Beclomethasone dipropionate, monohydrate, the active component of BECONASE AQ Nasal Spray, is an anti-inflammatory steroid having the chemical name 9-chloro-11β,17,21-trihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17,21-dipropionate, monohydrate and the following chemical structure:

Beclomethasone 17,21-dipropionate is a diester of beclomethasone, a synthetic halogenated corticosteroid. Beclomethasone dipropionate, monohydrate is a white to creamy-white, odorless powder with a molecular weight of 539.06. It is very slightly soluble in water, very soluble in chloroform, and freely soluble in acetone and in ethanol.
BECONASE AQ Nasal Spray is a metered-dose, manual pump spray unit containing a microcrystalline suspension of beclomethasone dipropionate, monohydrate equivalent to 42 mcg of beclomethasone dipropionate, calculated on the dried basis, in an aqueous medium containing microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, benzalkonium chloride, polysorbate 80, and 0.25% v/w phenylethyl alcohol. The pH through expiry is 5.0 to 6.8.
After initial priming (6 actuations), each actuation of the pump delivers from the nasal adapter 100 mg of suspension containing beclomethasone dipropionate, monohydrate equivalent to 42 mcg of beclomethasone dipropionate. If the pump is not used for 7 days, it should be primed until a fine spray appears. Each 25-g bottle of BECONASE AQ Nasal Spray provides 180 metered sprays.
Following topical administration, beclomethasone dipropionate produces anti-inflammatory and vasoconstrictor effects. The mechanisms responsible for the anti-inflammatory action of beclomethasone dipropionate are unknown. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, and cytokines) involved in inflammation. The direct relationship of these findings to the effects of beclomethasone dipropionate on allergic rhinitis symptoms is not known.
Biopsies of nasal mucosa obtained during clinical studies showed no histopathologic changes when beclomethasone dipropionate was administered intranasally.
Beclomethasone dipropionate is a pro-drug with weak glucocorticoid receptor binding affinity. It is hydrolyzed via esterase enzymes to its active metabolite beclomethasone-17-monopropionate (B-17-MP), which has high topical anti-inflammatory activity.
PharmacokineticsAbsorptionBeclomethasone dipropionate is sparingly soluble in water. When given by nasal inhalation in the form of an aqueous or aerosolized suspension, the drug is deposited primarily in the nasal passages. The majority of the drug is eventually swallowed. Following intranasal administration of aqueous beclomethasone dipropionate, the systemic absorption was assessed by measuring the plasma concentrations of its active metabolite B-17-MP, for which the absolute bioavailability following intranasal administration is 44% (43% of the administered dose came from the swallowed portion and only 1% of the total dose was bioavailable from the nose). The absorption of unchanged beclomethasone dipropionate following oral and intranasal dosing was undetectable (plasma concentrations less than 50 pg/mL).
DistributionThe tissue distribution at steady state for beclomethasone dipropionate is moderate (20 L) but more extensive for B-17-MP (424 L). There is no evidence of tissue storage of beclomethasone dipropionate or its metabolites. Plasma protein binding is moderately high (87%).
MetabolismBeclomethasone dipropionate is cleared very rapidly from the systemic circulation by metabolism mediated via esterase enzymes that are found in most tissues. The main product of metabolism is the active metabolite (B-17-MP). Minor inactive metabolites, beclomethasone-21-monopropionate (B-21-MP) and beclomethasone (BOH), are also formed, but these contribute little to systemic exposure.
EliminationThe elimination of beclomethasone dipropionate and B-17-MP after intravenous administration are characterized by high plasma clearance (150 and 120 L/hour) with corresponding terminal elimination half-lives of 0.5 and 2.7 hours. Following oral administration of tritiated beclomethasone dipropionate, approximately 60% of the dose was excreted in the feces within 96 hours, mainly as free and conjugated polar metabolites. Approximately 12% of the dose was excreted as free and conjugated polar metabolites in the urine. The renal clearance of beclomethasone dipropionate and its metabolites is negligible.
PharmacodynamicsThe effects of beclomethasone dipropionate on hypothalamic-pituitary-adrenal (HPA) function have been evaluated in adult volunteers by other routes of administration. Studies with beclomethasone dipropionate by the intranasal route may demonstrate that there is more or that there is less absorption by this route of administration. There was no suppression of early morning plasma cortisol concentrations when beclomethasone dipropionate was administered in a dose of 1,000 mcg/day for 1 month as an oral aerosol or for 3 days by intramuscular injection. However, partial suppression of plasma cortisol concentrations was observed when beclomethasone dipropionate was administered in doses of 2,000 mcg/day either by oral aerosol or intramuscular injection. Immediate suppression of plasma cortisol concentrations was observed after single doses of 4,000 mcg of beclomethasone dipropionate. Suppression of HPA function (reduction of early morning plasma cortisol levels) has been reported in adult patients who received 1,600-mcg daily doses of oral beclomethasone dipropionate for 1 month. In clinical studies using beclomethasone dipropionate aerosol intranasally, there was no evidence of adrenal insufficiency. The effect of BECONASE AQ Nasal Spray on HPA function was not evaluated but would not be expected to differ from intranasal beclomethasone dipropionate aerosol.
In 1 study in children with asthma, the administration of inhaled beclomethasone at recommended daily doses for at least 1 year was associated with a reduction in nocturnal cortisol secretion. The clinical significance of this finding is not clear. It reinforces other evidence, however, that topical beclomethasone may be absorbed in amounts that can have systemic effects and that physicians should be alert for evidence of systemic effects, especially in chronically treated patients (see PRECAUTIONS).
BECONASE AQ Nasal Spray is indicated for the relief of the symptoms of seasonal or perennial allergic and nonallergic (vasomotor) rhinitis.
Results from 2 clinical trials have shown that significant symptomatic relief was obtained within 3 days. However, symptomatic relief may not occur in some patients for as long as 2 weeks. BECONASE AQ Nasal Spray should not be continued beyond 3 weeks in the absence of significant symptomatic improvement. BECONASE AQ Nasal Spray should not be used in the presence of untreated localized infection involving the nasal mucosa.
BECONASE AQ Nasal Spray is also indicated for the prevention of recurrence of nasal polyps following surgical removal.
Clinical studies have shown that treatment of the symptoms associated with nasal polyps may have to be continued for several weeks or more before a therapeutic result can be fully assessed. Recurrence of symptoms due to polyps can occur after stopping treatment, depending on the severity of the disease.
Hypersensitivity to any of the ingredients of this preparation contraindicates its use.
The replacement of a systemic corticosteroid with BECONASE AQ Nasal Spray can be accompanied by signs of adrenal insufficiency.
Careful attention must be given when patients previously treated for prolonged periods with systemic corticosteroids are transferred to BECONASE AQ Nasal Spray. This is particularly important in those patients who have associated asthma or other clinical conditions where too rapid a decrease in systemic corticosteroids may cause a severe exacerbation of their symptoms.
If recommended doses of intranasal beclomethasone are exceeded or if individuals are particularly sensitive or predisposed by virtue of recent systemic steroid therapy, symptoms of hypercorticism may occur, including very rare cases of menstrual irregularities, acneiform lesions, cataracts, and cushingoid features. If such changes occur, BECONASE AQ Nasal Spray should be discontinued slowly consistent with accepted procedures for discontinuing oral steroid therapy.
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In children or adults who have not had these diseases or been properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents may be considered.
Avoid spraying in eyes.
Intranasal corticosteroids may cause a reduction in growth velocity when administered to pediatric patients (see PRECAUTIONS: Pediatric Use).
During withdrawal from oral corticosteroids, some patients may experience symptoms of withdrawal, e.g., joint and/or muscular pain, lassitude, and depression.
Rarely, immediate hypersensitivity reactions may occur after the intranasal administration of beclomethasone (see ADVERSE REACTIONS).
Rare instances of nasal septum perforation have been spontaneously reported.
Rare instances of wheezing, cataracts, glaucoma, and increased intraocular pressure have been reported following the intranasal use of beclomethasone dipropionate.
In clinical studies with beclomethasone dipropionate administered intranasally, the development of localized infections of the nose and pharynx with Candida albicans has occurred only rarely. When such an infection develops, it may require treatment with appropriate local therapy and discontinuation of treatment with BECONASE AQ Nasal Spray.
If persistent nasopharyngeal irritation occurs, it may be an indication for stopping BECONASE AQ Nasal Spray.
Beclomethasone dipropionate is absorbed into the circulation. Use of excessive doses of BECONASE AQ Nasal Spray may suppress HPA function.
Intranasal corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculous infections of the respiratory tract, untreated local or systemic fungal or bacterial infections, systemic viral or parasitic infections, or ocular herpes simplex.
For BECONASE AQ Nasal Spray to be effective in the treatment of nasal polyps, the spray must be able to enter the nose. Therefore, treatment of nasal polyps with BECONASE AQ Nasal Spray should be considered adjunctive therapy to surgical removal and/or the use of other medications that will permit effective penetration of BECONASE AQ Nasal Spray into the nose. Nasal polyps may recur after any form of treatment.
As with any long-term treatment, patients using BECONASE AQ Nasal Spray over several months or longer should be examined periodically for possible changes in the nasal mucosa.
Because of the inhibitory effect of corticosteroids on wound healing, patients who have experienced recent nasal septal ulcers, nasal surgery, or nasal trauma should not use a nasal corticosteroid until healing has occurred.
Although systemic effects have been minimal with recommended doses, this potential increases with excessive doses. Therefore, larger than recommended doses should be avoided.
In general, side effects in clinical studies have been primarily associated with irritation of the nasal mucous membranes.
Adverse reactions reported in controlled clinical trials and open studies in patients treated with BECONASE AQ Nasal Spray are described below.
Mild nasopharyngeal irritation following the use of beclomethasone aqueous nasal spray has been reported in up to 24% of patients treated, including occasional sneezing attacks (about 4%) occurring immediately following use of the spray. In patients experiencing these symptoms, none had to discontinue treatment. The incidence of transient irritation and sneezing was approximately the same in the group of patients who received placebo in these studies, implying that these complaints may be related to vehicle components of the formulation.
Fewer than 5 per 100 patients reported headache, nausea, or lightheadedness following the use of BECONASE AQ Nasal Spray. Fewer than 3 per 100 patients reported nasal stuffiness, nosebleeds, rhinorrhea, or tearing eyes.
Rare cases of ulceration of the nasal mucosa and instances of nasal septum perforation have been spontaneously reported (see PRECAUTIONS).
Reports of dryness and irritation of the nose and throat and unpleasant taste and smell have been received. There are rare reports of loss of taste and smell.
Rare instances of wheezing, cataracts, glaucoma, and increased intraocular pressure have been reported following the use of intranasal beclomethasone dipropionate (see PRECAUTIONS).
Rare cases of immediate and delayed hypersensitivity reactions, including anaphylactoid/anaphylactic reactions, urticaria, angioedema, rash, and bronchospasm, have been reported following the oral and intranasal inhalation of beclomethasone dipropionate.
Cases of growth suppression have been reported for intranasal corticosteroids, including BECONASE AQ (see PRECAUTIONS: Pediatric Use).
When used at excessive doses, systemic corticosteroid effects such as hypercorticism and adrenal suppression may appear. If such changes occur, BECONASE AQ Nasal Spray should be discontinued slowly consistent with accepted procedures for discontinuing oral steroid therapy. No deaths occurred when beclomethasone dipropionate was given as single oral doses of 3,000 mg/kg to mice (approximately 36,000 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis, or approximately 21,000 times the maximum recommended daily intranasal dose in children on a mg/m2 basis) and 2,000 mg/kg to rats (approximately 48,000 times the maximum recommended daily intranasal dose in adults or approximately 29,000 times the maximum recommended daily intranasal dose in children on a mg/m2 basis). One bottle of BECONASE AQ Nasal Spray contains beclomethasone dipropionate, monohydrate equivalent to 10.5 mg of beclomethasone dipropionate; therefore, acute overdosage is unlikely.
The usual dosage is 1 or 2 nasal inhalations (42 to 84 mcg) in each nostril twice a day (total dose, 168 to 336 mcg/day).
Children 6 to 12 Years of AgePatients should be started with 1 nasal inhalation in each nostril twice daily; patients not adequately responding to 168 mcg or those with more severe symptoms may use 336 mcg (2 inhalations in each nostril). Once adequate control is achieved, the dosage should be decreased to 84 mcg (1 spray in each nostril) twice daily. BECONASE AQ Nasal Spray is not recommended for children below 6 years of age.
The maximum total daily dosage should not exceed 2 sprays in each nostril twice daily (336 mcg/day).
In patients who respond to BECONASE AQ Nasal Spray, an improvement of the symptoms of seasonal or perennial rhinitis usually becomes apparent within a few days after the start of therapy with BECONASE AQ Nasal Spray. However, symptomatic relief may not occur in some patients for as long as 2 weeks. BECONASE AQ Nasal Spray should not be continued beyond 3 weeks in the absence of significant symptomatic improvement.
The therapeutic effects of corticosteroids, unlike those of decongestants, are not immediate. This should be explained to the patient in advance in order to ensure cooperation and continuation of treatment with the prescribed dosage regimen.
In the presence of excessive nasal mucous secretion or edema of the nasal mucosa, the drug may fail to reach the site of intended action. In such cases it is advisable to use a nasal vasoconstrictor during the first 2 to 3 days of therapy with BECONASE AQ Nasal Spray.
Directions for UseIllustrated Patient's Instructions for Use accompany each package of BECONASE AQ Nasal Spray.
BECONASE AQ Nasal Spray, 42 mcg is supplied in an amber glass bottle fitted with a metering atomizing pump and nasal adapter in a box of 1 (NDC 0173-0388-79) with patient’s instructions for use. Each bottle contains 25 g of suspension and will provide 180 metered sprays.
The correct amount of medication in each spray cannot be assured after 180 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
Store between 15°and 30°C (59°and 86°F).
GlaxoSmithKline
Research Triangle Park, NC 27709
April 2005 RL-2182
Patients being treated with BECONASE AQ Nasal Spray should receive the following information and instructions. This information is intended to aid them in the safe and effective use of this medication. It is not a disclosure of all possible adverse or intended effects.
Patients should use BECONASE AQ Nasal Spray at regular intervals since its effectiveness depends on its regular use. The patient should take the medication as directed. It is not acutely effective, and the prescribed dosage should not be increased. Instead, nasal vasoconstrictors or oral antihistamines may be needed until the effects of BECONASE AQ Nasal Spray are fully manifested. One to 2 weeks may pass before full relief is obtained. The patient should contact the physician if symptoms do not improve, if the condition worsens, or if sneezing or nasal irritation occurs.
For the proper use of BECONASE AQ Nasal Spray and to attain maximum improvement, the patient should read and follow carefully the patient's instructions accompanying the product.
Persons who are using immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles. Patients should also be advised that if they are exposed, medical advice should be sought without delay.
Carcinogenesis, Mutagenesis, Impairment of FertilityThe carcinogenicity of beclomethasone dipropionate was evaluated in rats that were exposed for a total of 95 weeks, 13 weeks at inhalation doses up to 0.4 mg/kg and the remaining 82 weeks at combined oral and inhalation doses up to 2.4 mg/kg. There was no evidence of carcinogenicity in this study at the highest dose, approximately 60 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis or approximately 35 times the maximum recommended daily intranasal dose in children on a mg/m2 basis.
Beclomethasone dipropionate did not induce gene mutation in bacterial cells or mammalian Chinese hamster ovary (CHO) cells in vitro. No significant clastogenic effect was seen in cultured CHO cells in vitro or in the mouse micronucleus test in vivo.
In rats, beclomethasone dipropionate caused decreased conception rates at an oral dose of 16 mg/kg (approximately 390 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis). There was no significant effect of beclomethasone dipropionate on fertility in rats at oral doses of 1.6 mg/kg (approximately 40 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis). Inhibition of the estrous cycle in dogs was observed following oral dosing at 0.5 mg/kg (approximately 40 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis). No inhibition of the estrous cycle in dogs was seen following 12 months’ exposure at an estimated inhalation dose of 0.33 mg/kg (approximately 25 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis).
PregnancyTeratogenic EffectsPregnancy Category C. Like other corticosteroids, beclomethasone dipropionate was teratogenic and embryocidal in the mouse and rabbit at a subcutaneous dose of 0.1 mg/kg in mice or 0.025 mg/kg in rabbits (approximately equal to the maximum recommended daily intranasal dose in adults on a mg/m2 basis). No teratogenicity or embryocidal effects were seen in rats when exposed to an inhalation dose of 0.1 mg/kg plus oral doses of up to 10 mg/kg per day for a combined dose of 10.1 mg/kg (approximately 240 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis).
There are no adequate and well-controlled studies in pregnant women. Beclomethasone dipropionate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic EffectsHypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully observed.
Nursing MothersIt is not known whether beclomethasone dipropionate is excreted in human milk. Because other corticosteroids are excreted in human milk, caution should be exercised when BECONASE AQ Nasal Spray is administered to a nursing woman.
Pediatric UseThe safety and effectiveness of BECONASE AQ Nasal Spray have been established in children aged 6 years and above through evidence from extensive clinical use in adult and pediatric patients. The safety and effectiveness of BECONASE AQ Nasal Spray in children below 6 years of age have not been established.
Controlled clinical studies have shown that intranasal corticosteroids may cause a reduction in growth velocity in pediatric patients. This effect has been observed in the absence of laboratory evidence of HPA axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with intranasal corticosteroids, including the impact on final adult height, are unknown. The potential for “catch-up” growth following discontinuation of treatment with intranasal corticosteroids has not been adequately studied. The growth of pediatric patients receiving intranasal corticosteroids, including BECONASE AQ Nasal Spray, should be monitored routinely (e.g., via stadiometry). The potential growth effects of prolonged treatment should be weighed against the clinical benefits obtained and the risks/benefits of treatment alternatives. To minimize the systemic effects of intranasal corticosteroids, including BECONASE AQ Nasal Spray, each patient should be titrated to the lowest dose that effectively controls his/her symptoms.
In a double-blind, controlled trial, 100 children between the ages of 6 and 9½ years with allergic rhinitis were randomized to receive aqueous intranasal beclomethasone dipropionate 168 mcg twice daily or placebo for 1 year. As measured by stadiometry, children who received beclomethasone dipropionate grew more slowly than those who received placebo. A difference in mean change in height was observed within 1 month of drug initiation. At the end of 12 months, the beclomethasone dipropionate-treated group had a growth velocity on average of 4.75 cm/year compared to 6.20 cm/year in the placebo group (p less than 0.01). While the placebo group had an expected distribution of growth velocity, approximately 50% of the beclomethasone dipropionate-treated children grew below the 10th percentile.
In children 7.3 years of age, the mean age of children in this study, the range for expected growth velocity is: boys − 3rd percentile = 4.1 cm/year, 50th percentile = 5.8 cm/year, and 97th percentile = 7.5 cm/year; girls − 3rd percentile = 4.3 cm/year, 50th percentile = 5.9 cm/year, and 97th percentile = 7.5 cm/year. The potential reversibility of the reduction in growth velocity was not studied. No significant differences were observed between the 2 groups for mean basal plasma cortisol or ACTH-stimulated plasma cortisol levels.
Geriatric UseClinical studies of BECONASE AQ Nasal Spray did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
PHARMACIST—DETACH HERE AND GIVE INSTRUCTIONS TO PATIENT
_ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _
BECONASE AQ®
(beclomethasone dipropionate,
monohydrate)
Nasal Spray, 42 mcg
For Intranasal Use Only. SHAKE WELL BEFORE USE.
Patient’s Instructions for Use
Shake the suspension spray bottle well before using it.Read complete instructionscarefully and use only as directed.
To Use:
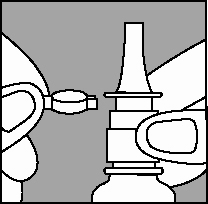
1. Remove the safety clip and the plastic dust cap from the nasal applicator (Figure 1).
2. The very first time the spray is used, prime the pump into the air by pressing downward on the white collar, using your forefinger and middle finger while supporting the base of the bottle with your thumb. When you prime the pump for the first time, press down and release the pump 6 times or until a fine spray appears (Figure 2).
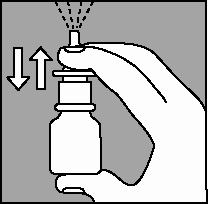
The pump is now ready for use. If the pump is not used for 7 days, prime until a fine spray appears.
3. Gently blow your nose to clear your nostrils. Close 1 nostril. Tilt your head forward slightly and, keeping the bottle upright, carefully insert the nasal applicator into the other nostril (Figure 3).
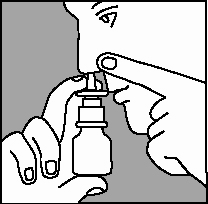
5. Breathe out through your mouth.
6. Repeat steps 5 through 7 in the other nostril.
7. Replace the plastic dust cap and safety clip.
8.DISCARD THE BOTTLE AFTER the date calculated by your doctor or pharmacist. The correct amount of medication in each spray cannot be assured after 180 sprays even though the bottle is not completely empty. Discard the bottle after 180 sprays. Before the discard date you should consult your doctor to see if a refill is needed. Do not take extra doses or stop taking BECONASE AQ Nasal Spray without consulting your doctor.
Cleansing: To clean the nasal applicator, remove the plastic dust cap and safety clip and then press gently upward on the white collar to free the nasal applicator. Wash the applicator and dust cap with cold water. Dry and replace with the plastic dust cap and safety clip back in position.
If the nasal applicator becomes blocked, remove the dust cap, unscrew the complete pump mechanism, and soak the pump in warm water for a few minutes. Rinse with cold water, dry, refit to bottle, and reprime the pump.
Caution: BECONASE AQ Nasal Spray is not intended to give rapid relief of your nasal symptoms. BECONASE AQ Nasal Spray controls the underlying disorders responsible for your attacks, so it is important that you use it regularly at the times recommended by your doctor. The full benefit of BECONASE AQ Nasal Spray may take a few days to develop.
Storage: Store between 15° and 30°C (59° and 86°F).
GlaxoSmithKline
Research Triangle Park, NC 27709
April 2005 RL-2182
Principal Display PanelNDC 0173-0388-79
Beconase AQ®
(beclomethasone dipropionate, monohydrate)
Nasal Spray, 42 mcg
25 g
180 Metered Sprays
Spray-For Intranasal Use Only
Rx only
Important: Read accompanying directions carefully.
See package insert for Dosage Information.
After initial priming (6 actuations), each actuation of the pump delivers 100 mg of aqueous suspension containing beclomethasone dipropionate, monohydrate equivalent to 42 mcg of beclomethasone dipropionate and microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, benzalkonium chloride, polysorbate 80, and 0.25% v/w phenylethyl alcohol.
Store between 15o and 30oC (59o and 86oF). Shake well before each use.
GlaxoSmithKline
Research Triangle Park, NC 27709
Made in Canada
A018016 Rev. 7/05
