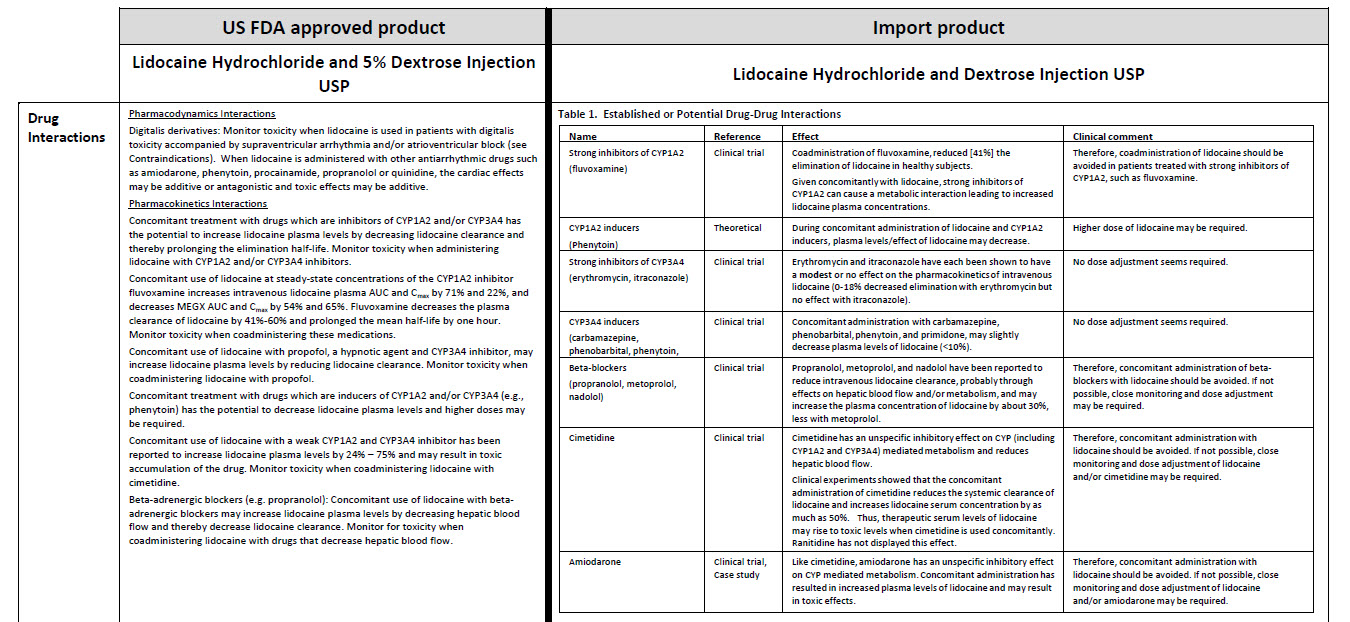
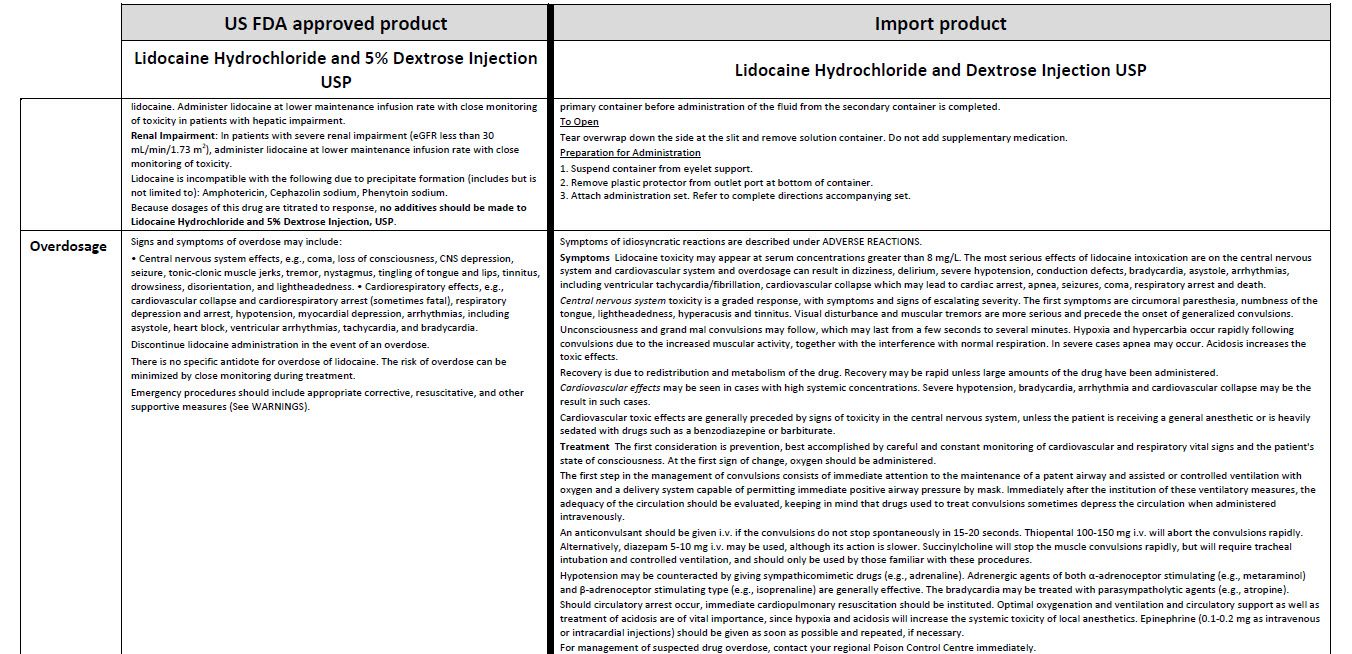
LIDOCAINE HYDROCHLORIDE AND DEXTROSE- lidocaine hydrochloride injection
Baxter Healthcare Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
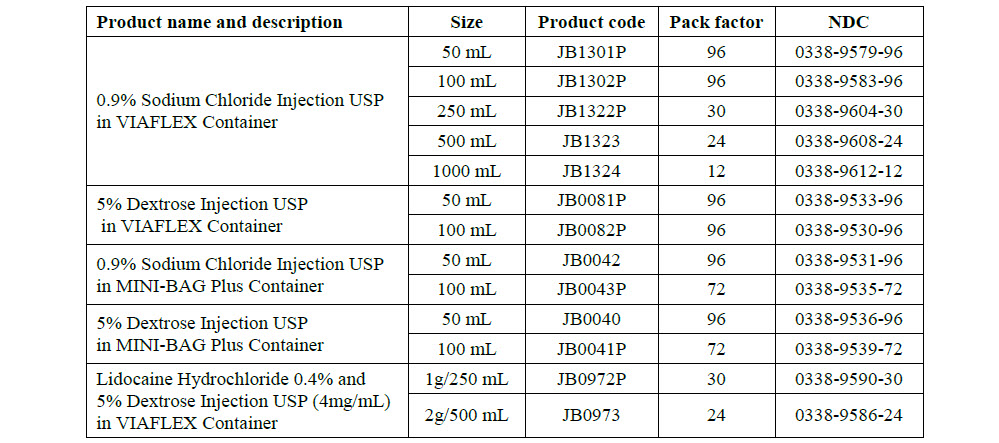
Lidocaine Hydrochloride 0.4% and 5% Dextrose
PACKAGE/LABEL PRINCIPAL DI SPLAY PANEL
JB0972
250 mL
DIN 00828602
Lidocaine Hydrochloride 0.4% and 5% Dextrose Injection /
Chlorhydrate de Lidocaine á 0.4% dans du Dextrose á
5%, Injectable
Lidocaine Hydrochloride and Dextrose Injection USP / Chlorhydrate
de Lidocaine dans du Dextrose USP, Injectable
Lidocaine
4 mg/mL
1 g / 250 mL
Bar Code
(01)00809080000548
EACH mL CONTAINS / CHAQUE mL CONTIENT
LIDOCAINE HYDROCHLORIDE /
CHLORHYDRATE DE LIDOCAINE 4 mg
DEXTROSE / DEXTROSE – 50 mg
SODIUM HYDROXIDE (PH ADJUSTER) /
HYDROXYDE DE SODIUM (AJUSTEUR DE PH)
WATER FOR INJECTION / EAU POUR INJECTION
APPROX mOsmol/L 282 APPROX pH 5.0
ANTIARRHYTHMIC AGENT FOR INTRAVENOUS
INFUSION ONLY / AGENT ANTIARYTHMIQUE
POUR PERFUSION INTRAVEINEUSE SEULEMENT
USUAL DOSAGE SINGLE DOSE 50 mg – 100 mg AT A RATE OF 25 – 50 mg
PER MIN / DO NOT EXCEED 300 mg PER HOUR / CONTINUOUS
INFUSION AT A RATE OF 1 TO 2 mg PER MIN / NOT RECOMMENDED FOR
USE IN CHILDREN
WARNINGS DO NOT ADD SUPPLEMENTARY MEDICATION / MUST NOT
BE USED IN SERIES CONNECTIONS / DO NOT USE SIMULTANEOUSLY
WITH BLOOD
CAUTIONS SQUEEZE AND INSPECT BAG / DO NOT USE UNLESS
SOLUTION IS CLEAR / SINGLE USE / DISCARD UNUSED PORTION /
STORE AT 15ºC TO 25ºC / PRODUCT MONOGRAPH AVAILABLE ON
REQUEST / HEALTHCARE PROFESSIONAL DISPENSE WITH
CONSUMER INFORMATION
POSOLOGIE HABITUELLE DOSE UNIQUE DE 50 mg – 100 MG A UN
DEBIT DE 25-50 mg PAR MIN / NE PAS DEPASSER 300 mg PAR HEURE /
PERFUSION CONTINUE A UN DEBIT DE 1 A 2 mg PAR MIN / N’EST PAS
RECOMMANDE EN PEDIATRIE
MISES EN GARDE NE PAS AJOUTER DAUTRES MEDICAMENTS / NE
PAS RACCORDER EN SERIE / NE PAS UTILISER SIMULTANEMENT AVEC
DU SANG
PRECAUTIONS COMPRIMER ET EXAMINER LA POCHE / NE PAS
UTILISER SI LA SOLUTION N’EST PAS LIMPIDE / N’UTILISER QU’UNE
SEULE FOIS / JETER TOUTE PORTION INUTILISEE / CONSERVER
ENTRE 15ºC ET 25ºC / MONOGRAPHIE OFFERTE SUR DEMANDE /
PROFESSIONNEL DE LA SANTE DISTRIBUER AVEC LES
RENSEIGNEMENTS POUR LE CONSOMMATEUR
NONPYROGENIC / STERILE / APYROGENE
QUESTIONS OR CONCERNS/
QUESTIONS OU PREOCCUPATIONS
Baxter Logo
Baxter Corporation
Mississauga ON L5N 0C2 1-800-719-9955
VIAFLEX® PVC CONTAINER/
CONTENANT DE PVC
No Latex Symbol
07-25-77-873
JB0973
500 mL
DIN 00828602
Lidocaine Hydrochloride 0.4% and 5% Dextrose
Injection / Chlorhydrate de Lidocaine á 0.4%
dans du Dextrose á 5% Injectable
Lidocaine Hydrochloride and Dextrose Injection USP /
Chlorhydrate de Lidocaine dans du Dextrose USP,
Injectable
Lidocaine
4 mg/mL
2 g / 500 mL
Bar Code
(01)00809080000555
EACH mL CONTAINS / CHAQUE mL CONTIENT
LIDOCAINE HYDROCHLORIDE / CHLORHYDRATE DE LIDOCAINE 4 mg
DEXTROSE / DEXTROSE – 50 mg
SODIUM HYDROXIDE (PH ADJUSTER) / HYDROXYDE DE SODIUM
(AJUSTEUR DE PH)
WATER FOR INJECTION / EAU POUR INJECTION
APPROX mOsmol/L 282 APPROX pH 5.0
ANTIARRHYTHMIC AGENT FOR INTRAVENOUS INFUSION ONLY /
AGENT ANTIARYTHMIQUE POUR PERFUSION INTRAVEINEUSE SEULEMENT
USUAL DOSAGE SINGLE DOSE 50 mg – 100 mg AT A RATE OF 25 – 50 mg PER
MIN / DO NOT EXCEED 300 mg PER HOUR / CONTINUOUS INFUSION AT A RATE
OF 1 TO 2 mg PER MIN / NOT RECOMMENDED FOR USE IN CHILDREN
WARNINGS DO NOT ADD SUPPLEMENTARY MEDICATION / MUST NOT BE USED
IN SERIES CONNECTIONS / DO NOT USE SIMULTANEOUSLY WITH BLOOD
CAUTIONS SQUEEZE AND INSPECT BAG / DO NOT USE UNLESS
SOLUTION IS CLEAR / SINGLE USE / DISCARD UNUSED PORTION / STORE AT
15ºC TO 25ºC / PRODUCT MONOGRAPH AVAILABLE ON REQUEST /
HEALTHCARE PROFESSIONAL DISPENSE WITH CONSUMER INFORMATION
POSOLOGIE HABITUELLE DOSE UNIQUE DE 50 mg – 100 MG A UN DEBIT DE
25-50 mg PAR MIN / NE PAS DEPASSER 300 mg PAR HEURE / PERFUSION
CONTINUE A UN DEBIT DE 1 A 2 mg PAR MIN / N’EST PAS RECOMMANDE EN
PEDIATRIE
MISES EN GARDE NE PAS AJOUTER D’AUTRES MEDICAMENTS / NE PAS
RACCORDER EN SERIE / NE PAS UTILISER SIMULTANEMENT AVEC DU SANG
PRECAUTIONS COMPRIMER ET EXAMINER LA POCHE / NE PAS UTILISER SI LA
SOLUTION N’EST PAS LIMPIDE / N’UTILISER QU’UNE SEULE FOIS / JETER TOUTE
PORTION INUTILISEE / CONSERVER ENTRE 15ºC ET 25ºC / MONOGRAPHIE
OFFERTE SUR DEMANDE / PROFESSIONNEL DE LA SANTE DISTRIBUER AVEC
LES RENSEIGNEMENTS POUR LE CONSOMMATEUR
NONPYROGENIC / STERILE / APYROGENE
QUESTIONS OR CONCERNS/
QUESTIONS OU PREOCCUPATIONS
Baxter Logo
Baxter Corporation
Mississauga ON L5N 0C2 1-800-719-9955
VIAFLEX® PVC CONTAINER/
CONTENANT DE PVC
No Latex Symbol
07-25-77-872
| LIDOCAINE HYDROCHLORIDE AND DEXTROSE
lidocaine hydrochloride injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LIDOCAINE HYDROCHLORIDE AND DEXTROSE
lidocaine hydrochloride injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Corporation | 205087968 | ANALYSIS(0338-9590, 0338-9586) , LABEL(0338-9590, 0338-9586) , MANUFACTURE(0338-9590, 0338-9586) , PACK(0338-9590, 0338-9586) , STERILIZE(0338-9590, 0338-9586) | |