushing and perspiring skin (later becoming cold and cyanotic), nausea and vomiting (possibly with colic and even bloody diarrhea), syncope (especially in the upright posture), methemoglobinemia with cyanosis and anorexia, initial hyperpnea, dyspnea and slow breathing, slow pulse (dicrotic and intermittent), heart block, increased intracranial pressure with cerebral symptoms of confusion and moderate fever, paralysis and coma followed by clonic convulsions, and possibly death due to circulatory collapse.
Case reports of clinically significant methemoglobinemia are rare at conventional doses of organic nitrates. The formation of methemoglobin is dose-related and in the case of genetic abnormalities of hemoglobin that favor methemoglobin formation, even conventional doses of organic nitrates could produce harmful concentrations of methemoglobin.
10.2 Treatment of Overdosage
As hypotension associated with nitroglycerin overdose is the result of venodilatation and arterial hypovolemia, prudent therapy in this situation should be directed toward increase in central fluid volume. No specific antagonist to the vasodilator effects of nitroglycerin is known. Keep the patient recumbent in a shock position and comfortably warm. Passive movement of the extremities may aid venous return. Intravenous infusion of normal saline or similar fluid may also be necessary. Administer oxygen and artificial ventilation, if necessary. If methemoglobinemia is present, administration of methylene blue (1 % solution), 1 – 2 mg per kilogram of body weight intravenously, may be required unless the patient is known to have G-6-PD deficiency. If an excessive quantity of Nitroglycerin Lingual Spray has been recently swallowed gastric lavage may be of use.
As epinephrine is ineffective in reversing the severe hypotensive events associated with overdosage, it is not recommended for resuscitation.
11. Description
Nitroglycerin, an organic nitrate, is a vasodilator which has effects on both arteries and veins. The chemical name for nitroglycerin is 1,2,3-propanetriol trinitrate (C3H5N3O9). The compound has a molecular weight of 227.09. The chemical structure is:
CH2–ONO2
|
CH–ONO2
|
CH2–ONO2
Nitroglycerin Lingual Spray (nitroglycerin lingual spray 400 mcg) is a metered dose spray containing nitroglycerin. This product delivers nitroglycerin (400 mcg per spray, 60 or 200 metered sprays) in the form of spray droplets onto or under the tongue. Inactive ingredients: medium-chain triglycerides, dehydrated alcohol, medium-chain partial glycerides, peppermint oil, sodium lactate, lactic acid.
12. Clinical Pharmacology
12.1 Mechanism of Action
Nitroglycerin forms free radical nitric oxide (NO), which activates guanylate cyclase, resulting in an increase of guanosine 3',5'-monophosphate (cyclic GMP) in smooth muscle and other tissues. This eventually leads to dephosphorylation of myosin light chains, which regulates the contractile state in smooth muscle and results in vasodilatation.
12.2 Pharmacodynamics
The principal pharmacological action of nitroglycerin is relaxation of vascular smooth muscle. Although venous effects predominate, nitroglycerin produces, in a dose-related manner, dilation of both arterial and venous beds. Dilation of the postcapillary vessels, including large veins, promotes peripheral pooling of blood, decreases venous return to the heart, and reduces left ventricular end-diastolic pressure (preload). Nitroglycerin also produces arteriolar relaxation, thereby reducing peripheral vascular resistance and arterial pressure (after load), and dilates large epicardial coronary arteries; however, the extent to which this latter effect contributes to the relief of exertional angina is unclear.
Therapeutic doses of nitroglycerin may reduce systolic, diastolic and mean arterial blood pressure. Effective coronary perfusion pressure is usually maintained, but can be compromised if blood pressure falls excessively or increased heart rate decreases diastolic filling time.
Elevated central venous and pulmonary capillary wedge pressures, and pulmonary and systemic vascular resistance are also reduced by nitroglycerin therapy. Heart rate is usually slightly increased, presumably a reflex response to the fall in blood pressure. Cardiac index may be increased, decreased, or unchanged. Myocardial oxygen consumption or demand (as measured by the pressure-rate product, tension-time index, and stroke-work index) is decreased and a more favorable supply-demand ratio can be achieved. Patients with elevated left ventricular filling pressure and increased systemic vascular resistance in association with a depressed cardiac index are likely to experience an improvement in cardiac index. In contrast, when filling pressures and cardiac index are normal, cardiac index may be slightly reduced following nitroglycerin administration.
12.3 Pharmacokinetics
A liver reductase enzyme is of primary importance in the metabolism of nitroglycerin to glycerol di- and mononitrate metabolites and ultimately to glycerol and organic nitrate. Known sites of extrahepatic metabolism include red blood cells and vascular walls. In addition to nitroglycerin, 2 major metabolites, 1,2- and 1,3-dinitroglycerin are found in plasma. The mean elimination half-life of both 1,2- and 1,3-dinitroglycerin is about 40 minutes. The 1,2- and 1,3-dinitroglycerin metabolites have been reported to possess some pharmacological activity, whereas the glycerol mononitrate metabolites of nitroglycerin are essentially inactive. Higher plasma concentrations of the dinitro metabolites, with their nearly 8-fold longer elimination half-lives, may contribute significantly to the duration of pharmacologic effect.
In a pharmacokinetic study when a single 0.8 mg dose of Nitroglycerin Lingual Spray was administered to healthy volunteers (n = 24), the mean Cmax and tmax were 1,041 pg/ml and 7.5 minutes, respectively. Additionally, in these subjects the mean area under the curve (AUC) was 12,769 pg/ml * min.
The volume of distribution of nitroglycerin following intravenous administration is 3.3 L/kg.
Drug interactions
Aspirin: Coadministration of nitroglycerin with high dose aspirin (1000 mg) results in increased exposure to nitroglycerin. The vasodilatory and hemodynamic effects of nitroglycerin may be enhanced by concomitant administration of nitroglycerin with high dose aspirin.
Tissue-type plasminogen activator (t-PA): Concomitant administration of t-PA and intravenous nitroglycerin has been shown to reduce plasma levels of t-PA and its thrombolytic effect.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal carcinogenesis studies with sublingual nitroglycerin have not been performed.
Rats receiving up to 434 mg/kg/day of dietary nitroglycerin for 2 years developed dose-related fibrotic and neoplastic changes in liver, including carcinomas, and interstitial cell tumors in testes. At high dose, the incidences of hepatocellular carcinomas in both sexes were 52 % vs. 0 % in controls, and incidences of testicular tumors were 52 % vs. 8 % in controls. Lifetime dietary administration of up to 1058 mg/kg/day of nitroglycerin was not tumorigenic in mice.
Nitroglycerin was weakly mutagenic in Ames tests performed in two different laboratories. There was no evidence of mutagenicity in an in vivo dominant lethal assay with male rats treated with doses up to about 363 mg/kg/day, p.o., or in in vitro cytogenic tests in rat and dog tissues and for chromosomal aberration in Chinese hamster ovary cells.
In a three-generation reproduction study, rats received dietary nitroglycerin at doses up to about 434 mg/kg/day for six months prior to mating of the F0 generation with treatment continuing through successive F1 and F2 generations. The high dose was associated with decreased feed intake and body weight gain in both sexes at all matings. No specific effect on the fertility of the F0 generation was seen. Infertility noted in subsequent generations, however, was attributed to increased interstitial cell tissue and aspermatogenesis in the high- dose males. In this three-generation study there was no clear evidence of teratogenicity.
14. Clinical Studies
In a randomized, double-blind single-dose, 5-period cross-over study in 51 patients with exertional angina pectoris significant dose-related increases in exercise tolerance, time to onset of angina and ST-segment depression were seen following doses of 0.2, 0.4, 0.8 and 1.6 mg of nitroglycerin delivered by metered lingual spray as compared to placebo.
The drug showed a profile of mild to moderate adverse events.
16. How Supplied/Storage and Handling
NITROGLYCERIN LINGUAL SPRAY is supplied in the following dosage forms.
NDC 51662-1244-1
NITROGLYCERIN LINGUAL SPRAY 400mcg PER SPRAY 60 METERED SPRAYS
HF Acquisition Co LLC, DBA HealthFirst
Mukilteo, WA 98275
Also supplied in the following manufacture supplied dosage forms
Each box of Nitroglycerin Lingual Spray contains one glass bottle coated with red transparent plastic which assists in containing the glass and medication should the bottle be shattered. Each bottle contains 4.9 g or 14.1 g (Net Contents) of nitroglycerin lingual spray which will deliver 60 or 200 metered sprays containing 400 mcg of nitroglycerin per spray after priming.
Nitroglycerin Lingual Spray is available as:
60-dose (4.9 g) single bottle NDC 70003-300-65
200-dose (14.1 g) single bottle NDC 70003-300-20
Store at 20°C – 25°C (68°F – 77°F); excursions permitted to 15 – 30°C (59 – 86°F) [see USP Controlled Room Temperature].
Note: Nitroglycerin Lingual Spray contains 20 % alcohol. Do not forcefully open or burn container after use. Do not spray toward flames.
Rx Only.
17. Patient Counseling
Advise the patient to read the FDA-approved patient labeling (Instructions for Use)
The following trademarks are either registered trademarks or trademarks of Pohl-Boskamp in the United States and/or other countries: Pohl-Boskamp word mark; Pohl-Boskamp logo; Nitroglycerin Lingual shapes Nitroglycerin Lingual Spray colors.
U.S. Patent No. 7,872,049
Manufactured for
Jacksonville Pharmaceuticals, Inc. Jacksonville, FL 32250
by G. Pohl-Boskamp GmbH & Co. KG,
25551 Hohenlockstedt, Germany.
Patient Package Insert
Instructions for Use
Nitroglycerin Lingual Spray (nī-trō-GLIS-er-in ling´gwal)
(nitroglycerin)
lingual spray
Read this Instructions for Use before you start using Nitroglycerin Lingual Spray and each time you get your prescription refilled. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Important information:
Nitroglycerin Lingual Spray is for use onto or under the tongue. Do not inhale Nitroglycerin Lingual Spray.
A dose of Nitroglycerin Lingual Spray may be either 1 or 2 sprays. Follow your healthcare provider’s instructions about how many sprays you should use for each dose. Doses should be separated by approximately 5 minutes.
You should not use more than 3 sprays of Nitroglycerin Lingual Spray within 15 minutes.
Get emergency medical help right away if you still have chest pain after using a total of 3 sprays of Nitroglycerin Lingual Spray.
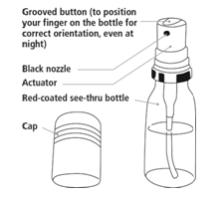
Nitroglycerin Lingual Spray parts:
Figure A
How should I use Nitroglycerin Lingual Spray?
It is best to use Nitroglycerin Lingual Spray while you are resting and in a sitting position.
Do not shake the Nitroglycerin Lingual Spray container.
Priming Nitroglycerin Lingual Spray:
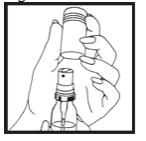
Before you use Nitroglycerin Lingual Spray for the first time, you must prime it. To prime your Nitroglycerin Lingual Spray, follow the steps below:
Step 1. Remove the plastic cap from the container. (See Figure B)
Figure B
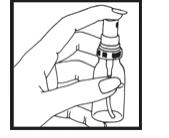
Step 2. Hold the container upright and facing away from yourself and others. Press down on the top of the grooved button 5 times. (See Figure C)
Figure C
Your Nitroglycerin Lingual Spray is now primed. You are ready to give your first dose.
If you do not use your Nitroglycerin Lingual Spray within 6 weeks, you will need to prime it again by pressing down on the top of the grooved button 1 time.
If you do not use your Nitroglycerin Lingual Spray within 3 months, you will need to re-prime it by pressing down on the top of the grooved button up to 5 times.
Giving a dose of Nitroglycerin Lingual Spray:
Step 3. Hold your Nitroglycerin Lingual Spray container upright with your index finger on top of the grooved button.
Step 4. Open your mouth and bring the Nitroglycerin Lingual Spray container as close to your mouth as possible.
Step 5. Press down on the top of the grooved button firmly with your index finger to release the spray onto or under your tongue. (See Figure D). The grooved button can help you make sure the canister is turned to the correct position if you are administering the spray in the dark.
Figure D
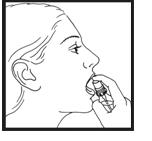
Step 6. Release the grooved button and close your mouth right away. Avoid swallowing right after using Nitroglycerin Lingual Spray. Do not spit out Nitroglycerin Lingual Spray or rinse your mouth for 5 to 10 minutes after using Nitroglycerin Lingual Spray.
Step 7. If a second dose of Nitroglycerin Lingual Spray is needed, repeat Steps 3 through 6 above.
Step 8. Replace the plastic cap.
Check the level of the fluid in your Nitroglycerin Lingual Spray container regularly.
Check the container in an upright position.
The end of the center tube should be covered by the fluid in the Nitroglycerin Lingual Spray container. If the level of the fluid falls below the end of the center tube, sprays will not provide enough Nitroglycerin Lingual Spray.
Replace your Nitroglycerin Lingual Spray container before the fluid level falls below the end of the center tube.
How should I store Nitroglycerin Lingual Spray?
Store Nitroglycerin Lingual Spray at room temperature between 68°F - 77°F (20°C - 25°C).
Do not forcefully open or burn the Nitroglycerin Lingual Spray container after use.
Do not spray Nitroglycerin Lingual Spray toward flames.
Keep Nitroglycerin Lingual Spray and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for Jacksonville Pharmaceuticals, Inc. Jacksonville, FL 32250
by G. Pohl-Boskamp GmbH & Co. KG, 25551 Hohenlockstedt, Germany.
Revised: 07/2018
The following trademarks are either registered trademarks or trademarks of Pohl-Boskamp in the United States and / or other countries: Pohl-Boskamp word mark; Pohl-Boskamp logo; Nitroglycerin Lingual Spray shapes, Nitroglycerin Lingual Spray colors.