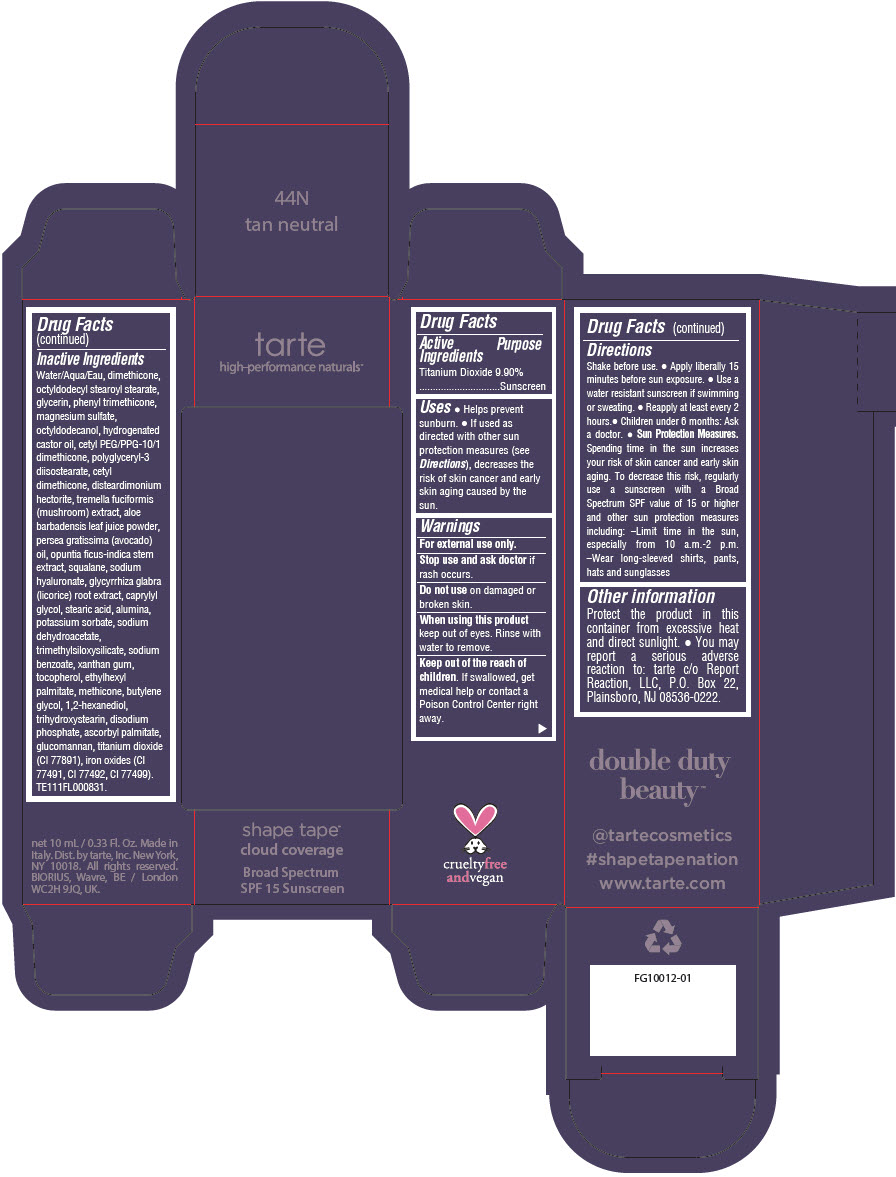
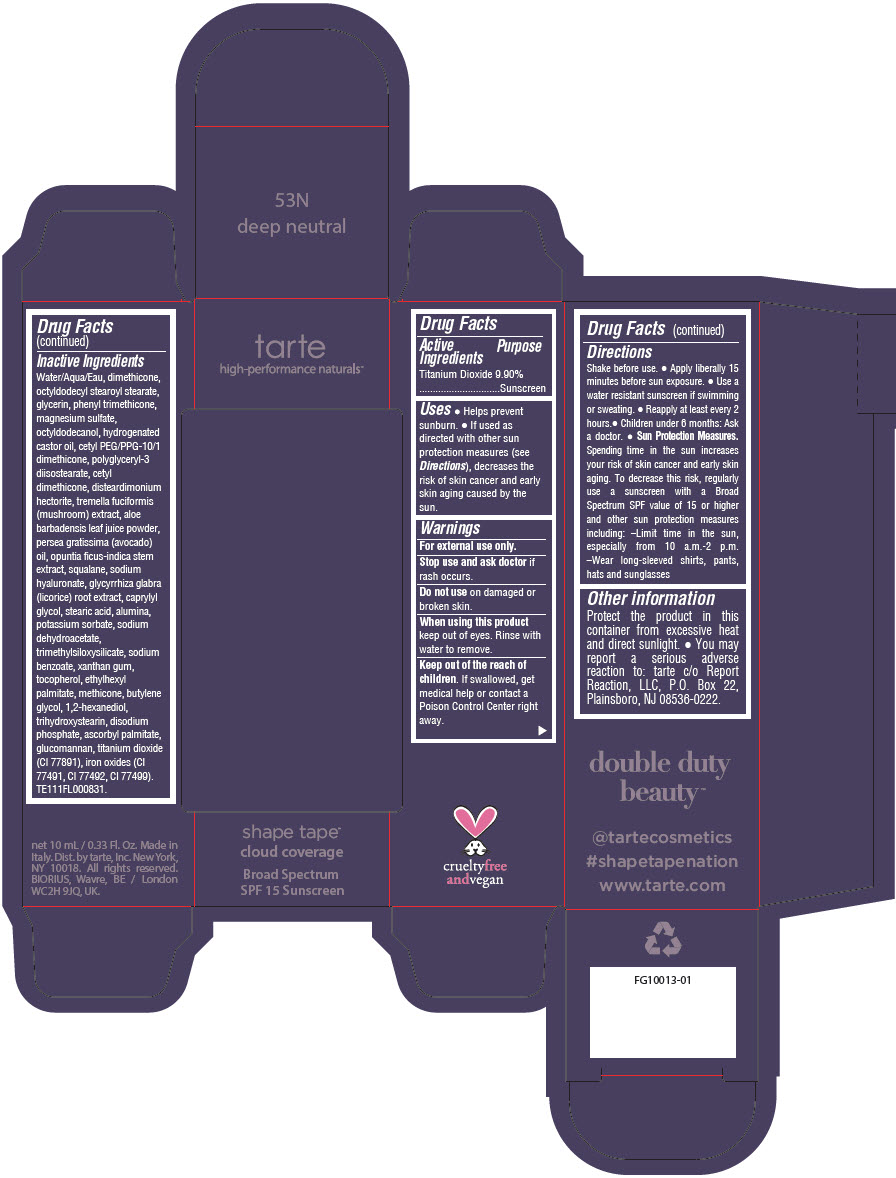
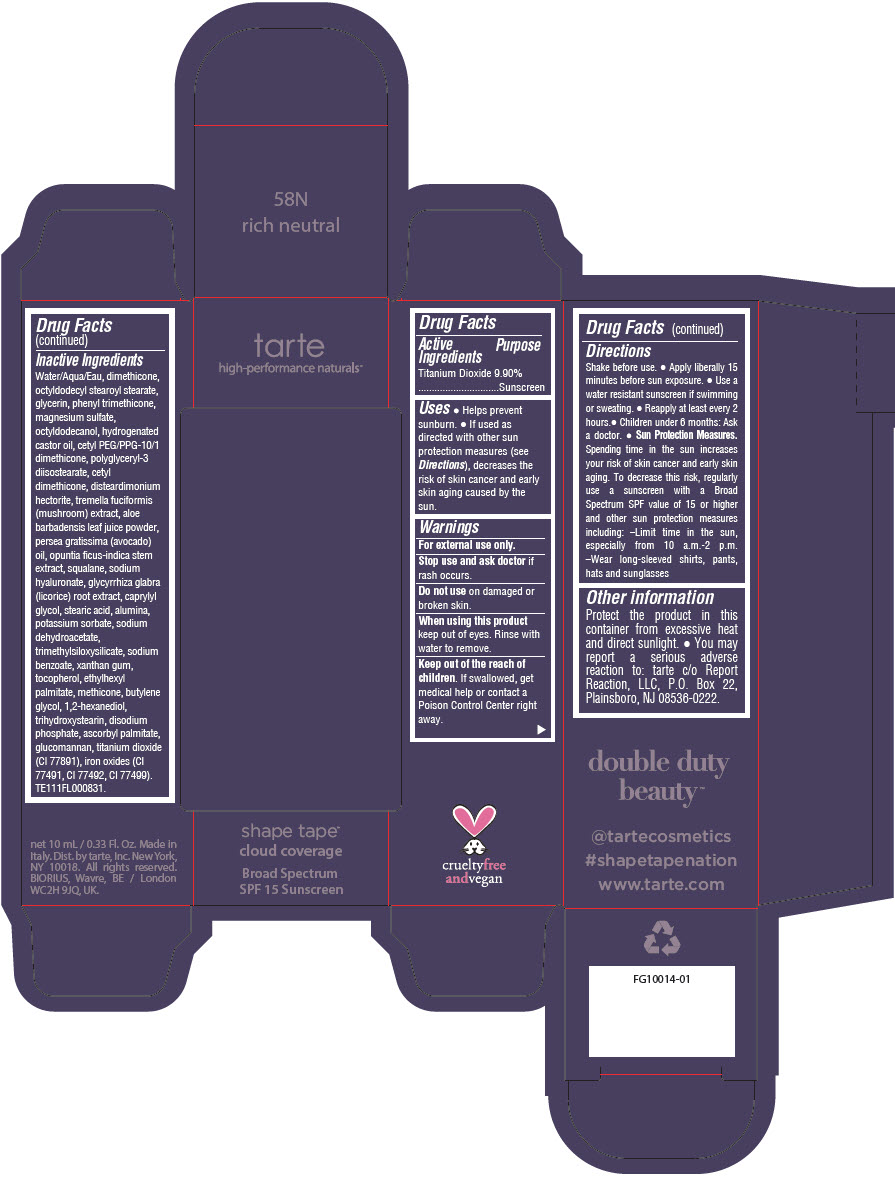
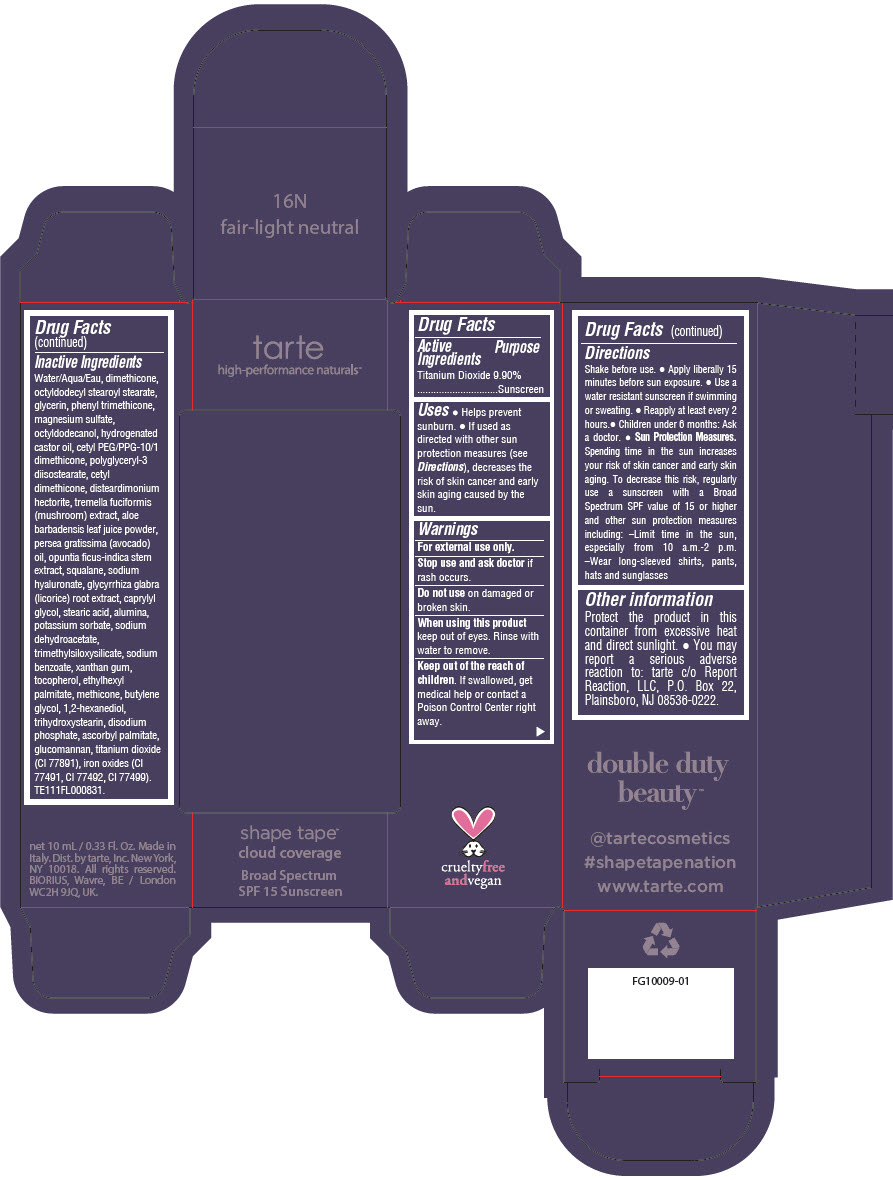
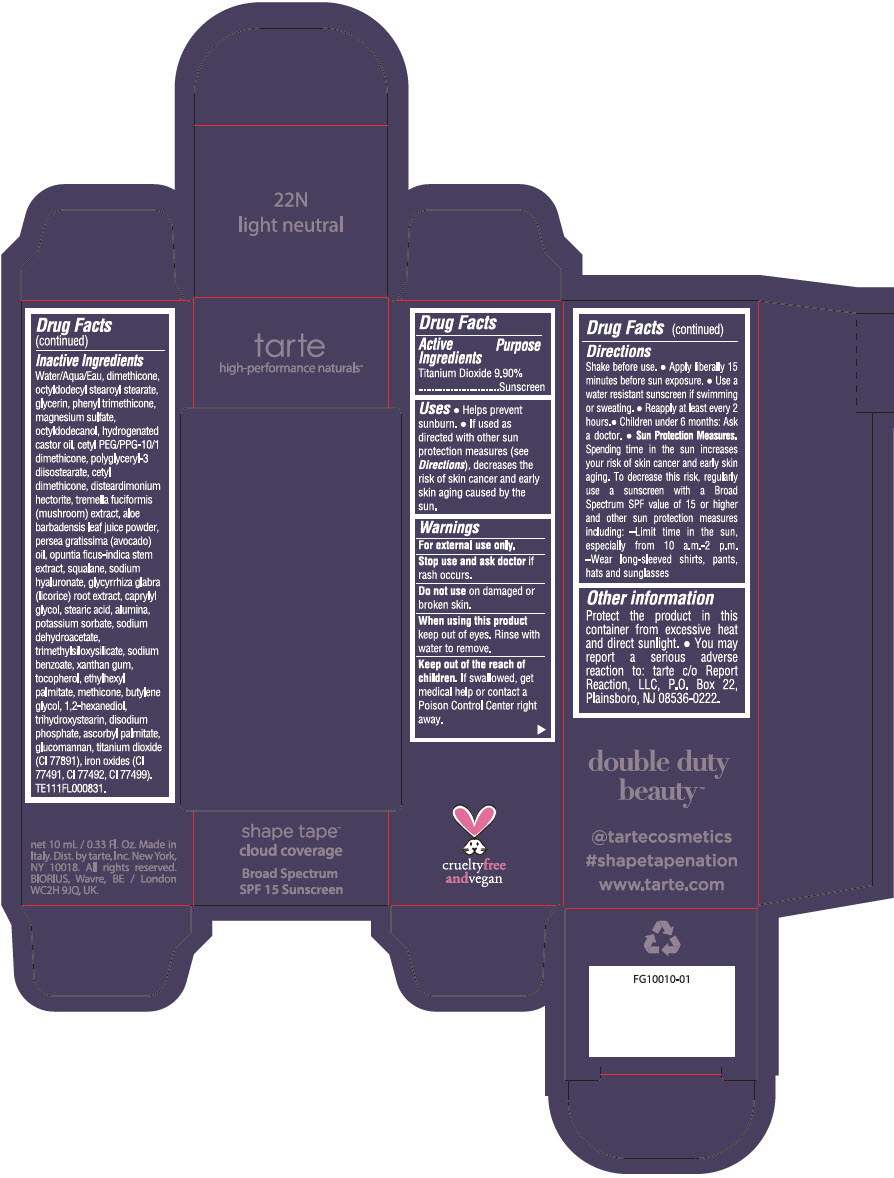
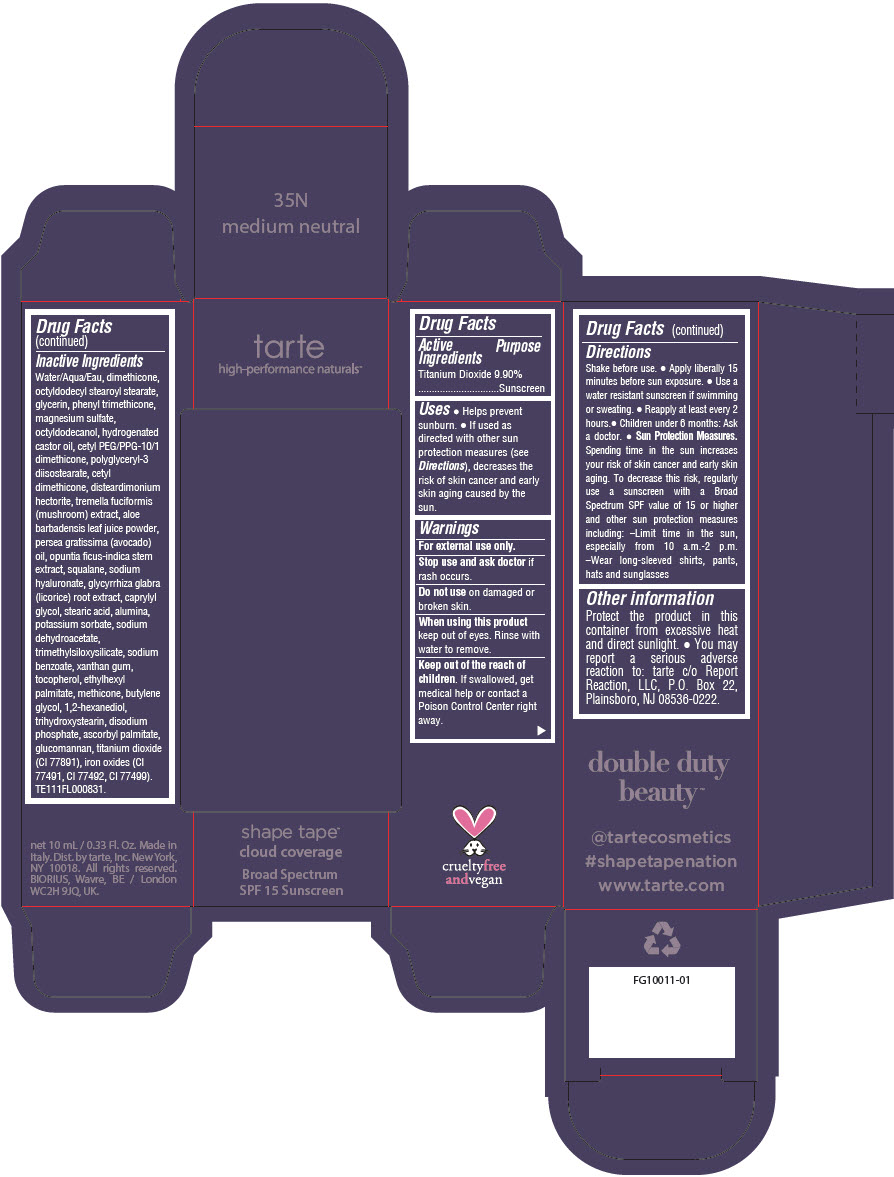
Uses
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
Directions
- Shake before use.
- Apply liberally 15 minutes before sun exposure.
- Use a water resistant sunscreen if swimming or sweating.
- Reapply at least every 2 hours.
- Children under 6 months: Ask a doctor.
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- –
- Limit time in the sun, especially from 10 a.m.-2 p.m.
- –
- Wear long-sleeved shirts, pants, hats and sunglasses
Other information
Protect the product in this container from excessive heat and direct sunlight.
- You may report a serious adverse reaction to: tarte c/o Report Reaction, LLC, P.O. Box 22, Plainsboro, NJ 08536-0222.
Inactive Ingredients
Water/Aqua/Eau, dimethicone, octyldodecyl stearoyl stearate, glycerin, phenyl trimethicone, magnesium sulfate, octyldodecanol, hydrogenated castor oil, cetyl PEG/PPG-10/1 dimethicone, polyglyceryl-3 diisostearate, cetyl dimethicone, disteardimonium hectorite, tremella fuciformis (mushroom) extract, aloe barbadensis leaf juice powder, persea gratissima (avocado) oil, opuntia ficus-indica stem extract, squalane, sodium hyaluronate, glycyrrhiza glabra (licorice) root extract, caprylyl glycol, stearic acid, alumina, potassium sorbate, sodium dehydroacetate, trimethylsiloxysilicate, sodium benzoate, xanthan gum, tocopherol, ethylhexyl palmitate, methicone, butylene glycol, 1,2-hexanediol, trihydroxystearin, sodium hyaluronate, disodium phosphate, ascorbyl palmitate, glucomannan, titanium dioxide (CI 77891), iron oxides (CI 77491, CI 77492, CI 77499). TE111FL000831.
PRINCIPAL DISPLAY PANEL - 10 mL Tube Carton - 16N Fair-Light Neutral
tarte
high-performance naturals™
shape tape™
cloud coverage
Broad Spectrum
SPF 15 Sunscreen
PRINCIPAL DISPLAY PANEL - 10 mL Tube Carton - 22N Light Neutral
tarte
high-performance naturals™
shape tape™
cloud coverage
Broad Spectrum
SPF 15 Sunscreen
PRINCIPAL DISPLAY PANEL - 10 mL Tube Carton - 35N Medium Neutral
tarte
high-performance naturals™
shape tape™
cloud coverage
Broad Spectrum
SPF 15 Sunscreen
PRINCIPAL DISPLAY PANEL - 10 mL Tube Carton - 44N Tan Neutral
tarte
high-performance naturals™
shape tape™
cloud coverage
Broad Spectrum
SPF 15 Sunscreen