ANJESO- meloxicam injection
Baudax Bio, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ANJESO safely and effectively. See full prescribing information for ANJESO.
ANJESO® (meloxicam) injection, for intravenous use Initial U.S. Approval: 2000 WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTSSee full prescribing information for complete boxed warning.
RECENT MAJOR CHANGESINDICATIONS AND USAGEANJESO is an NSAID indicated for use in adults for the management of moderate-to-severe pain, alone or in combination with non-NSAID analgesics. Limitation of Use Because of delayed onset of analgesia, ANJESO alone is not recommended for use when rapid onset of analgesia is required. (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSANJESO (meloxicam) injection, single-dose vial containing 30 mg/mL per vial. (3) CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions (≥ 2% and greater than placebo) in controlled clinical trials include constipation, GGT increased, and anemia. (6) To report SUSPECTED ADVERSE REACTIONS, contact Baudax Bio at 1-844-303-5510 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION. Revised: 2/2022 |
FULL PRESCRIBING INFORMATION
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
Cardiovascular Risk
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use. [see Warnings and Precautions (5.1)].
- ANJESO is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4) and Warnings and Precautions (5.1)].
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events [see Warnings and Precautions (5.2)].
1 INDICATIONS AND USAGE
ANJESO is indicated for use in adults for the management of moderate-to-severe pain, alone or in combination with non-NSAID analgesics.
Limitation of Use
Because of delayed onset of analgesia, ANJESO alone is not recommended for use when rapid onset of analgesia is required.
2 DOSAGE AND ADMINISTRATION
Use for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5)].
For intravenous administration only.
The recommended dose of ANJESO is 30 mg once daily, administered by intravenous bolus injection over 15 seconds.
When initiating ANJESO, monitor patient analgesic response. Because the median time to meaningful pain relief was 2 and 3 hours after ANJESO administration in two clinical studies, a non-NSAID analgesic with a rapid onset of effect may be needed, for example, upon anesthetic emergence or resolution of local or regional anesthetic blocks [see Clinical Studies (14)].
Some patients may not experience adequate analgesia for the entire 24-hour dosing interval and may require administration of a short-acting, non-NSAID, immediate-release analgesic [see Clinical Studies (14)].
To reduce the risk of renal toxicity, patients must be well hydrated prior to administration of ANJESO.
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration. Should the contents appear discolored or contain particulate matter, discard the vial [see Dosage Forms and Strengths (3)]. The vial contains overfill to allow for withdrawal of 1 mL of the drug product. The overfill is to provide assurance that 1 mL can be withdrawn from the vial to deliver 30 mg of meloxicam. Discard any unused portion from the single-dose vial [see Dosage Forms and Strengths (3)].
3 DOSAGE FORMS AND STRENGTHS
ANJESO (meloxicam) injection is a sterile, opaque, pale yellow, non-pyrogenic, aqueous dispersion intended for intravenous use available as a clear, 2 mL, single-dose vial containing 30 mg/mL per vial.
Each vial is filled with a target 1.3mg (1.25mL) of drug product. The fill overage is to provide assurance that 1mL can be withdrawn from the vial to deliver 30 mg of drug product for dosing.
4 CONTRAINDICATIONS
ANJESO is contraindicated in the following patients:
- Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to meloxicam or any components of the drug product [see Warnings and Precautions (5.7)].
- History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal anaphylactic-like reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.8)].
- In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)].
- Moderate to severe renal insufficiency patients who are at risk for renal failure due to volume depletion [see Warnings and Precautions (5.6)].
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as meloxicam, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)].
Status Post Coronary Artery Bypass Graft (CABG) Surgery
Two large, controlled clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10-14 days following CABG surgery found an increased incidence of myocardial infarction and stroke. NSAIDs are contraindicated in the setting of CABG [see Contraindications (4)].
Post-MI Patients
Observational studies conducted in the Danish National Registry have demonstrated that patients treated with NSAIDs in the post-MI period were at increased risk of reinfarction, CV-related death, and all-cause mortality beginning in the first week of treatment. In this same cohort, the incidence of death in the first year post-MI was 20 per 100 person years in NSAID-treated patients compared to 12 per 100 person years in non-NSAID exposed patients. Although the absolute rate of death declined somewhat after the first year post-MI, the increased relative risk of death in NSAID users persisted over at least the next four years of follow-up.
Avoid the use of ANJESO in patients with a recent MI unless the benefits are expected to outweigh the risk of recurrent CV thrombotic events. If ANJESO is used in patients with a recent MI, monitor patients for signs of cardiac ischemia.
5.2 Gastrointestinal Effects- Risk of Ulceration, Bleeding, and Perforation
NSAIDs, including meloxicam, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with ANJESO. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3-6 months, and in about 2-4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
Risk Factors for GI Bleeding, Ulceration, and Perforation
Patients with a prior history of peptic ulcer disease and/or GI bleeding who used NSAIDs had a greater than 10-fold increased risk for developing a GI bleed compared to patients without these risk factors. Other factors that increase the risk of GI bleeding in patients treated with NSAIDs include longer duration of NSAID therapy; concomitant use of oral corticosteroids, aspirin, anticoagulants, or selective serotonin reuptake inhibitors (SSRIs); smoking; use of alcohol; older age; and poor general health status. Most postmarketing reports of fatal GI events occurred in elderly or debilitated patients. Additionally, patients with advanced liver disease and/or coagulopathy are at increased risk for GI bleeding.
Strategies to Minimize the GI Risks in NSAID-treated patients:
- Use the lowest effective dosage for the shortest possible duration.
- Avoid administration of more than one NSAID at a time.
- Avoid use in patients at higher risk unless benefits are expected to outweigh the increased risk of bleeding. For such patients, as well as those with active GI bleeding, consider alternate therapies other than NSAIDs.
- Remain alert for signs and symptoms of GI ulceration and bleeding during NSAID therapy.
- If a serious GI adverse event is suspected, promptly initiate evaluation and treatment, and discontinue ANJESO until a serious GI adverse event is ruled out.
- In the setting of concomitant use of low-dose aspirin for cardiac prophylaxis, monitor patients more closely for evidence of GI bleeding [see Drug Interactions (7)].
5.3 Hepatotoxicity
Elevations of ALT or AST (three or more times the upper limit of normal [ULN]) have been reported in approximately 1% of NSAID-treated patients in clinical trials. In addition, rare, sometimes fatal, cases of severe hepatic injury, including fulminant hepatitis, liver necrosis, and hepatic failure have been reported.
Elevations of ALT or AST (less than three times ULN) may occur in up to 15% of patients treated with NSAIDs including meloxicam.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue ANJESO immediately, and perform a clinical evaluation of the patient [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.4 Hypertension
NSAIDs, including ANJESO, can lead to new onset of hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists’ Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of meloxicam may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)].
Avoid the use of ANJESO in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If ANJESO is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
5.6 Renal Toxicity and Hyperkalemia
Renal Toxicity
Long-term administration of NSAIDs has resulted in renal papillary necrosis, renal insufficiency, acute renal failure, and other renal injury.
Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of an NSAID may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, dehydration, hypovolemia, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors or ARBs, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
ANJESO is not recommended in patients with moderate to severe renal insufficiency and is contraindicated in patients with moderate to severe renal insufficiency who are at risk for renal failure due to volume depletion.
No information is available from controlled clinical studies regarding the use of ANJESO in patients with advanced renal disease. The renal effects of ANJESO may hasten the progression of renal dysfunction in patients with pre-existing renal disease.
Correct volume status in dehydrated or hypovolemic patients prior to initiating ANJESO. Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia during use of ANJESO [see Drug Interactions (7)]. Avoid the use of ANJESO in patients with advanced renal disease unless the benefits are expected to outweigh the risk of worsening renal function. If ANJESO is used in patients with advanced renal disease, monitor patients for signs of worsening renal function [see Clinical Pharmacology (12.3)].
Hyperkalemia
Increases in serum potassium concentration, including hyperkalemia, have been reported with use of NSAIDs, even in some patients without renal impairment. In patients with normal renal function, these effects have been attributed to a hyporeninemic-hypoaldosteronism state.
5.7 Anaphylactic Reactions
Meloxicam has been associated with anaphylactic reactions in patients with and without known hypersensitivity to meloxicam and in patients with aspirin-sensitive asthma [see Contraindications (4) and Warnings and Precautions (5.8)].
Seek emergency help if an anaphylactic reaction occurs.
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, ANJESO is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)]. When ANJESO is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.9 Serious Skin Reactions
NSAIDs, including ANJESO, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity. ANJESO is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)].
5.10 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as ANJESO. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue ANJESO and evaluate the patient immediately.
5.11 Fetal Toxicity
Premature Closure of Fetal Ductus Arteriosus
Avoid use of NSAIDs, including ANJESO, in pregnant women at about 30 weeks gestation and later. NSAIDs, including ANJESO, increase the risk of premature closure of the fetal ductus arteriosus at approximately this gestational age.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs, including ANJESO, at about 20 weeks gestation or later in pregnancy may cause fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. Oligohydramnios is often, but not always, reversible with treatment discontinuation. Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some postmarketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If NSAID treatment is necessary between about 20 weeks and 30 weeks gestation, limit ANJESO use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if ANJESO treatment extends beyond 48 hours. Discontinue ANJESO if oligohydramnios occurs and follow up according to clinical practice [see Use in Specific Populations (8.1)]
5.12 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with ANJESO has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including ANJESO, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5.13 Masking of Inflammation and Fever
The pharmacological activity of ANJESO in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6)]. ANJESO is not indicated for long-term treatment.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in the other sections of the labeling:
- Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1)]
- GI Bleeding, Ulceration, and Perforation [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Hypertension [see Warnings and Precautions (5.4)]
- Heart Failure and Edema [see Warnings and Precautions (5.5)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6)]
- Anaphylactic Reactions [see Warnings and Precautions (5.7)]
- Serious Skin Reactions [see Warnings and Precautions (5.9)]
- Hematologic Toxicity [see Warnings and Precautions (5.12)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During clinical development, 1426 patients were exposed to ANJESO in controlled and open-label Phase 2 and Phase 3 trials. ANJESO was studied across a range of surgical procedures, including bunionectomy, abdominoplasty, soft tissue surgery, total knee replacement surgery, gynecologic surgery, complex foot surgery and total hip replacement surgery. In these trials, 381 patients received a single dose of ANJESO and 1045 patients received multiple doses of ANJESO daily for up to 7 days. The incidence rates of adverse reactions listed in Table 1 are derived from the three Phase 3 trials comparing ANJESO to placebo in patients who may have also received opioid rescue medication.
| ANJESO | Placebo |
|
|---|---|---|
| Adverse Reaction | N=748 | N=393 |
| Constipation | 57 (7.6%) | 24 (6.1%) |
| Gamma-Glutamyl transferase Increased | 21 (2.8%) | 6 (1.5%) |
| Anemia | 18 (2.4%) | 4 (1.0%) |
The following is a list of adverse drug reactions occurring in <2% of patients receiving ANJESO in clinical trials.
| Body as a Whole | asthenia, back pain, edema, fatigue, hyperthermia, infusion site reactions (including pain, pruritus, phlebitis and thrombosis), muscle spasms, non-cardiac chest pain, pyrexia, vaginal discharge, weight decrease |
| Central and Peripheral Nervous System | disturbance in attention, migraine, presyncope, somnolence, syncope |
| Gastrointestinal | abdominal discomfort, abdominal distension, abdominal pain, diarrhea, dry mouth, epigastric discomfort, flatulence, frequent bowel movements, gastritis, gastroesophageal reflux, gastrointestinal pain, rectal hemorrhage |
| Heart Rate and Rhythm | tachycardia |
| Hematologic | increased bleeding time, neutropenia, thrombocytosis |
| Infections and Infestations | cellulitis, gastroenteritis, urinary tract infection, vulval abscess |
| Liver and Biliary System | liver function test abnormal |
| Metabolic and Nutritional | hypokalemia, hypomagnesemia |
| Procedural Complications | incision site hemorrhage, incision site rash, wound dehiscence, wound hematoma |
| Psychiatric | confusion, hallucination, insomnia |
| Respiratory | dyspnea, epistaxis, hypoxia, oropharyngeal pain |
| Skin and Appendages | contact dermatitis, ecchymosis, rash |
| Urinary System | pollakiuria, urinary retention |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of meloxicam. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions reported in worldwide post marketing experience or the literature include: acute urinary retention; agranulocytosis; alterations in mood (such as mood elevation); anaphylactoid reactions including shock; erythema multiforme; exfoliative dermatitis; interstitial nephritis; jaundice; liver failure; Stevens-Johnson syndrome; toxic epidermal necrolysis, and infertility female.
7 DRUG INTERACTIONS
See Table 3 for clinically significant drug interactions with meloxicam.
| Drugs That Interfere with Hemostasis | |
| Clinical Impact: |
|
| Intervention: | Monitor patients with concomitant use of ANJESO with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.12)]. |
| Aspirin | |
| Clinical Impact: | Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)]. |
| Intervention: | Concomitant use of ANJESO and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12)].
In the setting of concomitant use of low-dose aspirin for cardiac prophylaxis, monitor patients more closely for evidence of GI bleeding [see Warnings and Precautions (5.2)]. ANJESO is not a substitute for low dose aspirin for cardiovascular protection. |
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single and multiple dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam |
| Intervention: | During concomitant use of ANJESO with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6)]. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. [see Clinical Pharmacology (12.3)]. |
| Intervention: | During concomitant use of ANJESO and lithium, monitor patients for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of ANJESO and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of ANJESO and cyclosporine may increase cyclosporine’s nephrotoxicity. |
| Intervention: | During concomitant use of ANJESO and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of meloxicam with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2)]. |
| Intervention: | The concomitant use of meloxicam with other NSAIDs or salicylates is not recommended. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of ANJESO and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of ANJESO and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity.
Patients taking meloxicam should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. In patients with creatinine clearance below 45 mL/min, the concomitant administration of meloxicam with pemetrexed is not recommended. |
| CYP2C9 Inhibitors | |
| Clinical Impact: | In vitro studies indicate that CYP2C9 (cytochrome P450 metabolizing enzyme) plays an important role in this metabolic pathway with a minor contribution of the CYP3A4 isozyme. Thus concomitant usage of CYP2C9 inhibitors (such as amiodarone, fluconazole, and sulphaphenazole) may lead to abnormally high plasma levels of meloxicam due to reduced metabolic clearance [see Use in Specific Populations (8.8); and Clinical Pharmacology (12.3, 12.5)]. |
| Intervention: | Consider dose reduction in patients undergoing treatment with CYP2C9 inhibitors, and monitor patients for adverse effects. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Use of NSAIDs, including ANJESO, of can cause premature closure of the fetal ductus arteriosus and fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. Because of these risks, limit dose and duration of ANJESO use between about 20 and 30 weeks of gestation, and avoid ANJESO use at about 30 weeks of gestation and later in pregnancy (see Clinical Considerations, Data).
Premature Closure of Fetal Ductus Arteriosus
Use of NSAIDs, including ANJESO, at about 30 weeks gestation or later in pregnancy increases the risk of premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs at about 20 weeks gestation or later in pregnancy has been associated with cases of fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment.
Data from observational studies regarding potential embryofetal risks of NSAID use in women in the first or second trimesters of pregnancy are inconclusive.
In animal reproduction studies, embryofetal death was observed in rats and rabbits treated during the period of organogenesis with meloxicam at oral doses equivalent to 0.32- and 3.24-times the maximum recommended human dose (MRHD) of 30 mg of ANJESO, based on body surface area (BSA). Increased incidence of septal heart defects were observed in rabbits treated throughout embryogenesis with meloxicam at an oral dose equivalent to 39-times the MRHD of 30 mg of ANJESO. In pre- and post-natal reproduction studies, there was an increased incidence of dystocia, delayed parturition, and decreased offspring survival at 0.04-times the MRHD of 30 mg of ANJESO. No teratogenic effects were observed in rats and rabbits treated with meloxicam during organogenesis at an oral dose equivalent to 1.3 and 13-times the MRHD of 30 mg of ANJESO [see Data].
Based on animal data, prostaglandins have been shown to have an important role in endometrial vascular permeability, blastocyst implantation, and decidualization. In animal studies, administration of prostaglandin synthesis inhibitors, such as meloxicam, resulted in increased pre- and post-implantation loss. Prostaglandins also have been shown to have an important role in fetal kidney development. In published animal studies, prostaglandin synthesis inhibitors have been reported to impair kidney development when administered at clinically relevant doses.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy, because NSAIDs, including ANJESO, can cause premature closure of the fetal ductus arteriosus [see Data].
Oligohydramnios/Neonatal Renal Impairment:
If an NSAID is necessary at about 20 weeks gestation or later in pregnancy, limit the use to the lowest effective dose and shortest duration possible. If ANJESO treatment extends beyond 48 hours, consider monitoring with ultrasound for oligohydramnios. If oligohydramnios occurs, discontinue ANJESO and follow up according to clinical practice [see Data].
Labor or delivery
There are no studies on the effects of meloxicam during labor or delivery. In animal studies, NSAIDs, including meloxicam, inhibit prostaglandin synthesis, cause delayed parturition, and increase the incidence of stillbirth.
Human Data
Premature Closure of Fetal Ductus Arteriosus:
Published literature reports that the use of NSAIDs at about 30 weeks of gestation and later in pregnancy may cause premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment:
Published studies and postmarketing reports describe maternal NSAID use at about 20 weeks gestation or later in pregnancy associated with fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. In many cases, but not all, the decrease in amniotic fluid was transient and reversible with cessation of the drug. There have been a limited number of case reports of maternal NSAID use and neonatal renal dysfunction without oligohydramnios, some of which were irreversible. Some cases of neonatal renal dysfunction required treatment with invasive procedures, such as exchange transfusion or dialysis.
Methodological limitations of these postmarketing studies and reports include lack of a control group; limited information regarding dose, duration, and timing of drug exposure; and concomitant use of other medications. These limitations preclude establishing a reliable estimate of the risk of adverse fetal and neonatal outcomes with maternal NSAID use. Because the published safety data on neonatal outcomes involved mostly preterm infants, the generalizability of certain reported risks to the full-term infant exposed to NSAIDs through maternal use is uncertain.
Animal Data
Meloxicam was not teratogenic when administered to pregnant rats during fetal organogenesis at oral doses up to 4 mg/kg/day (1.3-fold greater than the MRHD of 30 mg of ANJESO based on BSA comparison). Administration of meloxicam to pregnant rabbits throughout embryogenesis produced an increased incidence of septal defects of the heart at an oral dose of 60 mg/kg/day (39-fold greater than the MRHD of 30 mg of ANJESO based on BSA comparison). The no effect level was 20 mg/kg/day (13-fold greater than the MRHD of 30 mg of ANJESO based on BSA conversion). In rats and rabbits, embryolethality occurred at oral meloxicam doses of 1 mg/kg/day and 5 mg/kg/day, respectively (0.32 and 3.24-fold greater, respectively, than the MRHD of 30 mg of ANJESO based on BSA comparison) when administered throughout organogenesis.
Oral administration of meloxicam to pregnant rats during late gestation through lactation increased the incidence of dystocia, delayed parturition, and decreased offspring survival at meloxicam doses of 0.125 mg/kg/day or greater (0.04-times the MRHD of 30 mg of ANJESO based on BSA comparison).
8.2 Lactation
Risk Summary
There are no human data available on whether meloxicam is present in human milk, or on the effects on breastfed infants, or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ANJESO and any potential adverse effects on the breastfed infant from the ANJESO or from the underlying maternal condition.
Data
Animal Data
Meloxicam was present in the milk of lactating rats at concentrations higher than those in plasma.
8.3 Females and Males of Reproductive Potential
Infertility
Females
Based on the mechanism of action, the use of prostaglandin-mediated NSAIDs, including ANJESO, may delay or prevent rupture of ovarian follicles, which has been associated with reversible infertility in some women. Published animal studies have shown that administration of prostaglandin synthesis inhibitors has the potential to disrupt prostaglandin-mediated follicular rupture required for ovulation. Small studies in women treated with NSAIDs have also shown a reversible delay in ovulation. Consider withdrawal of NSAIDs, including ANJESO, in women who have difficulties conceiving or who are undergoing investigation of infertility.
Males
ANJESO may compromise fertility in males of reproductive potential. In a published study, oral administration of meloxicam to male rats for 35 days resulted in decreased sperm count and motility and histopathological evidence of testicular degeneration at 0.3-times the MRHD based on BSA comparison [see Nonclinical Toxicology (13.1)]. It is not known if these effects on fertility are reversible. The clinical relevance of these findings is unknown.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.14)].
Of the total number of patients in clinical studies of ANJESO, 197 patients were age 65 and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients.
8.6 Hepatic Impairment
ANJESO has not been studied in patients with hepatic impairment. For oral meloxicam, no dose adjustment is necessary in patients with mild to moderate hepatic impairment. Since meloxicam is significantly metabolized in the liver and hepatotoxicity may occur, monitor for adverse events in patients with severe hepatic impairment [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Pharmacokinetics of ANJESO in elderly subjects with mild renal impairment is similar to healthy young subjects. Patients with moderate or severe renal impairment have not been studied. ANJESO is not recommended in patients with moderate to severe renal insufficiency. ANJESO is contraindicated in patients with moderate to severe renal insufficiency who are at risk for renal failure due to volume depletion [see Contraindications (4), Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
8.8 Poor Metabolizers of CYP2C9 Substrates
In patients who are known or suspected to be poor CYP2C9 metabolizers based on genotype or previous history/experience with other CYP2C9 substrates (such as warfarin and phenytoin) consider dose reduction as they may have abnormally high plasma levels due to reduced metabolic clearance, and monitor patients for adverse effects.
10 OVERDOSAGE
Symptoms following acute NSAID overdoses have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression and coma have occurred, but were rare. [see Warnings and Precautions (5.1, 5.2, 5.4, 5.6)].
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may be employed but are not likely to be useful due to high protein binding.
There is limited experience with meloxicam overdose. In four reported cases of meloxicam overdose, patients took 6 to 11 times the highest available oral dose of meloxicam tablets (15 mg); all recovered. Cholestyramine is known to accelerate the clearance of meloxicam. Accelerated removal of meloxicam by 4 g doses of cholestyramine given three times a day was demonstrated in a clinical trial. Administration of cholestyramine may be useful following an overdosage.
In case of an overdosage, discontinue ANJESO therapy and contact a regional poison control center at 1-800-222-1222.
11 DESCRIPTION
ANJESO (meloxicam) is a nonsteroidal anti-inflammatory drug (NSAID) of the enolic acid class. It is a sterile opaque, pale-yellow, aqueous dispersion containing the active pharmaceutical ingredient meloxicam for intravenous administration. Each mL of aqueous dispersion contains 30 mg of meloxicam. The inactive ingredients of ANJESO include: 9 mg povidone, 3 mg sodium deoxycholate (deoxycholic acid), 60 mg sucrose, and water for injection.
Meloxicam is practically insoluble in water, slightly soluble in acetone, soluble in dimethylformamide and very slightly soluble in 96% ethanol and in methanol. Meloxicam is designated chemically as 4-hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide. The molecular weight is 351.4. Its molecular formula is C14H13N3O4S2 and the structural formula of meloxicam is:
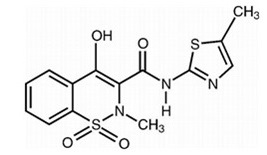
Figure 1: Structural Formula of Meloxicam
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Meloxicam has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of meloxicam, like that of other NSAIDs, is not completely understood but involves inhibition of cyclooxygenase (COX-1 and COX-2).
Meloxicam is a potent inhibitor of prostaglandin synthesis in vitro. Meloxicam concentrations reached during therapy have produced in vivo effects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Because meloxicam is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of ANJESO on QTc prolongation was evaluated in a randomized, double-blind, placebo-controlled crossover study in healthy subjects. A total of 48 subjects were administered intravenous meloxicam 30 mg, 120 mg, and 180 mg. In a study with demonstrated ability to detect small effects, the upper bound of the 90% confidence interval for the largest placebo-adjusted, baseline- corrected QTc based on Fridericia correction method (QTcF) was below 10 ms, the threshold for regulatory concern.
12.3 Pharmacokinetics
Absorption
Following intravenous administration of ANJESO to healthy volunteers, plasma concentrations of 30 mg ANJESO exceed that of 15 mg oral meloxicam for the first 24 hours. The single-dose pharmacokinetics of meloxicam following intravenous administration of ANJESO and oral doses of meloxicam are compared in Table 4.
| ANJESO | Oral Meloxicam | |
|---|---|---|
| 30 mg IV | 15 mg PO | |
| Parameter | ||
| Cmax (ng/mL) | 5642.9±1009.0 | 1221.9±289.5 |
| Tmax (h) | 0.12±0.04 | 6.57 ±4.12 |
| AUCinf (ng*hr/mL) | 107508.7±34443.0 | 53988.8±23207.7 |
| T½ (h) | 23.3±9.36 | 26.4 ±12.1 |
Following multiple dosing, ANJESO exhibited predictable accumulation (slightly higher than 2-fold) without a change in the terminal elimination half-life (Table 5).
| ANJESO | ANJESO | |
|---|---|---|
| 30 mg | 30 mg | |
| IV Single Dose | IV Repeat Dose | |
| Parameter | ||
| Cmax (ng/mL) | 7972.5±2579.9 | 10632.5±4729.8 |
| AUCinf (ng.hr/mL) | 121437.6±64505.6 | 297771.6±241604.01 |
| T½ (h) | 23.6±10.1 | 26.4±10.1 |
ANJESO exhibits linear pharmacokinetics over intravenous doses ranging from 15 mg to 180 mg.
Distribution
Following administration of ANJESO, the apparent volume of distribution during the terminal elimination phase (Vz) of meloxicam is 9.63 L.
Meloxicam is ~99.4% bound to human plasma proteins (primarily albumin) within the therapeutic dose range. The fraction of protein binding is independent of drug concentration, over the clinically relevant concentration range, but decreases to ~99% in patients with renal disease. Meloxicam penetration into human red blood cells, after oral dosing, is less than 10%. Following a radiolabeled dose, over 90% of the radioactivity detected in the plasma was present as unchanged meloxicam.
Meloxicam concentrations in synovial fluid, after a single oral dose, range from 40% to 50% of those in plasma. The free fraction in synovial fluid is 2.5 times higher than in plasma, due to the lower albumin content in synovial fluid as compared to plasma. The significance of this penetration is unknown.
Elimination
Metabolism
Meloxicam is extensively metabolized in the liver. Meloxicam metabolites include 5'-carboxy meloxicam (60% of dose), from P-450 mediated metabolism formed by oxidation of an intermediate metabolite 5'-hydroxymethyl meloxicam which is also excreted to a lesser extent (9% of dose). In vitro studies indicate that CYP2C9 (cytochrome P450 metabolizing enzyme) plays an important role in this metabolic pathway with a minor contribution of the CYP3A4 isozyme. Patients’ peroxidase activity is probably responsible for the other two metabolites which account for 16% and 4% of the administered dose, respectively. The four metabolites are not known to have any in vivo pharmacological activity.
Excretion
Meloxicam excretion is predominantly in the form of metabolites, and occurs to equal extents in the urine and feces. Only traces of the unchanged parent compound are excreted in the urine (0.2%) and feces (1.6%). The extent of the urinary excretion was confirmed for unlabeled multiple 7.5 mg doses: 0.5%, 6%, and 13% of the dose were found in urine in the form of meloxicam, and the 5'-hydroxymethyl and 5'-carboxy metabolites, respectively. There is significant biliary and/or enteral secretion of the drug. This was demonstrated when oral administration of cholestyramine following a single IV dose of meloxicam decreased the AUC of meloxicam by 50%.
The mean elimination half-life (t1/2) for ANJESO 30 mg is approximately 24 hours.
Specific Populations
Based on population pharmacokinetics analyses, age, sex and race do not have a clinically meaningful effect on the PK of ANJESO.
Hepatic Impairment: The pharmacokinetics of ANJESO have not been investigated in patients with hepatic impairment. However, following a single 15 mg dose of meloxicam tablets there was no marked difference in plasma concentrations in patients with mild (Child-Pugh Class I) or moderate (Child-Pugh Class II) hepatic impairment compared to healthy volunteers. Protein binding of meloxicam was not affected by hepatic impairment. No dosage adjustment is necessary in patients with mild to moderate hepatic impairment. Patients with severe hepatic impairment (Child-Pugh Class III) have not been adequately studied. Monitor for adverse events when used in patients with severe hepatic impairment. [see Warnings and Precautions (5.3), Use in Specific Populations (8.6)].
Renal Impairment: ANJESO pharmacokinetics have been investigated in elderly subjects with mild renal impairment (eGFR 60 – 90) compared to young healthy volunteers. A 5% and 7% increase of Cmax and AUC, respectively, was observed in elderly subjects with mild renal impairment. No dosage adjustment is necessary in patients with mild renal impairment. Patients with moderate and severe renal impairment have not been adequately studied. The use of ANJESO in patients with moderate to severe renal impairment is not recommended. ANJESO is contraindicated in patients with moderate to severe renal insufficiency who are at risk for renal failure due to volume depletion [see Contraindications (4), Warnings and Precautions (5.6), Use in Specific Populations (8.7)].
Drug Interaction Studies
Aspirin: When NSAIDs were administered with aspirin, the protein binding of NSAIDs were reduced, although the clearance of free NSAID was not altered. The clinical significance of this interaction is not known. See Table 3 clinically significant drug interactions of NSAIDs with aspirin [see Drug Interactions (7)].
Cholestyramine: Pretreatment for four days with cholestyramine significantly increased the clearance of meloxicam by 50%. This resulted in a decrease in t1/2, from 19.2 hours to 12.5 hours, and a 35% reduction in AUC. This suggests the existence of a recirculation pathway for meloxicam in the gastrointestinal tract. The clinical relevance of this interaction has not been established.
Cimetidine: Concomitant administration of 200 mg cimetidine four times daily did not alter the single-dose pharmacokinetics of 30 mg meloxicam.
Digoxin: Meloxicam tablets 15 mg once daily for 7 days did not alter the plasma concentration profile of digoxin after β-acetyldigoxin administration for 7 days at clinical doses. In vitro testing found no protein binding drug interaction between digoxin and meloxicam [see Drug Interactions (7)].
Lithium: In a study conducted in healthy subjects, mean pre-dose lithium concentration and AUC were increased by 21% in subjects receiving lithium doses ranging from 804 to 1072 mg twice daily with meloxicam tablets 15 mg once per day every day as compared to subjects receiving lithium alone [see Drug Interactions (7)].
Methotrexate: A study in 13 rheumatoid arthritis (RA) patients evaluated the effects of multiple oral doses of meloxicam on the pharmacokinetics of methotrexate taken once weekly. Meloxicam did not have a significant effect on the pharmacokinetics of single doses of methotrexate. In vitro, methotrexate did not displace meloxicam from its human serum binding sites [see Drug Interactions (7)].
Warfarin: The effect of meloxicam tablets on the anticoagulant effect of warfarin was studied in a group of healthy subjects receiving daily doses of warfarin that produced an INR (International Normalized Ratio) between 1.2 and 1.8. In these subjects, meloxicam did not alter warfarin pharmacokinetics and the average anticoagulant effect of warfarin as determined by prothrombin time. However, one subject showed an increase in INR from 1.5 to 2.1. Caution should be used when administering ANJESO with warfarin since patients on warfarin may experience changes in INR and an increased risk of bleeding complications when a new medication is introduced [see Drug Interactions (7)].
12.5 Pharmacogenomics
CYP2C9 activity is reduced in individuals with genetic variants such as CYP2C9*2 and CYP2C9*3 polymorphisms. Limited data from three published reports showed that meloxicam AUC was substantially higher in individuals with reduced CYP2C9 activity, particularly in poor metabolizers (e.g., *3/*3), compared to normal metabolizers (*1/*1). The frequency of CYP2C9 poor metabolizer genotypes varies based on racial/ethnic background but is generally present in <5% of the population.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis There was no increase in tumor incidence in long-term carcinogenicity studies in rats (104 weeks) and mice (99 weeks) administered meloxicam at oral doses up to 0.8 mg/kg/day in rats and up to 8.0 mg/kg/day in mice (up to 0.26 and 1.3 times, respectively, the MRHD of 30 mg/day of ANJESO based on BSA comparison).
Mutagenesis ANJESO was not mutagenic in an Ames assay. Meloxicam was not clastogenic in a chromosome aberration assay with human lymphocytes and an in vivo micronucleus test in mouse bone marrow.
Impairment of Fertility Meloxicam did not impair male and female fertility in rats at oral doses up to 9 mg/kg/day in males and 5 mg/kg/day in females (up to 2.9- and 1.6-times greater, respectively, than the MRHD of 30 mg of ANJESO based on BSA comparison).
In a published study, oral administration of 1 mg/kg (0.3-times the MRHD based on BSA comparison) meloxicam to male rats for 35 days resulted in decreased sperm count and motility and histopathological evidence of testicular degeneration. The clinical relevance of these findings is unknown.
14 CLINICAL STUDIES
The efficacy and safety of ANJESO in the treatment of moderate to severe pain was evaluated in two Phase 3 randomized, double-blind, placebo-controlled, multiple-dose clinical trials in patients with postoperative pain. In both trials, oral oxycodone 5 mg was permitted as rescue medication for pain management.
Study 1 (Bunionectomy Surgery)
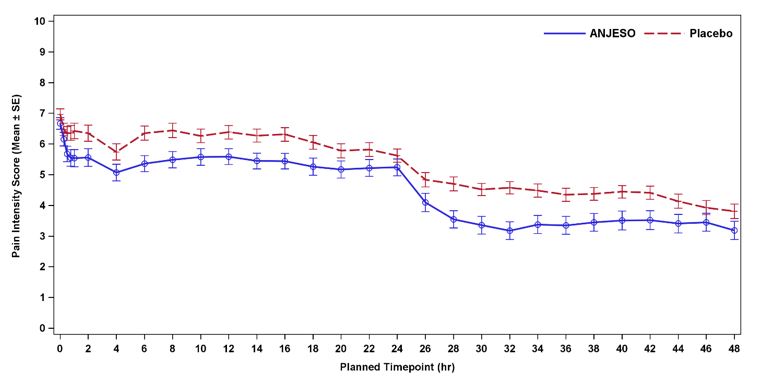
In the first controlled, multiple-dose trial (NCT02675907) of adult patients with postoperative pain who underwent bunionectomy surgery, 201 patients were treated with ANJESO 30 mg or placebo administered once daily for two days starting on the day after surgery. An optional third dose was permitted just prior to discharge. A minimum postoperative baseline pain intensity of 4 on the Numeric Pain Rating Scale (NPRS) (range 0-10) and pain categorized as moderate or severe were required for randomization. The majority of patients were female (85%). The average age was 48 years. The mean overall baseline pain intensity on the NPRS was 6.8. A statistically significant difference demonstrating efficacy was observed in the primary efficacy endpoint of the summed pain intensity difference over the first 48 hours (SPID48). The average pain intensity over time is depicted for the treatment groups in Figure 2. A generally consistent separation in pain scores between the ANJESO and placebo groups was observed from time of onset through most of the dosing interval with a narrowing at the end of the first 24-hour dosing interval.
Study 2 (Abdominoplasty Surgery)
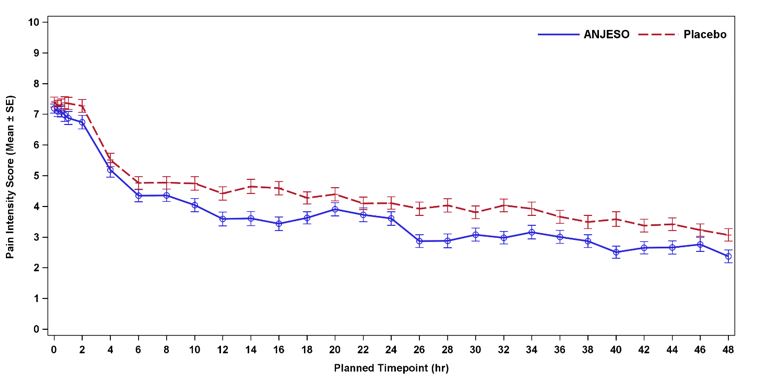
In the second controlled, multiple-dose trial (NCT02678286) of adult patients with postoperative pain who underwent elective abdominoplasty surgery, 219 patients were treated with ANJESO 30 mg or placebo administered once daily for two days starting on the day of surgery. An optional third dose was permitted just prior to discharge. A minimum postoperative baseline pain intensity of 4 on the NPRS (range 0-10) and pain categorized as moderate or severe were required for randomization. The majority of patients were female (98%). The average age was 40 years. The mean overall baseline pain intensity on the NPRS was 7.3. A statistically significant difference demonstrating efficacy was observed in the primary efficacy endpoint of the summed pain intensity difference over the first 24 hours (SPID24) as well as over the first 48 hours (SPID48). The average pain intensity over time is depicted for the treatment groups in Figure 3. A generally consistent separation in pain scores between the ANJESO and placebo group was observed from time of onset through most of the dosing interval with a narrowing at the end of the first 24-hour dosing interval.
Onset of Meaningful Pain Relief and Use of Rescue Analgesic Medication
The median time to first rescue analgesic use in patients treated with ANJESO (2 hours in Study 1 and 1 hour in Study 2) came before the median time to patient-reported meaningful pain relief in both studies (2 hours in Study 1 and 3 hours in Study 2). Fifty percent of patients treated with ANJESO and 49% of patients treated with placebo in Study 1 received rescue analgesia medication in the first 2 hours after the start of dosing. Seventy-eight percent of patients treated with ANJESO and 78% of patients treated with placebo in Study 2 received rescue in the first 3 hours after the start of dosing.
16 HOW SUPPLIED/STORAGE AND HANDLING
ANJESO (meloxicam) injection, is an opaque, pale-yellow aqueous dispersion intended for intravenous use supplied as a 1 mL fill (30 mg/mL) in a clear 2 mL single-dose vial with a blue tamper-evident top.
Single-dose vial: NDC 71518-001-01
17 PATIENT COUNSELING INFORMATION
Inform patients of the following information before initiating therapy with ANJESO.
Cardiovascular Thrombotic Effects
Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their healthcare provider immediately[see Warnings and Precautions (5.1)].
Gastrointestinal Bleeding, Ulceration, and Perforation
Advise patients to report symptoms of ulcerations and bleeding, including epigastric pain, dyspepsia, melena, and hematemesis to their healthcare provider. In the setting of concomitant use of low-dose aspirin for cardiac prophylaxis, inform patients of the increased risk for the signs and symptoms of GI bleeding [see Warnings and Precautions (5.2)].
Hepatotoxicity
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, instruct patients to stop ANJESO and seek immediate medical therapy [see Warnings and Precautions (5.3)].
Heart Failure and Edema
Advise patients to be alert for the symptoms of congestive heart failure including shortness of breath, unexplained weight gain, or edema and to contact their healthcare provider if such symptoms occur [see Warnings and Precautions (5.5)].
Anaphylactic Reactions
Inform patients of the signs of an anaphylactic reaction (e.g., difficulty breathing, swelling of the face or throat). Instruct patients to seek immediate emergency help if these occur [see Contraindications (4) and Warnings and Precautions (5.7)].
Serious Skin Reactions including DRESS
Advise patients to stop taking ANJESO immediately if they develop any type of rash or fever and to contact their healthcare provider as soon as possible [see Warnings and Precautions (5.9,5.10)].
Female Fertility
Advise females of reproductive potential who desire pregnancy that NSAIDs, including ANJESO, may be associated with a reversible delay in ovulation [see Use in Specific Populations (8.3)].
Fetal Toxicity
Inform pregnant women to avoid use of ANJESO and other NSAIDs starting at 30 weeks gestation because of the risk of the premature closing of the fetal ductus arteriosus. If treatment with ANJESO is needed for a pregnant woman between about 20 to 30 weeks gestation, advise her that she may need to be monitored for oligohydramnios, if treatment continues for longer than 48 hours [see Warnings and Precautions (5.11) and Use in Specific Populations (8.1)].
Avoid Concomitant Use of NSAIDs
Inform patients that the concomitant use of ANJESO with other NSAIDs or salicylates (e.g., diflunisal, salsalate) is not recommended due to the increased risk of gastrointestinal toxicity, and little or no increase in efficacy [see Warnings and Precautions (5.2) and Drug Interactions (7)]. Alert patients that NSAIDs may be present in “over the counter” medications for treatment of colds, fever, or insomnia.
Use of NSAIDs and Low-Dose Aspirin
Inform patients not to use low-dose aspirin concomitantly with ANJESO until they talk to their healthcare provider [see Drug Interactions (7)].
This PRESCRIBING INFORMATION has been approved by the US Food and Drug
Administration.
Manufactured for:
Baudax Bio, Inc.
Malvern, PA 19355 USA
Made in Italy
Prescribing Information issued: July 2021
216277
| ANJESO
meloxicam injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baudax Bio, Inc. (117213919) |