IMPEKLO- clobetasol propionate lotion
Mylan Specialty L.P.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use IMPEKLO, safely and effectively. See full prescribing information for IMPEKLO.
IMPEKLO™ (clobetasol propionate) lotion, for topical use Initial U.S. Approval: 1985 INDICATIONS AND USAGEIMPEKLO lotion is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses, in patients 18 years of age or older (1). Limitations of Use: Do not in the treatment of rosacea or perioral dermatitis. DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSLotion, 0.05% w/w (3) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions (incidence > 1%) are skin atrophy, telangiectasia, discomfort skin and skin dry (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 5/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Indication
IMPEKLO lotion is a super-high potent topical corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 18 years of age or older.
1.2 Limitations of Use
IMPEKLO lotion should not be used in the treatment of rosacea or perioral dermatitis.
Use in patients under 18 years of age is not recommended due to numerically high rates of HPA axis suppression [see Warnings and Precautions (5.1) and Use in Specific Populations (8.4)].
2 DOSAGE AND ADMINISTRATION
IMPEKLO lotion is for topical use only, and not for ophthalmic, oral or intravaginal use.
Avoid use of IMPEKLO lotion on the face, axillae, or groin and avoid use if there is atrophy at the treatment site.
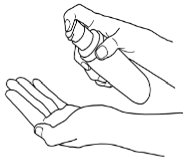
IMPEKLO lotion should be applied to the affected skin areas twice daily and rubbed in gently and completely. Avoid contact with eyes. Wash hands after each application.
The total dosage should not exceed 50 g per week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis. Do not use more than 10 pump actuations per application twice daily or 20 pump actuations per day for more than 7 days.
IMPEKLO lotion contains a topical corticosteroid; therefore treatment should be limited to 2 consecutive weeks for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses and up to 2 additional weeks in localized lesions (less than 10% body surface area) of moderate to severe plaque psoriasis that have not sufficiently improved after the initial 2 weeks of treatment. Discontinue use when control is achieved.
Unless directed by physician, IMPEKLO lotion should not be used with occlusive dressings.
3 DOSAGE FORMS AND STRENGTHS
IMPEKLO lotion is a white to off white, opaque to translucent, homogenous and lump free lotion without any phase separation packed in a white bottle with a metered-dose pump having an integral pump locking feature. Each pump actuation delivers on average 0.15 mg of clobetasol propionate, USP in 0.30 g of lotion.
5 WARNINGS AND PRECAUTIONS
5.1 Effects on the Endocrine System
Clobetasol propionate is a highly potent topical corticosteroid that has been shown to suppress the HPA axis at the lowest doses tested.
Systemic absorption of topical corticosteroids has caused reversible adrenal suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of the topical corticosteroid.
The effect of clobetasol propionate lotion, 0.05% on HPA axis function was assessed in adults in two trials, one for psoriasis and one for atopic dermatitis. In total, 8 of 10 evaluable subjects with moderate to severe plaque psoriasis experienced adrenal suppression following 4 weeks of clobetasol propionate lotion, 0.05% therapy. In follow-up testing, 1 of 2 subjects remained suppressed after 8 days.
Furthermore, 5 of 9 evaluable subjects with moderate to severe atopic dermatitis experienced adrenal suppression following 2 weeks of clobetasol propionate lotion, 0.05% therapy. Of the 3 subjects that had follow-up testing, one subjects failed to recover adrenal function 7 days post-treatment. The proportion of subjects suppressed may be underestimated because the adrenal glands were stimulated weekly with cosyntropin in these trials.
Because of the potential for systemic absorption, use of topical corticosteroids may require that patients be periodically evaluated for HPA axis suppression. Factors that predispose a patient using a topical corticosteroid to HPA axis suppression include the use of more potent steroids, use over large surface areas, use over prolonged periods, use under occlusion, use on an altered skin barrier, young age, concomitant use of multiple corticosteroid-containing products and use in patients with liver failure.
Cushing’s syndrome, hyperglycemia, glycosuria, and unmasking of latent diabetes mellitus can also result from systemic absorption of topical corticosteroids.
An adrenocorticotropic hormone (ACTH) stimulation test may be helpful in evaluating patients for HPA axis suppression. If HPA axis suppression is documented, an attempt should be made to gradually withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt and complete upon discontinuation of topical corticosteroids.
Pediatric patients may be more susceptible to systemic toxicity from use of topical corticosteroids. Use in patients under 18 years of age is not recommended due to numerically high rates of HPA axis suppression [see Use in Specific Populations (8.4)].
5.2 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including IMPEKLO, may increase the risks of glaucoma and posterior subcapsular cataract. Glaucoma and cataracts have been reported in postmarketing experience with the use of topical corticosteroid products, including topical clobetasol products.
Avoid contact of IMPEKLO with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
5.3 Local Adverse Reactions with Topical Corticosteroids
Local adverse reactions may occur more frequently with the use of occlusive dressings and higher potency corticosteroids, including clobetasol propionate. These reactions are listed in an approximate decreasing order of occurrence: folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, striae, miliaria, skin atrophy and telangiectasia. Some local adverse reactions may be irreversible. Clobetasol propionate is not recommended in patients with acne vulgaris, rosacea or perioral dermatitis.
5.4 Allergic Contact Dermatitis
Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than a clinical exacerbation. Clinical diagnosis of allergic contact dermatitis can be confirmed by patch testing.
5.5 Concomitant Skin Infections
In the presence of dermatologic infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, use of IMPEKLO lotion should be discontinued until the infection has been adequately controlled.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- •
- Effects on the Endocrine System [see Warnings and Precautions (5.1)]
- •
- Ophthalmic Adverse Reactions [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In controlled, clinical trials with clobetasol propionate lotion, 0.05%, the following adverse reactions have been reported: burning/stinging, skin dryness, irritation, erythema, folliculitis, pruritus, skin atrophy, and telangiectasia. The pooled incidence of local adverse reactions in trials for psoriasis and atopic dermatitis with clobetasol propionate lotion, 0.05% at 1% or greater was:
|
Adverse Reaction |
Incidence |
|
Skin Atrophy |
4.2% |
|
Telangiectasia |
3.2% |
|
Discomfort Skin |
1.3% |
|
Skin Dry |
1.0% |
Most local adverse events were rated as mild to moderate and they are not affected by age, race or gender.
Systemic absorption of topical corticosteroids has produced hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during post-approval use of clobetasol propionate lotion, 0.05%.
- •
- Endocrine Disorders: Cushing’s syndrome, adrenal suppression
- •
- Eye Disorders: cataracts, glaucoma and increased intraocular pressure.
- •
- Skin: rash, pain of skin, skin exfoliation, skin chapped, scaling, induration/papulation, lichenification.
- •
- Other: psoriasis (aggravation), plaque elevation, excoriation.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data for the use of IMPEKLO during pregnancy to inform any drug-associated risks of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Published data report an increased risk of low birthweight with the use of greater than 300 grams of potent or very potent topical corticosteroid during a pregnancy. Advise pregnant women of the potential risk to a fetus and to use IMPEKLO on the smallest area of skin and for the shortest duration possible (see Data). In animal reproduction studies, increased malformations, such as cleft palate and skeletal abnormalities, were observed after subcutaneous administration of clobetasol propionate to pregnant mice and rabbits during the period of organogenesis. The available data do not support relevant comparisons of systemic clobetasol propionate exposures achieved in the animal studies to exposures observed in humans after topical use of IMPEKLO.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Multiple observational studies found no significant associations between maternal use of topical corticosteroids of any potency and congenital malformations, preterm delivery, or fetal mortality. However, when the dispensed amount of potent or very potent topical corticosteroid exceeded 300 g during the entire pregnancy, use was associated with an increase in low birth weight infants. In addition, a small cohort study, in which 28 sub-Saharan women using potent topical corticosteroids (27/28 used clobetasol propionate 0.05%) for skin lightening during pregnancy, noted a higher incidence of low birth weight infants in the exposed group. The majority of exposed subjects treated large areas of the body (a mean quantity of 60 g/month (range, 12-170g) over long periods of time.
Animal Data
In embryofetal development studies in mice, subcutaneous administration of clobetasol propionate during the period of organogenesis resulted in malformations at all dose levels, ranging from 0.03 to 1 mg/kg. Malformations included cleft palate and skeletal abnormalities; fetotoxicity was observed at the high dose (1 mg/kg).
In an embryofetal development study in rabbits, subcutaneous administration of clobetasol propionate during the period of organogenesis resulted in malformations at doses of 0.003 and 0.01 mg/kg. Malformations included cleft palate, cranioschisis, and other skeletal abnormalities.
In a prenatal and postnatal development study in rats, clobetasol propionate was administered subcutaneously to female rats twice daily (0, 12.5, 25, and 50 µg/kg/day) from gestation day 7 through lactation day 25. In dams, body weight gain and food consumption were reduced during gestation at all doses and prolonged delivery occurred at the high dose. A maternal no-observed-effect-level (NOEL) could not be determined and the reproductive NOEL for dams was 25 µg/kg/day. In offspring, doses ≥25 µg/kg/day increased incidence of stillbirths, reduced pup body weights on lactation days 1 and 7, increased pup mortality, increased the incidence of umbilical hernia, increased the incidence of cysts on the kidney, and significantly reduced epididymides and testes weights. However, no effects were observed on the mating and fertility of the offspring. The no-observed-adverse-effect-level (NOAEL) for viability and growth in the offspring was 12.5 µg/kg/day.
8.2 Lactation
Risk Summary
There is no information regarding the presence of clobetasol propionate in breast milk or its effects on the breastfed infant or on milk production. Systemically administered corticosteroids appear in human milk and can suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of clobetasol propionate could result in sufficient systemic absorption to produce detectable quantities in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for IMPEKLO and any potential adverse effects on the breastfed infant from clobetasol propionate or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the breastfed infant via breast milk, use IMPEKLO on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply IMPEKLO directly to the nipple and areola to avoid direct infant exposure.
8.4 Pediatric Use
Use of clobetasol propionate lotion, 0.05% in pediatric patients is not recommended due to the potential for HPA axis suppression [see Warnings and Precautions (5.1)].
The HPA axis suppression potential of clobetasol propionate lotion, 0.05% has been studied in adolescents (12 to 17 years of age) with moderate to severe atopic dermatitis covering a minimum of 20% of the total body surface area. Subjects were treated twice daily for 2 weeks with clobetasol propionate lotion, 0.05%. After 2 weeks of treatment, 9 out of 14 of the subjects experienced adrenal suppression. One out of 4 subjects who were retested remained suppressed two weeks post-treatment.
Because of higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing’s syndrome when they are treated with topical corticosteroids. They are therefore also at greater risk of glucocorticosteroid insufficiency during and/or after withdrawal of treatment. Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children.
HPA axis suppression, Cushing’s syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
8.5 Geriatric Use
Clinical studies of clobetasol propionate lotion, 0.05% did not include sufficient numbers of subjects aged 65 and over to adequately determine whether they respond differently than younger subjects. In general, dose selection for an elderly patient should be made with caution, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
10 OVERDOSAGE
Topically applied IMPEKLO lotion can be absorbed in sufficient amount to produce systemic effects [see Warnings and Precautions (5.1)].
11 DESCRIPTION
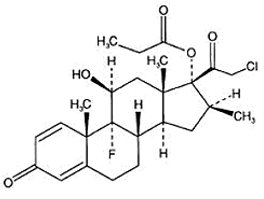
IMPEKLO (clobetasol propionate) lotion contains clobetasol propionate, a synthetic fluorinated corticosteroid, for topical use. The corticosteroids constitute a class of primarily synthetic steroids used topically as anti-inflammatory and antipruritic agents. Clobetasol propionate is 21-chloro-9-fluoro-11β, 17-dihydroxy-16β -methylpregna-1,4-diene-3,20-dione 17-propionate, with the empirical formula C25H32CIFO5, and a molecular weight of 466.97 (CAS Registry Number 25122-46-7).
The following is the chemical structure:
Clobetasol propionate, USP is a white to almost white crystalline powder that is practically insoluble in water. IMPEKLO (clobetasol propionate) lotion is a white to off white, opaque to translucent, homogenous and lump free lotion composed of carbomer 1342, hypromellose, mineral oil, PEG-6 isostearate, propylene glycol, purified water and sodium hydroxide. Each pump actuation delivers 0.15 mg of clobetasol propionate in 0.30 g of lotion.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in corticosteroid-responsive dermatoses is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
IMPEKLO lotion is in the super-high range of potency as demonstrated in vasoconstrictor studies in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
In adults treated with clobetasol propionate lotion, 0.05%, hypothalamic-pituitary-adrenal (HPA) axis suppression, rate was 80% in subjects with psoriasis and 56% in subjects with atopic dermatitis. In adolescent subjects the rate of HPA axis suppression was 64% in subjects with atopic dermatitis. [see Warnings and Precautions (5.1) and Use in Specific Population (8.4)].
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier and occlusion.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and other disease processes in the skin may increase percutaneous absorption.
There are no human data regarding the distribution of corticosteroids to body organs following topical application. Nevertheless, once absorbed through the skin, topical corticosteroids are handled through metabolic pathways similar to systemically administered corticosteroids. They are metabolized, primarily in the liver, and are then excreted by the kidneys. In addition, some corticosteroids and their metabolites are also excreted in the bile.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study, clobetasol propionate was topically applied to rats at concentrations up to 0.005%. No drug-related increase in tumor incidence was observed.
Clobetasol propionate was negative in the in vitro mammalian chromosomal aberration test and in the in vivo mammalian erythrocyte micronucleus test.
In a fertility and reproductive toxicity study, clobetasol propionate was administered subcutaneously to rats at doses of 0, 12.5, 25, and 50 μg/kg/day. Males were treated beginning 70 days before mating and females beginning 15 days before mating through day 7 of gestation. A NOEL could not be determined for paternal and maternal toxicity or male reproductive toxicity because of decreased weight gain in both sexes and increased seminal vesicle weights in males. The female reproductive NOEL was 12.5 μg/kg/day because higher doses reduced the number of estrous cycles during the pre-cohabitation period and increased in the number of nonviable embryos.
14 CLINICAL STUDIES
The efficacy of clobetasol propionate lotion, 0.05% in psoriasis and atopic dermatitis has been demonstrated in two adequate and well-controlled clinical trials. The first trial was conducted in subjects with moderate to severe plaque psoriasis. Subjects were treated twice daily for 4 weeks with either clobetasol propionate lotion, 0.05% or vehicle lotion. Trial results demonstrated that the efficacy of clobetasol propionate lotion, 0.05% in treating moderate to severe plaque psoriasis was superior to that of vehicle.
At the end of treatment (4 weeks), 30 of 82 subjects (36.6%) treated with clobetasol propionate lotion, 0.05% compared with 0 of 29 (0%) treated with vehicle achieved success. Success was defined as a score of none or very mild (no or very slight clinical signs or symptoms of erythema, plaque elevation, or scaling) on the Global Severity scale of psoriasis.
The second trial was conducted in subjects with moderate to severe atopic dermatitis. Subjects were treated twice daily for 2 weeks with either clobetasol propionate lotion, 0.05% or vehicle lotion. Trial results demonstrated that the efficacy of clobetasol propionate lotion, 0.05% in treating moderate to severe atopic dermatitis was superior to that of vehicle.
At the end of treatment (2 weeks), 41 of 96 subjects (42.7%) treated with clobetasol propionate lotion, 0.05% compared with 4 of 33 (12.1%) treated with vehicle achieved success. Success was defined as a score of none or very mild (no or very slight clinical signs or symptoms of erythema, induration/papulation, oozing/crusting, or pruritus) on the Global Severity scale of atopic dermatitis.
16 HOW SUPPLIED/STORAGE AND HANDLING
IMPEKLO™ lotion, 0.05% is a white to off white, opaque to translucent, homogenous and lump free lotion without any phase separation provided in a white bottle with a metered-dose pump having an integral pump locking feature. Each pump actuation delivers 0.15 mg of clobetasol propionate, USP in 0.30 g of lotion. The metered-dose pump is capable of dispensing not less than 138 actuations to deliver not less than 41.4 g of lotion. It is available as follows:
NDC 49502-537-35
one metered dose pump contains 68 g lotion
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Protect from freezing.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all administration instructions or all possible adverse or unintended effects.
Advise patients using IMPEKLO lotion of the following information and instructions:
Important Administration Instructions
- •
- This medication is to be used as directed by the physician. It is for external use only. Avoid use on the face or in skin-fold areas, such as the underarms or groin. Avoid contact with the eyes or other mucous membranes. Wash hands after use.
- •
- Therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician. Avoid use of IMPEKLO longer than the prescribed time period.
- •
- Avoid use for more than 50 g per week (i.e., 20 actuations/day for 7 days) of IMPEKLO lotion. [see Dosage and Administration (2)].
Effects on the Endocrine System
IMPEKLO may cause HPA axis suppression. Advise patients that use of topical corticosteroids, including IMPEKLO, may require periodic evaluation for HPA axis suppression. Topical corticosteroids may have other endocrine effects. Concomitant use of multiple corticosteroid-containing products may increase the total systemic exposure to topical corticosteroids. Patients should inform their physician(s) that they are using IMPEKLO if surgery is contemplated [see Warnings and Precautions (5.1)].
Ophthalmic Adverse Reactions
Advise patients to report any visual symptoms to their healthcare providers [see Warnings and Precautions (5.2)].
Local Adverse Reactions
Advise patients that local reactions and skin atrophy are more likely to occur with occlusive use or prolonged use [see Warnings and Precautions (5.2)].
Pregnancy
Advise pregnant women of the potential risk to a fetus and to use IMPEKLO on the smallest area of skin and for the shortest duration possible [see Use in Specific Populations (8.1)].
Lactation
Advise a woman to use IMPEKLO on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply IMPEKLO directly to the nipple and areola to avoid direct infant exposure [see Use in Specific Populations (8.2)].
Patient Information
|
IMPEKLO™ (im pek' loe) (clobetasol propionate) lotion |
|
Important: IMPEKLO lotion is for use on the skin only. Do not get IMPEKLO lotion near or in your eyes, mouth or vagina. |
|
What is IMPEKLO lotion? IMPEKLO lotion is a prescription corticosteroid medicine used on the skin (topical) for the relief of swelling (inflammation) and itching caused by certain skin conditions in people 18 years of age and older. IMPEKLO lotion should not be used for the treatment of acne vulgaris, rosacea or red, dry itchy skin (dermatitis) around the mouth. IMPEKLO lotion is not recommended in children under 18 years of age. |
|
Before you use IMPEKLO lotion, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription or over-the-counter medicines, vitamins, and herbal supplements. Do not use other products containing corticosteroid medicine during treatment with IMPEKLO lotion without talking to your healthcare provider first. |
|
How should I use IMPEKLO lotion? See the “Instructions for Use” for detailed information about the right way to apply IMPEKLO lotion.
|
|
What are the possible side effects of IMPEKLO lotion? IMPEKLO lotion may cause serious side effects, including:
The most common side effects of IMPEKLO lotion include thinning of the skin (atrophy), widening of small veins in the skin, skin discomfort and skin dryness. These are not all of the possible side effects of IMPEKLO lotion. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store IMPEKLO lotion?
Keep IMPEKLO lotion and all medicines out of reach of children. |
|
General information about the safe and effective use of IMPEKLO lotion. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use IMPEKLO lotion for a condition for which it was not prescribed. Do not give IMPEKLO lotion to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about IMPEKLO lotion that is written for health professionals. |
|
What are the ingredients in IMPEKLO lotion? Active ingredient: clobetasol propionate Inactive ingredients: carbomer 1342, hypromellose, mineral oil, PEG-6 isostearate, propylene glycol, purified water and sodium hydroxide.
Manufactured for:
Manufactured by:
IMPEKLO is a trademark of Mylan Specialty L.P. For more information, call Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or go to www.impeklo.com. |
This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 5/2020
INSTRUCTIONS FOR USE
IMPEKLO™ (im pek' loe)
(clobetasol propionate)
lotion
This Instructions for Use contains information on how to apply IMPEKLO lotion.
Important: IMPEKLO lotion is for use on the skin only (topical). Do not get IMPEKLO lotion near or in your eyes, mouth or vagina.
IMPEKLO lotion comes in a pump bottle that delivers a measured amount of IMPEKLO lotion each time you press the pump.
Before you use a new bottle of IMPEKLO lotion for the first time, you will need to prime the pump.
Manufactured for:
Mylan Specialty L.P.
Morgantown, WV 26505 U.S.A.
Manufactured by:
Lupin Limited
Pithampur (M.P.) – 454 775, India
IMPEKLO is a trademark of Mylan Specialty L.P.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 5/2020
Revised: 5/2020
CLOBLON:R1p/PL:CLOBLON:PIL:R1p/PL:CLOBLON:IFU:R1p
265498
PRINCIPAL DISPLAY PANEL – 0.05%
NDC 49502-537-35 Rx only
Impeklo™
Clobetasol Propionate Lotion
0.05% Metered-Dose Pump
0.15 mg of clobetasol
propionate per
pump actuation*
*Each actuation delivers 0.30 g of lotion
Metered-dose pump
capable of dispensing
not less than
138 actuations to
deliver not less than
41.4 g of lotion.
Topical Lotion
For external use only.
Not for eye use.
68 g
Contains: Clobetasol propionate
USP, carbomer 1342, hypromellose,
mineral oil, PEG-6 isostearate,
propylene glycol, purified water and
sodium hydroxide.
Recommended Dosage: Apply
twice daily, once in the morning
and once at night (not to exceed
10 actuations/application or
20 actuations/day for more than
7 days). Use only enough to cover
the affected areas. Do not apply
ImpekloTM to the face, underarms,
or groin, and avoid contact with
eyes and lips. See accompanying
prescribing information.
Warnings: Keep this and all
medication out of the reach of
children. Not a child resistant
closure.
Storage: Store at 25°C (77°F);
excursions permitted to 15° to 30°C
(59° to 86°F) [see USP Controlled
Room Temperature].
Protect from freezing.
Impeklo is a trademark of
Mylan Specialty L.P.
©2020 Mylan Specialty L.P.
Code No.: 25/01/2016
MS:LUP:537:1C:R4
Instructions for Pump Use:
 |
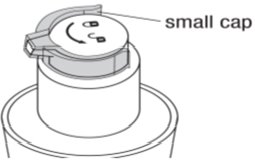
Remove the large cap |
 |
Remove the small cap around the tip of the pump |
 |
Turn the pump counter clockwise from the “lock” ( |
Manufactured for: Mylan Specialty L.P.
Morgantown, WV 26505 U.S.A.
Manufactured by: Lupin Limited
Pithampur (M.P.) - 454 775, INDIA
Mylan.com
| IMPEKLO
clobetasol propionate lotion |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Mylan Specialty L.P. (194775557) |
 ) position to the “unlock” (
) position to the “unlock” ( ) position.
) position.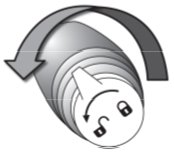
 ) position to the “lock” (
) position to the “lock” ( ) position. Unlock the pump when you are ready to use.
) position. Unlock the pump when you are ready to use. 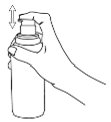
 ) position to the “lock” (
) position to the “lock” ( ) position.
) position.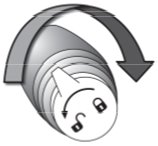
 ) position to the “unlock” (
) position to the “unlock” ( ) position
) position