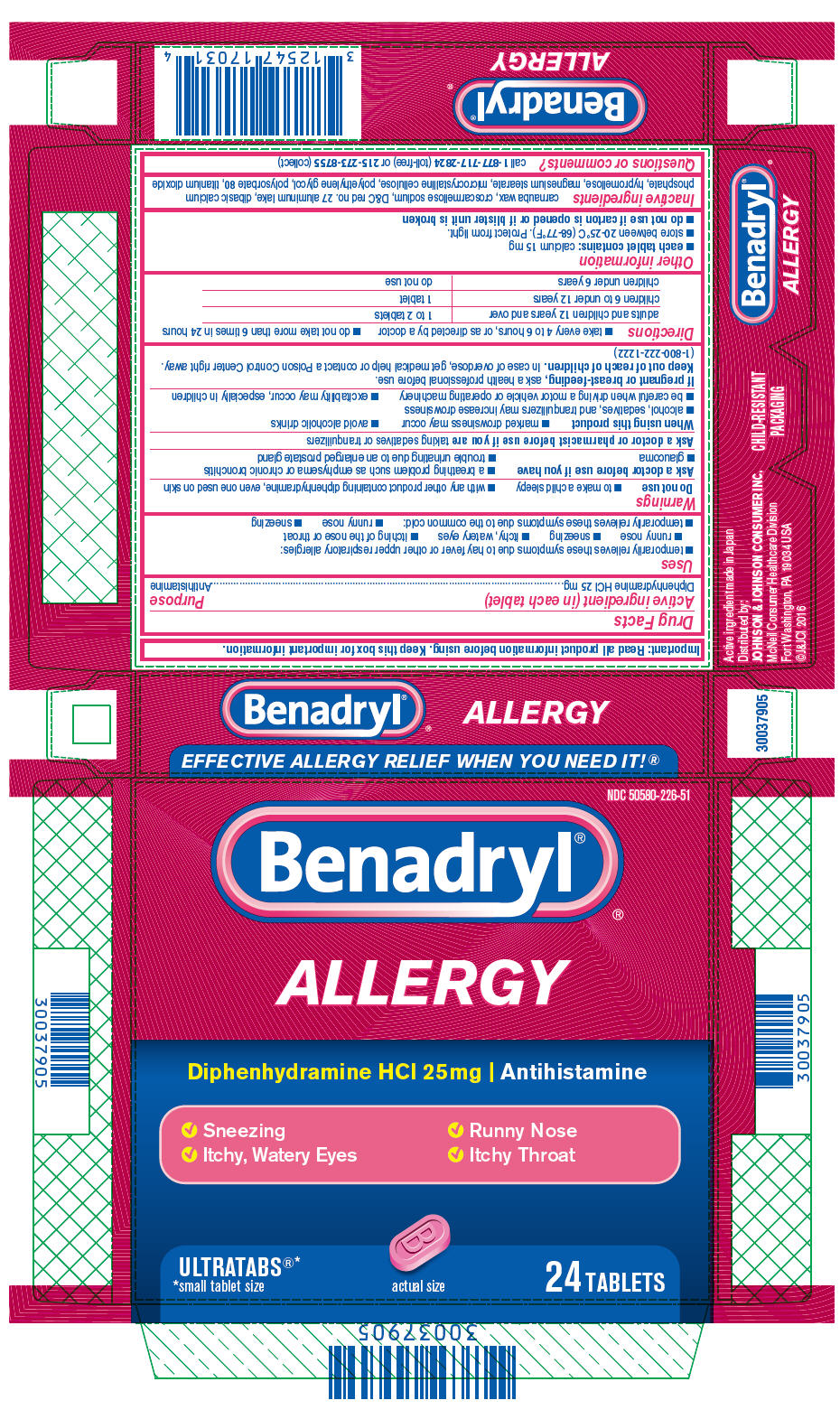
Active ingredient (in each tablet)
Diphenhydramine HCl 25 mg
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Directions
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use |
Other information
-
each tablet contains: calcium 15 mg
- store between 20-25°C (68-77°F). Protect from light.
-
do not use if carton is opened or if blister unit is broken
Inactive ingredients
carnauba wax, croscarmellose sodium, D&C red no. 27 aluminum lake, dibasic calcium phosphate, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, titanium dioxide
Questions or comments?
call
1-877-717-2824 (toll-free) or
215-273-8755 (collect)
PRINCIPAL DISPLAY PANEL
NDC 50580-226-51
Benadryl
®
ALLERGY
Diphenhydramine HCl 25mg | Antihistamine
✓ Sneezing
✓ Runny Nose
✓ Itchy, Watery Eyes
✓ Itchy Throat
ULTRATABS
®*
*small tablet size
actual size
24 TABLETS
Johnson & Johnson Consumer Inc.
