FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Intralipid® is indicated as a source of calories and essential fatty acids for adult and pediatric patients requiring parenteral nutrition (PN) and as a source of essential fatty acids for prevention of essential fatty acid deficiency (EFAD).
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
- Intralipid is prepared and administered by a healthcare provider in the inpatient setting. Patients and caregivers may prepare and administer Intralipid for home use after appropriate training by a trained healthcare provider.
- Intralipid is for intravenous infusion into a central or peripheral vein.
- Do not exceed the recommended maximum infusion rate in Table 1 [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
- Intralipid admixtures with osmolarity
- Greater than or equal to 900 mOsm/L must be infused through a central vein.
- Less than 900 mOsm/L may be administered either through a central or peripheral vein.
- Use a 1.2 micron in-line filter during administration.
- Use a dedicated infusion line without any connections. Do not connect multiple medications in series.
- To prevent air embolism, use a non-vented infusion set or close the vent on a vented set and fully evacuate residual gas in the bag prior to administration.
- Do not pressurize the flexible bag to increase flow rates, and if administration is controlled by a pumping device, turn off the pump before the bag runs dry.
- Do not use infusion sets and lines that contain di-2-ethylhexyl phthalate (DEHP), including infusion sets that contain polyvinyl chloride (PVC) components, because they contain DEHP as a plasticizer.
- Intralipid can be infused concurrently into the same vein as dextrose-amino acid solutions (as part of PN) by a Y-connector located near the infusion site; flow rates of each solution should be controlled separately by infusion pumps.
- After connecting the infusion set, start infusion of Intralipid immediately. Complete the infusion within 12 hours when using a Y-connector and within 24 hours when used as part of an admixture.
2.2 Preparation Instructions
Use the following instructions to prepare single-dose 100 mL, 250 mL, and 500 mL Flexible containers for administration:
Intralipid 100 mL, 250 mL, and 500 mL single-dose Flexible Containers
- After removing the overpouch, infuse immediately. If not used immediately, the product should be stored at 2°C to 8°C (36°F to 46°F) for no longer than 24 hours. After removal from storage, infuse within 12 hours when using a Y-connector and within 24 hours when used as part of an admixture.
Intralipid 1,000 mL Pharmacy Bulk Package
- For admixing use only and not for direct intravenous infusion. Prior to administration, transfer to a separate PN container for individual patient use.
- Transfer the contents through the blue infusion port using a suitable sterile transfer device or dispensing set. Discard any unused contents.
- Use the Pharmacy Bulk Package immediately for admixing after removal from the overpouch. If not used immediately, the product can be stored for no longer than 24 hours at 2°C to 8°C (36°F to 46°F). After removal from storage, and once the closure is penetrated, use Pharmacy Bulk Package contents within 4 hours.
Admixing Instructions
- Prepare the admixture in PN containers using strict aseptic techniques to avoid microbial contamination.
- Do not add Intralipid to the PN container first; destabilization of the lipid may occur. The prime destabilizers of emulsions are excessive acidity (such as a pH <5) and inappropriate electrolyte content. Amino acid solutions exert buffering effects that protect the emulsion from destabilization. Give careful consideration to the addition of divalent cations (Ca++ and Mg++), which have been shown to cause emulsion instability.
- Do not inject additives directly into Intralipid.
- Intralipid may be mixed with amino acid and dextrose injections to produce “all-in-one” PN admixtures. The mixing sequence below must be followed for manual compounding to minimize pH-related problems by ensuring that typically acidic dextrose injections are not mixed with lipid emulsions alone; shake bags gently after each addition.
- Transfer dextrose injection to the PN container.
- Transfer amino acid injection.
- Transfer Intralipid.
- Simultaneous transfer of amino acid injection, dextrose injection, and Intralipid to the PN container is also permitted; follow automated compounding device instructions as indicated. Use gentle agitation during admixing to minimize localized concentration effects.
- Additions to the PN admixtures should be evaluated by a pharmacist for compatibility. Questions about compatibility may be directed to Baxter Healthcare.
- Inspect the admixture to ensure that precipitates have not formed during preparation of the admixture and the emulsion has not separated. Discard the admixture if any of the above are observed.
- Infuse admixtures containing Intralipid immediately. If not used immediately, store admixtures under refrigeration at 2°C to 8°C (36°F to 46°F) for no longer than 24 hours. Infusion must be complete within 24 hours after removal from refrigeration. Discard any remaining admixture.
- Protect the admixed PN solution from light.
2.3 Recommended Dosage and Administration
- The recommended nutritional requirements of lipid and recommended dosages of Intralipid to be administered to meet those requirements for adults and pediatric patients are provided in Table 1, along with recommendations for the initial and maximum infusion rates.
- The dosing of Intralipid depends on the patient's individual energy requirements influenced by age, body weight, tolerance, clinical status, and the ability to metabolize and eliminate lipids.
- When determining dose, energy supplied by dextrose and amino acids from PN, as well as energy from oral or enteral nutrition, has to be taken into account. Energy and lipid provided from lipid-based medications should also be taken into account (e.g., propofol).
- Prior to administration of Intralipid, correct severe fluid and electrolyte disorders and measure serum triglyceride levels to establish a baseline value. In patients with elevated triglyceride levels, initiate Intralipid at a lower dosage and titrate in smaller increments, monitoring the triglyceride levels with each adjustment [see Warnings and Precautions (5.7)].
|
* The neonatal period is defined as including term, post-term, and preterm neonates. The neonatal period for term and post-term neonates is the day of birth plus 27 days. For preterm neonates, the neonatal period is defined as the day of birth through the expected age of delivery plus 27 days (i.e., 44 weeks post-menstrual age). |
|||
|
** Daily dosage should also not exceed a maximum of 60% of total energy requirements [see Overdosage (10)]. |
|||
| Age | Nutritional Requirements | Direct Infusion Rate | |
| Recommended Initial Dosage and Maximum Dosage | Initial | Maximum | |
| Birth to 2 years of age (including preterm and term neonates*) [see Warnings and Precautions (5.1)] | Initial 0.5 g/kg/day not to exceed 3 g/kg/day** | 0.1 mL/kg/hour for the first 10 to 15 minutes; gradually increase to the required rate after 15 minutes | 0.75 mL/kg/hour |
| Pediatric patients 2 to <12 years of age | Initial 1 to 2 g/kg/day not to exceed 2.5 g/kg/day*** | 0.2 to 0.4 mL/kg/hour for the first 10 to 15 minutes; gradually increase to the required rate after 15 minutes | 0.75 mL/kg/hour |
| Pediatric patients 12 to 17 years of age | Initial 1 g/kg/day not to exceed 2 g/kg/day** | 0.2 mL/kg/hour for the first 10 to 15 minutes; gradually increase to the required rate after 15 minutes | 0.75 mL/kg/hour |
| Adults | 1 g/kg/day in stable patients ≤1 g/kg/day in critically ill patients not to exceed 2.5 g/kg/day; not more than 500 mL of Intralipid should be infused on the first day of therapy** | 0.2 mL/kg/hour for the first 10 to 15 minutes; gradually increase to the required rate after 30 minutes | 0.5 mL/kg/hour |
3 DOSAGE FORMS AND STRENGTHS
Intralipid 20% is a sterile, homogenous, milky, white lipid injectable emulsion in Flexible Containers supplied as:
- 20 g/100 mL (0.2 g/mL) of lipid in 100 mL single-dose Flexible Container
- 50 g /250 mL (0.2 g/mL) of lipid in 250 mL single-dose Flexible Container
- 100 g/500 mL (0.2 g/mL) of lipid in 500 mL single-dose Flexible Container
- 200 g /1,000 mL (0.2 g/mL) of lipid in 1,000 mL Pharmacy Bulk Package
5 WARNINGS AND PRECAUTIONS
5.1 Clinical Decompensation with Rapid Infusion of Intravenous Lipid Emulsions in Neonates and Infants
In the postmarketing setting, serious adverse reactions including acute respiratory distress, metabolic acidosis, and death have been reported in neonates and infants after rapid infusion of intravenous lipid emulsions. Hypertriglyceridemia was commonly reported.
Strictly adhere to the recommended total daily dosage; the hourly infusion rate should not exceed 0.75 mL/kg/hour [see Dosage and Administration (2.3)].
Preterm and small for gestational age infants have poor clearance of intravenous lipid emulsion and increased free fatty acid plasma levels following lipid emulsion infusion.
Carefully monitor the infant's ability to eliminate the infused lipids from the circulation (e.g., measure serum triglycerides and/or plasma free fatty acid levels). If signs or poor clearance of lipids from the circulation occur, stop the infusion and initiate a medical evaluation [see Warnings and Precautions (5.5, 5.7) and Overdosage (10)].
5.2 Parenteral Nutrition-Associated Liver Disease and Other Hepatobiliary Disorders
Risk of Parenteral Nutrition-Associated Liver Disease
Parenteral nutrition-associated liver disease (PNALD), also referred to as intestinal failure- associated liver disease (IFALD), can present as cholestasis or hepatic steatosis, and may progress to steatohepatitis with fibrosis and cirrhosis (possibly leading to chronic hepatic failure). The etiology of PNALD is multifactorial; however, intravenously administered phytosterols (plant sterols) contained in plant-derived lipid emulsions, including Intralipid, have been associated with development of PNALD.
In a randomized study of neonates and infants expected to be treated with PN for at least 28 days, parenteral nutrition-associated cholestasis (PNAC), a precursor to PNALD, developed more frequently in Intralipid-treated patients than patients treated with a 4-oil mixed lipid emulsion. [see Adverse Reactions (6.1), Use in Specific Populations (8.4)].
Monitor liver tests in patients treated with Intralipid and consider discontinuation or dosage reduction if abnormalities occur.
Other Hepatobiliary Disorders
Hepatobiliary disorders including cholecystitis and cholelithiasis have developed in some PN-treated patients without preexisting liver disease.
Monitor liver tests when administering Intralipid. Patients developing signs of hepatobiliary disorders should be assessed early to determine whether these conditions are related to Intralipid use.
5.3 Hypersensitivity Reactions
Intralipid contains soybean oil and egg phospholipids, which may cause hypersensitivity reactions. Cross reactions have been observed between soybean and peanut. Intralipid is contraindicated in patients with known hypersensitivity to egg, soybean, peanut or any of the active or inactive ingredients in Intralipid. If a hypersensitivity reaction occurs, stop infusion of Intralipid immediately and initiate appropriate treatment and supportive measures.
5.4 Infections
Parenteral nutrition, such as Intralipid, can support microbial growth and is an independent risk factor for the development of catheter-related bloodstream infections. To decrease the risk of infectious complications, ensure aseptic techniques are used for catheter placement, catheter maintenance, and preparation and administration of Intralipid.
Monitor for signs and symptoms of infection including fever and chills, as well as laboratory test results that might indicate infection (including leukocytosis and hyperglycemia). Perform frequent checks of the intravenous catheter insertion site for edema, redness, and discharge.
5.5 Fat Overload Syndrome
Fat overload syndrome is a rare condition that has been reported with intravenous lipid injectable emulsions and is characterized by a sudden deterioration in the patient's condition (e.g., fever, anemia, leukopenia, thrombocytopenia, coagulation disorders, hyperlipidemia, hepatomegaly, deteriorating liver function, and central nervous system manifestations such as coma). A reduced or limited ability to metabolize lipids, accompanied by prolonged plasma clearance (resulting in higher lipid levels), may result in this syndrome. Although fat overload syndrome has been most frequently observed when the recommended lipid dose or infusion rate was exceeded, cases have also been described when the lipid formulation was administered according to instructions.
If signs or symptoms of fat overload syndrome occur, stop the infusion of Intralipid. The syndrome is usually reversible when the infusion of the lipid emulsion is stopped.
5.6 Refeeding Syndrome
Administering PN to severely malnourished patients may result in refeeding syndrome, which is characterized by the intracellular shift of potassium, phosphorus, and magnesium as patients become anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, closely monitor severely malnourished patients and slowly increase their nutrient intake.
5.7 Hypertriglyceridemia
The use of Intralipid is contraindicated in patients with hypertriglyceridemia with serum triglyceride concentrations >1,000 mg/dL.
Patients with conditions such as inherited lipid disorders, obesity, diabetes mellitus, or metabolic syndromes have a higher risk of developing hypertriglyceridemia with the use of Intralipid. In addition, patients with hypertriglyceridemia may have worsening of their hypertriglyceridemia with administration of Intralipid. Excessive dextrose administration may further increase such risk.
Evaluate patients' capacity to metabolize and eliminate the infused lipid emulsion by measuring serum triglycerides before the start of infusion (baseline value) and regularly throughout treatment. If triglyceride levels are above 400 mg/dL in adults, stop the Intralipid infusion and monitor serum triglyceride levels to avoid clinical consequences of hypertriglyceridemia such as pancreatitis. In pediatric patients with hypertriglyceridemia, lower triglyceride levels (i.e., below 400 mg/dL) may be associated with adverse reactions. Monitor serum triglyceride levels to avoid potential complications with hypertriglyceridemia such as pancreatitis, lipid pneumonitis, and neurologic changes, including kernicterus.
To minimize the risk of new or worsening of hypertriglyceridemia, assess high-risk patients for their overall energy intake including other sources of lipids and dextrose, as well as concomitant drugs that may affect lipid and dextrose metabolism.
5.8 Aluminum Toxicity
Intralipid contains no more than 25 mcg/L of aluminum. Prolonged PN administration in patients with renal impairment may result in aluminum reaching toxic levels. Preterm neonates are at greater risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions that contain aluminum.
Patients with impaired kidney function, including preterm neonates, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day can accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading in these patients may occur at even lower rates of administration.
5.9 Monitoring/Laboratory Tests
Monitor fluid status closely in patients with pulmonary edema or heart failure.
Throughout treatment, monitor serum triglycerides [see Warnings and Precautions (5.7)], essential fatty acids, fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count (including platelets), and coagulation parameters.
The lipids contained in Intralipid may interfere with some laboratory tests (e.g., hemoglobin, lactate dehydrogenase, bilirubin, oxygen saturation) if blood is sampled before lipids have cleared from the bloodstream. Conduct these tests at least 6 hours after stopping the infusion.
Intralipid contains vitamin K that may counteract anticoagulant activity [see Drug Interactions (7)].
6 ADVERSE REACTIONS
Adverse reactions described elsewhere in this Prescribing Information are:
- Clinical Decompensation with Rapid Infusion of Intravenous Lipid Emulsion in Neonates and Infants [see Warnings and Precautions (5.1)]
- Parenteral Nutrition-Associated Liver Disease and Other Hepatobiliary Disorders [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Infections [see Warnings and Precautions (5.4)]
- Fat Overload Syndrome [see Warnings and Precautions (5.5)]
- Refeeding Syndrome [see Warnings and Precautions (5.6)]
- Hypertriglyceridemia [see Warnings and Precautions (5.7)]
- Aluminum Toxicity [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Intralipid or equivalent soybean oil lipid emulsions functioned as the comparator in trials of the 4-oil mixed lipid emulsion [see Clinical Studies (14)]. The adverse reactions from these studies are included to present the clinical experience with Intralipid.
The safety database for Intralipid or equivalent soybean oil lipid emulsion exposure in these studies includes 393 patients (230 adults; 163 pediatric) in 9 clinical trials. Adult patients were exposed for 5 days to 4 weeks in 5 clinical trials. Intralipid or equivalent soybean oil lipid emulsion was used as a component of PN which also included dextrose, amino acids, vitamins, and trace elements. Two of the 5 studies in adults were performed with Intralipid as a component of PN delivered in a 3-chamber bag.
| Adverse Reaction | Number of Patients in Soybean Oil Lipid Emulsion Group (N=230) | Number of Patients in 4-Oil Mixed Lipid Emulsion Comparator Group (N=229) |
|---|---|---|
| Nausea | 26 (11%) | 20 (9%) |
| Vomiting | 12 (5%) | 15 (7%) |
| Pyrexia | 11 (5%) | 9 (4%) |
| Hypertension | 9 (4%) | 6 (3%) |
| Headache | 7 (3%) | 3 (1%) |
| Hyperglycemia | 5 (2%) | 12 (5%) |
| Abdominal pain | 5 (2%) | 8 (4%) |
| Flatulence | 4 (2%) | 10 (4%) |
| Blood triglycerides increased | 4 (2%) | 6 (3%) |
| Sepsis | 4 (2%) | 5 (2%) |
| Diarrhea | 4 (2%) | 3 (1%) |
| Pneumonia | 4 (2%) | 3 (1%) |
| Pruritus | 4 (2%) | 3 (1%) |
| Gamma-glutamyltransferase increased | 4 (2%) | 2 (1%) |
Less common adverse reactions occurring in ≤1% of adult patients who received Intralipid or equivalent soybean oil lipid emulsion were dyspepsia, urinary tract infection, anemia, infection, dyspnea, cholestasis, dysgeusia, increased blood alkaline phosphatase, tachycardia, liver function test abnormalities, dizziness, rash, and thrombophlebitis.
The 163 patients treated with Intralipid in four pediatric trials consisted of 147 patients <28 days of age, 9 patients 28 days to <2 years of age, and 7 patients 2 to 7 years of age; the duration of exposure was 7 to 84 days. Fifty-six percent of the pediatric patients were female, and 85% were Caucasian. Most pediatric patients were preterm neonates with feeding intolerance or other conditions requiring short-term (<29 days) PN.
| Adverse Reaction | Number of Patients in Intralipid Group (N=163) | Number of Patients in 4-Oil Mixed Lipid Emulsion Comparator Group (N=170) |
|---|---|---|
| Anemia | 33 (20%) | 30 (18%) |
| Vomiting | 16 (10%) | 16 (9%) |
| Gamma-glutamyltransferase increased | 12 (7%) | 10 (6%) |
| Cholestasis | 10 (6%) | 7 (4%) |
| Pyrexia | 7 (4%) | 7 (4%) |
| C-reactive protein increased | 7 (4%) | 6 (4%) |
| Hyperbilirubinemia | 7 (4%) | 5 (3%) |
| Bilirubin conjugated increased | 7 (4%) | 3 (2%) |
| Nosocomial infection | 6 (4%) | 10 (6%) |
| Blood alkaline phosphatase increased | 6 (4%) | 1 (1%) |
| Abdominal pain | 5 (3%) | 4 (2%) |
| Hematocrit decreased | 5 (3%) | 2 (1%) |
| Metabolic acidosis | 5 (3%) | 2 (1%) |
| Diarrhea | 4 (3%) | 3 (2%) |
| Tachycardia | 4 (3%) | 3 (2%) |
| Thrombocytopenia | 4 (3%) | 3 (2%) |
| Alanine aminotransferase increased | 3 (2%) | 1 (1%) |
| Aspartate aminotransferase increased | 3 (2%) | 0 (0%) |
| Parenteral nutrition-associated liver disease | 3 (2%) | 0 (0%) |
Less common adverse reactions occurring in ≤1% of pediatric patients who received Intralipid were hyperglycemia, sepsis, increased blood triglycerides, infection, fluid overload, hypertension, hypertriglyceridemia, rash, and hyperlipidemia.
In a randomized active-controlled, double-blind, parallel-group, multi-center study that included 152 neonates and 9 patients ranging in age from 29 to 153 days who were expected to require PN for at least 28 days, PNAC, a precursor to PNALD, developed more frequently in Intralipid-treated patients than in patients treated with a comparator 4-oil mixed lipid emulsion.
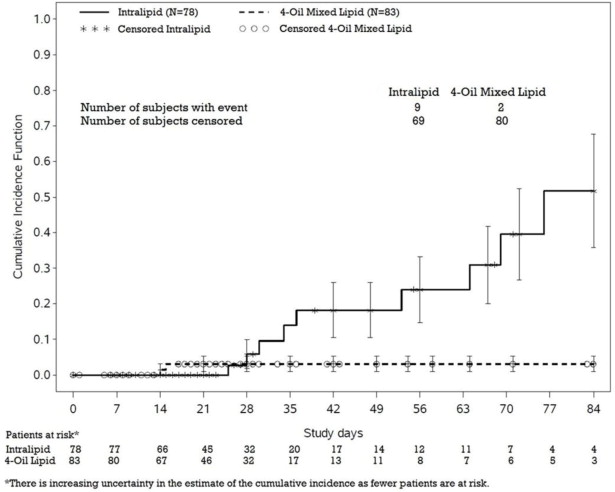
PNAC (defined as direct bilirubin >2 mg/dL with a second confirmed elevation >2 mg/dL at least 7 days later) occurred in 11.5% (9/78) in Intralipid-treated patients and 2.4% (2/83) of patients treated with a 4-oil mixed lipid emulsion. Most PNAC events occurred in patients who were treated for longer than 28 days.
The estimated cumulative incidence of PNAC is shown in the Kaplan-Meier cumulative incidence curve in Figure 1 [see Pediatric Clinical Studies (14.2)].
Figure 1: Cumulative Incidence Curve of Time to Parenteral Nutrition-Associated Cholestasis (PNAC) with Standard Error Bars
6.2 Postmarketing Experience
The following adverse reactions from voluntary reports have been reported with Intralipid. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: palpitations
Gastrointestinal disorders: vomiting, nausea
General disorders and administration site conditions: chills, chest discomfort, pyrexia
Nervous system disorders: dizziness
Respiratory, thoracic, and mediastinal disorders: dyspnea
Immune system disorders: hypersensitivity
Vascular disorders: phlebitis
Blood and lymphatic system disorders: hypercoagulability
7 DRUG INTERACTIONS
Soybean oil in Intralipid contains vitamin K1 which may counteract the anticoagulant activity of vitamin K antagonists such as warfarin. In patients who receive concomitant Intralipid and warfarin, increase monitoring of laboratory parameters for anticoagulant activity.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Administration of the recommended dose of Intralipid is not expected to cause major birth defects, miscarriage, or other adverse maternal or fetal outcomes. No animal reproduction studies have been conducted with Intralipid. There are risks to the fetus associated with severe malnutrition during pregnancy (see Clinical Considerations).
The background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo-Fetal Risk
Severe malnutrition in pregnant women is associated with preterm delivery, low birth weight, intrauterine growth restriction, congenital malformations, and perinatal mortality. Parenteral nutrition should be considered if the pregnant woman's nutritional requirements cannot be fulfilled by oral or enteral intake.
8.2 Lactation
Risk Summary
Administration of the recommended dose of Intralipid is not expected to cause harm to a breastfed infant. There are no data on the presence of Intralipid in human or animal milk or its effects on milk production. Available published literature includes fewer than five reported cases of breastfed infants exposed to various lipid emulsions via lactation, and these cases did not report adverse events. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Intralipid and any potential adverse effects of Intralipid on the breastfed infant, or from the underlying maternal condition.
8.4 Pediatric Use
Intralipid is contraindicated in pediatric patients with severe disorders of lipid metabolism [(see Contraindications (4)].
The safety and effectiveness of Intralipid have been established as a source of calories and essential fatty acids for PN in pediatric patients, including term and preterm neonates. Use of Intralipid in neonates is supported by evidence from short-term (i.e., 1- to 4- week) studies, and one study following neonates beyond 4 weeks [see Clinical Studies (14.2)]. Use of Intralipid in older pediatric patients is supported by evidence from short-term (i.e., <28 days) studies in pediatric patients 28 days to 12 years of age and additional evidence from studies in adults [see Clinical Studies (14)]. The most common adverse reactions in Intralipid-treated pediatric patients were anemia, vomiting, gamma-glutamyltransferase increased, and cholestasis. PNAC, a precursor to PNALD, developed more frequently in Intralipid-treated patients than in patients treated with a comparator 4-oil mixed lipid emulsion [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
In the postmarketing setting, clinical decompensation with rapid infusion of intravenous lipid emulsion in neonates and infants, sometimes fatal, has been reported [see Warnings and Precautions (5.1)]. Because of immature renal function, preterm neonates receiving prolonged treatment with Intralipid may be at risk for aluminum toxicity [see Warnings and Precautions (5.8)].
8.5 Geriatric Use
Reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
10 OVERDOSAGE
In the event of an overdose, serious adverse reactions may result [see Warnings and Precautions (5.1, 5.5)]. Stop the infusion of Intralipid until triglyceride levels have normalized and symptoms have abated. The effects are usually reversible by stopping the lipid infusion. If medically appropriate, further intervention may be indicated. Lipids are not dialyzable from plasma.
11 DESCRIPTION
Intralipid is a sterile, non-pyrogenic, white, homogenous lipid emulsion for intravenous infusion as a source of calories and essential fatty acids. The lipid content of Intralipid is 0.2 g/mL and comprises soybean oil. The phosphate content is 15 mmol/L.
The total energy content, including fat, phospholipids, and glycerin is 2,000 kcal/L.
Each 100 mL of Intralipid contains approximately 20 g soybean oil, 1.2 g egg yolk phospholipids, 2.25 g glycerin, water for injection, and sodium hydroxide for pH adjustment (pH 6 to 8.9). Intralipid has an osmolality of approximately 350 mOsmol/kg water (which represents an osmolarity of 260 mOsmoL/L).
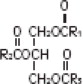
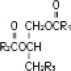
The soybean oil is a refined natural product consisting of a mixture of neutral triglycerides of predominantly unsaturated fatty acids with the following structure:
where 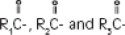 are saturated and unsaturated fatty acid residues.
are saturated and unsaturated fatty acid residues.
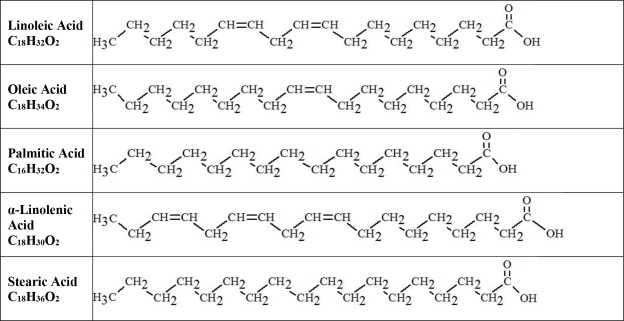
The major component fatty acids in Intralipid are linoleic acid (44% to 62%), oleic acid (19% to 30%), palmitic acid (7% to 14%), alpha-linolenic acid (4% to 11%), and stearic acid (1.4% to 5.5%). These fatty acids have the following chemical and structural formulas:
Purified egg phosphatides are a mixture of naturally occurring phospholipids which are isolated from the egg yolk. These phospholipids have the following general structure:
 contain saturated and unsaturated fatty acids that abound in neutral fats. R3 is primarily either the choline or ethanolamine ester of phosphoric acid.
contain saturated and unsaturated fatty acids that abound in neutral fats. R3 is primarily either the choline or ethanolamine ester of phosphoric acid.
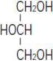
Glycerin is chemically designated C3H8O3 and is a clear colorless, hygroscopic syrupy liquid. It has the following structural formula:
The container-solution unit is a closed system and is not dependent upon entry of external air during administration. The container is overwrapped to provide protection from the physical environment and to provide an additional oxygen and moisture barrier when necessary.
Intralipid contains no more than 25 mcg/L of aluminum.
The container is not made with natural rubber latex, PVC, or DEHP.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Intralipid provides a biologically utilizable source of calories and essential fatty acids.
Fatty acids serve as an important substrate for energy production. The most common mechanism of action for energy production derived from fatty acid metabolism is beta oxidation. Fatty acids are also important for membrane structure and function, as precursors for bioactive molecules (such as prostaglandins), and as regulators of gene expression.
12.3 Pharmacokinetics
Intralipid provides fatty acids in the form of triglycerides which are hydrolyzed by lipoprotein lipase to release free fatty acids. Linoleic acid and alpha-linolenic acid are metabolized within a common biochemical pathway through a series of desaturation and elongation steps.
14 CLINICAL STUDIES
Intralipid or equivalent soybean oil lipid emulsion functioned as the comparator for the 4-oil mixed lipid emulsion in the clinical studies described in sections 14.1 and 14.2. The trial results are included to present the clinical experience with Intralipid.
14.1 Adult Clinical Studies
The efficacy of Intralipid or equivalent soybean oil lipid emulsion compared to a 4-oil mixed lipid emulsion was evaluated in 3 clinical studies in adult patients. Nutritional efficacy in adult studies was assessed by changes in anthropometric indices (body weight, height, and body mass index [BMI]), changes in lipid and protein metabolism (albumin), and fatty acid parameters. Of the 354 adult patients (178 Intralipid; 176 comparator), 62% were male, 99% were Caucasian, and ages ranged from 19 to 96 years. All patients received Intralipid/equivalent soybean oil lipid emulsion or the comparator as part of a PN regimen. Although Adult Study 1, Adult Study 2, and Adult Study 3 were not designed for formal statistical comparisons between Intralipid/equivalent soybean oil lipid emulsion and the comparator, they support Intralipid as a source of calories and essential fatty acids in adults. The lipid dosage was variable in these studies and adjusted to the patient's nutritional requirements.
Adult Study 1 was a double-blind, randomized, active-controlled, parallel-group, multicenter study in patients who required PN for at least 28 days. Seventy-five patients were enrolled, and 73 patients were treated with either Intralipid or the comparator. Changes in mean triglyceride levels from baseline values to Week 4 were similar in both the Intralipid and comparator groups. Mean albumin levels demonstrated a comparable decrease in both groups. Mean changes in body weight (kg) and BMI (kg/m2) were similar in both the Intralipid and the comparator groups.
Adult Study 2 was a phase 3, randomized, double-blind, active-controlled, multicenter study. A total of 249 postoperative adult patients were randomized to receive either an equivalent soybean oil lipid emulsion to Intralipid or the comparator for at least 5 days as part of their total parenteral nutrition (TPN) regimen. From baseline to Day 6, mean triglyceride levels increased similarly in both the soybean oil lipid emulsion and the comparator groups.
Adult Study 3 was a double-blind randomized, active-controlled, parallel-group, single-center study in 32 adult patients who required TPN for 10 to 14 days. Patients were treated with either an equivalent soybean oil lipid emulsion to Intralipid or the comparator. The increase in mean triglyceride levels from baseline to the final assessment was similar in both the soybean oil lipid emulsion and the comparator groups.
14.2 Pediatric Clinical Studies
The efficacy of Intralipid compared to a 4-oil mixed lipid emulsion in pediatric patients of all age groups, including term and preterm neonates, was evaluated in 333 patients in 4 randomized active-controlled, double-blind, parallel-group controlled clinical studies. Although Pediatric Studies 1, 2, 3, and 4 were not designed for formal statistical comparisons between Intralipid and the comparator, they support Intralipid as a source of calories and essential fatty acids in pediatric patients. The 333 pediatric patients (163 Intralipid; 170 comparator) consisted of 296 patients who were <28 days old, 22 patients 29 days to <2 years old, and 15 patients 2 to <12 years old. Fifty percent of the pediatric patients were male and 87% were Caucasian. All patients received Intralipid or the comparator as part of a PN regimen. Nutritional efficacy in neonates was assessed by changes in anthropometric indices (body weight, height, head circumference). Nutritional efficacy in pediatric patients, 28 days to 12 years of age, was assessed by changes in triglyceride concentrations and fatty acid parameters.
Pediatric Study 1 enrolled 152 preterm and term neonates (birth up to 28 days) and 9 patients ranging in age from 29 to 153 days. Patients were treated with either Intralipid (n=78) or the comparator (n=83). A total of 119 patients (58 Intralipid; 61 comparator) received study treatment for ≥14 days. A total of 27 patients received Intralipid for ≥29 days; 5 patients received Intralipid for the maximum study duration of 78-84 days.
Pediatric Studies 2 and 3 enrolled 60 and 84 preterm neonates, respectively, who were treated with either Intralipid or the comparator (72 neonates in each group). The median treatment duration for Intralipid group was 9 days in Pediatric Study 2 and 6 days in Pediatric Study 3.
Pediatric Study 4 enrolled 13 patients 5 months to <2 years of age and 15 patients 2 to 11.5 years of age. Patients were treated with either Intralipid (n=13) or the comparator (n=15) with a median treatment duration of 27 days.
In Pediatric Studies 1, 2 and 3, which enrolled neonates, Intralipid-treated patients showed increases in the median body weight, height/length, and head circumference (which was measured in Studies 1 and 3) comparable to the comparator-treated patients. Mean triglyceride levels from baseline to the final assessment in Pediatric Studies 1, 2, and 3 were variable in these neonates, but overall differences between groups were not considered clinically relevant. Mean triglyceride levels in Pediatric Study 4 were variable but remained within the normal range.
16 HOW SUPPLIED/STORAGE AND HANDLING
Intralipid 20% (lipid injectable emulsion, USP) is a sterile, homogeneous, milky, white lipid emulsion supplied in Flexible Containers as follows:
| Product Code | Unit of Use | Unit of Sale |
| 831800311 | NDC 65219-531-01 One 100 mL freeflex® bag | NDC 65219-531-10 Package of 10 freeflex® bags |
| 831818311 | NDC 65219-533-01 One 250 mL freeflex® bag | NDC 65219-533-25 Package of 10 freeflex® bags |
| 831826311 | NDC 65219-535-01 One 500 mL freeflex® bag | NDC 65219-535-50 Package of 12 freeflex® bags |
Store below 25°C (77°F). Avoid excessive heat. Do not freeze. If accidentally frozen, discard container. Store in the overpouch until ready for use.
Intralipid 100 mL, 250 mL and 500 mL single-dose Flexible Containers
After removing the overpouch, infuse immediately. If not used immediately, the product should be stored at 2°C to 8°C (36°F to 46°F) for no longer than 24 hours. After removal from storage, infuse within 12 hours when using a Y-connector or within 24 hours if used as part of an admixture [see Dosage and Administration (2.2)].
Intralipid 1,000 mL Pharmacy Bulk Package
Use the Pharmacy Bulk Package immediately for admixing after removal from the overpouch. If not used immediately, the product should be stored for no longer than 24 hours at 2°C to 8°C (36°F to 46°F). After removal from storage, and once the closure is penetrated, use Pharmacy Bulk Package contents within 4 hours [see Dosage and Administration (2.2)].
Admixtures
Infuse admixtures containing Intralipid immediately. If not used immediately, admixtures should be stored at 2°C to 8°C (36°F to 46°F) for no longer than 24 hours. After removal from storage, infuse within 24 hours [see Dosage and Administration (2.2)].
Protect the admixed PN solution from light [see Dosage and Administration (2.2)].
17 PATIENT COUNSELING INFORMATION
When initiating Intralipid administration, discuss the following information with the patient or caregiver:
Clinical Decompensation with Rapid Infusion of Intravenous Lipid Emulsion in Neonates and Infants
Inform caregivers that acute respiratory distress and death may occur in neonates and infants after rapid infusion of intravenous lipid emulsions. If Intralipid is infused at home, instruct caregivers not to exceed the maximum infusion rate [see Warnings and Precautions (5.1)].
Parenteral Nutrition-Associated Liver Disease and Other Hepatobiliary Disorders
Inform patients and caregivers that use of parenteral nutrition may result in parenteral nutrition- associated liver disease and/or other hepatobiliary disorders [see Warnings and Precautions (5.2)].
Hypersensitivity Reactions
Inform patients and caregivers that Intralipid may cause hypersensitivity reactions. If Intralipid is infused at home, instruct patients or caregivers to stop the infusion of Intralipid immediately and seek medical attention if a hypersensitivity reaction occurs, [see Warnings and Precautions (5.3)].
Infections
Inform patients and caregivers that patients who receive Intralipid are at risk of infection. If Intralipid is infused at home, instruct patients or caregivers to ensure aseptic techniques are used for the preparation and administration of Intralipid and to monitor for signs and symptoms of infection [see Warnings and Precautions (5.4)].
Fat Overload Syndrome
Inform patients and caregivers that fat overload syndrome has been reported with the use of intravenous lipid emulsions. If Intralipid is infused at home, instruct patients or caregivers to stop the infusion of Intralipid if signs or symptoms of fat overload syndrome occur [see Warnings and Precautions (5.5)].
Refeeding Syndrome
If the patient is severely malnourished, inform patients and caregivers that administering parenteral nutrition including Intralipid may result in refeeding syndrome [see Warnings and Precautions (5.6)].
Hypertriglyceridemia
Inform patients and their caregivers about the risks of hypertriglyceridemia with Intralipid use [see Warnings and Precautions (5.7)].
Aluminum Toxicity
Inform patients and their caregivers that prolonged PN administration in patients with renal impairment, including preterm neonates, may result in aluminum reaching toxic levels associated with central nervous system and bone toxicity [see Warnings and Precautions (5.8)].
Preparation and Administration Instructions
If it is acceptable for a patient or caregiver to administer Intralipid at home, then provide recommendations on how to inspect and prepare, add compatible additives (when appropriate), administer, and store Intralipid [see Dosage and Administration (2.1, 2.2)]. Inform patients or caregivers not to deviate from the administration instructions given by the healthcare provider.
Manufactured by:
Uppsala, Sweden
Intralipid® is a registered trademark of Fresenius Kabi AB.
www.fresenius-kabi.com/us
451713B
PACKAGE LABEL – PRINCIPAL DISPLAY – Intralipid 100 mL Bag Label
NDC 65219-531-01
Intralipid® 20% 100 ml
A 20% I.V. Fat Emulsion
100 mL
For Intravenous Use
Rx only
PACKAGE LABEL – PRINCIPAL DISPLAY – Intralipid 100 mL Shipper Label
NDC 65219-531-10
Intralipid® 20%
A 20% I.V. Fat Emulsion
100 mL
For Intravenous Use
10 x 100 mL
PACKAGE LABEL - PRINCIPAL DISPLAY - Intralipid 250 mL Bag Label
NDC 65219-533-01
Intralipid ® 20% 250 ml
A 20% I.V. Fat Emulsion
250 mL
For Intravenous Use
Rx only
PACKAGE LABEL – PRINCIPAL DISPLAY – Intralipid 250 mL Shipper Label
NDC 65219-533-25
Intralipid® 20%
A 20% I.V. Fat Emulsion
250 mL
For Intravenous Use
10 x 250 mL